Implant-based breast reconstruction following conservative mastectomy: one-stage vs. two-stage approach
Conservative mastectomy has become the newest option in the armamentarium of oncoplastic surgery. Conservative mastectomy is defined as preservation of the entire skin envelope including the nipple areolar complex. Other commonly referred to names for this procedure include nipple sparing mastectomy and total skin sparing mastectomy. This can be performed for therapeutic as well as prophylactic indications (1,2). The benefit of this approach is that reconstructive outcomes are optimized as breast volume, contour, and appearance are usually maintained or enhanced. Reconstruction can be performed using prosthetic devices or autologous tissues. In the United States, approximately 80% of reconstructions are performed using prosthetic devices, with the vast majority performed immediately at the time of mastectomy (3,4).
One of the controversies associated with prosthetic reconstruction is whether to perform the reconstruction in 1 stage (direct to implant) or 2 stages (tissue expander/implant). Advocates for the 1 stage technique emphasize a low revision rate, fewer operations, reduced overall cost, and excellent patient outcomes (5-8). Advocates for the 2 stage technique emphasize improved patient outcomes based on recontouring and selecting an ideal device for the second stage, reduced capsular contracture in the setting of post mastectomy radiation, a lower unplanned revision rate, and excellent patient outcomes (9,10). Success with either technique is ultimately based on proper patient selection, surgical technique, and surgeon experience.
In a multi-institutional study evaluating short-term outcomes following 1,528 1 stage and 9,033 2 stage reconstructions, Davila et al. demonstrated a higher incidence of complications following 1 stage (6.8% vs. 5.4%, P=0.02) and higher failure following 1 stage (1.4% vs. 0.8%, P=0.04) (11). There were no differences between 1 and 2 stage reconstructions with regard to surgical site infections (3.9% vs. 3.4%, P=0.34) or reoperation (7.5% vs. 6.9%, P=0.4) rates. Roostaeian et al. in review of a single institutions experience comparing outcomes following 1- and 2-stage prosthetic reconstruction demonstrated no differences with respect to complication rates, need for revision, and aesthetic outcomes (12). One stage reconstruction did result in fewer office visits and less time to completion.
In a large single institution study, the differences in complications between conservative and skin sparing mastectomy were evaluated in 233 cases (13). Nipple sparing mastectomy was performed in 113 cases and skin-sparing mastectomy was performed in 120 cases. The overall complication rate was 28% following nipple sparing and 27% following skin sparing. Of interest, in patients that had risk-reducing mastectomy (without axillary procedures), the complication rate was higher in the nipple-sparing cohort (26% vs. 9%, P=0.06) compared to the skin-sparing cohort.
Patient selection
Patient selection is an important factor when considering nipple sparing mastectomy, prosthetic reconstruction, and 1 or 2 stage techniques (14). In general, conservative mastectomy is considered for women with smaller tumors (<3 cm in diameter) that are more than 3 cm from the nipple areolar complex. Conservative mastectomy is also dependent upon breast size. Women with larger breasts are often not considered suitable candidates for conservative mastectomy because the vascularity to the nipple areolar complex may be compromised and may become necrotic.
Prosthetic reconstruction can be considered in the majority of women having skin or nipple sparing mastectomy. Ideal patients for prosthetic reconstruction include women of virtually any size breast, unilateral or bilateral cases, as well as women considering immediate or delayed reconstruction. Poor candidates for prosthetic reconstruction often include women that have had prior radiation therapy, morbidly obese patients, and patient that are actively smoking tobacco products. The decision regarding skin vs. nipple sparing mastectomy is dependent on patient selection with larger volumes usually having skin sparing techniques. The ability to achieve symmetry is another important consideration. Secondary procedures are more common following prosthetic reconstruction and may involve the ipsilateral or contralateral breast (15). In the setting of bilateral reconstruction, the specific characteristics of the breast are less important because the two reconstructed breasts will be very similar.
Immediate 1-stage reconstruction following conservative mastectomy
The staging of prosthetic reconstruction as a 1-stage or 2-stage procedure in the setting of conservative mastectomy depends on patient and breast characteristics as well as the quality of the mastectomy. Direct to implant reconstruction is sometimes considered in women with small to moderate breast volume with a cup size ranging from A-C (16). With these patients, the mastectomy should meet certain specifications that include adequate thickness of the skin flaps, no to minimal undermining of the regions outside the breast, and retention of a meniscus of fat along the medial and inframammary folds. These maneuvers will increase the likelihood of natural shape an contour without clefts or folds in the breast.
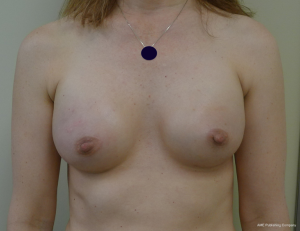
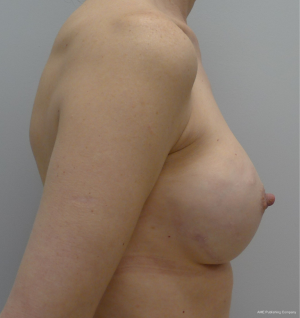
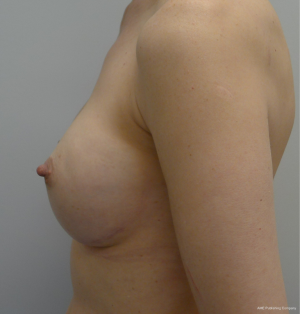
The technique of 1-stage reconstruction in the setting of conservative mastectomy has been previously described in detail (6,17). The salient points will be reviewed in this section. Mastectomy incisions can be created lateral to the areola, periareolar, along the inframammary fold, and via an inverted T approach. Munhoz et al. has demonstrated that complications related to delay healing are increased in patients having hemiareolar and inverted T incisional patterns (18). Regardless of the incisional pattern, the length of the incision should be adequate to perform a complete mastectomy. Suboptimal mastectomy in the setting of conservative mastectomy can increase the incidence of local recurrence (19). Sentinel lymph node biopsy can be performed through a separate incision near the axilla or via the laterally based incision if used. A subareolar biopsy is usually obtained. The permanent implant is selected based on the external and internal base diameter of the breast as well as mastectomy weight. Round or shaped devices can be used based on the breast and patient characteristics as well as patient desire (20). The selected devices are usually silicone gel, but saline devices can also be considered. The device can be placed in a variety of locations that include prepectoral, total subpectoral or partial subpectoral. Acellular dermal matrices are often used for prepectoral and partial subpectoral but are not usually necessary for subpectoral. Adequate compartmentalization of the device is necessary to ensure that the device does not migrate laterally or inferiorly. With shaped devices, proper orientation and compartmentalization is critical to ensure that the device does not rotate. Once the device has been properly positioned and secured, the mastectomy skin envelope is carefully redraped in order to prevent malposition of the nipple areolar complex. A closed suction drain is used in all cases. Figures 1-4 demonstrate a woman that had immediate direct to implant reconstruction following conservative mastectomy.

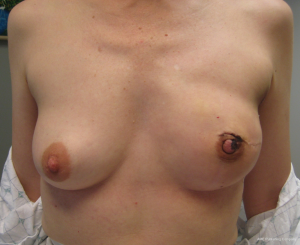
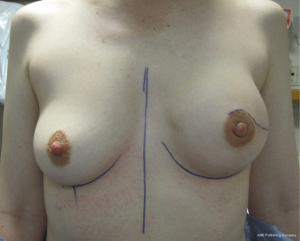
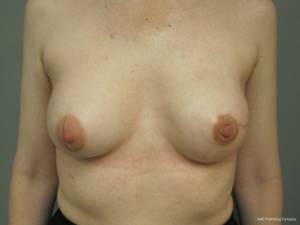
Outcomes following 1-stage reconstruction in the setting of conservative mastectomy have been favorable; however the unplanned secondary revision rate may be higher when compared to the two-stage technique. Over an 8-year follow-up period, Salzberg et al. has demonstrated consistently low complications rates with excellent aesthetic outcomes in the majority of patients (5). In 466 breasts reconstructed in a single stage, the overall complication rate was 3.9% that included explanation in 1.3%, delayed healing in 1.1%, infection in 0.2%, and capsular contracture in 0.4%. Although many of these patients had conservative mastectomy, the percentage was not quantified. Colwell et al. studied 331 reconstructed breasts that had immediate 1-stage prosthetic breast reconstruction of which 66 (20%) were in the setting of conservative mastectomy (6). The overall complication rate following immediate single-stage implant reconstruction was 14.8% that included ten infections (3%), five seromas (1.5%), and delayed healing in 30 (9.1%). There was no difference in the complication rate following skin sparing and nipple sparing techniques. Dent et al. have evaluated nipple areolar ischemia following conservative mastectomy in the setting of 1-stage reconstruction (21). They reviewed 318 nipple-sparing mastectomies that were performed through an inframammary incision and demonstrated partial thickness nipple-areolar necrosis in 44 breasts (13.8%) and full thickness nipple-areolar necrosis in 21 breasts (6.6%). Operative debridement was not performed following partial thickness necrosis and in four cases of full thickness necrosis. Factors associated with nipple areolar ischemia included advanced age, higher body mass index (BMI), greater breast volume, tobacco use, ADM use, and 1-stage reconstruction.
Immediate 2-stage reconstruction following conservative mastectomy
With larger breast volume and increasing BMI or in the event of surgeon preference, the 2-stage reconstruction can be considered (10). Other indications for a 2-stage approach would be excessively thin mastectomy skin flaps, excessive undermining beyond the breast borders, need for postoperative radiation, a large quantity of skin with random blood supply, or evidence of poor skin and questionable nipple vascularity. The second stage provides a planned opportunity to improve the position of the inframammary and lateral mammary fold, perform a capsulotomy to improve contour, perform a capsulorrhaphy to minimize device migration, select an optimal permanent implant to achieve symmetry and projection, and to perform a contralateral symmetry procedure if needed (9). These are the reasons why most surgeons in the United States prefer the 2-stage technique.
The technique of 2-stage reconstruction in the setting of skin sparing mastectomy is well described (9,22,23); however, the technique of 2-stage reconstruction in the setting of conservative mastectomy is not. Nipple sparing mastectomy is usually performed through an inframammary or laterally based breast incision. The incision can traverse through the areola or can extend around the edge superiorly or inferiorly. Following the mastectomy, the inferior edge of the pectoralis major muscle is usually elevated and the subpectoral space is created. A tabbed tissue expander is usually used and secured with absorbable sutures placed along the inframammary fold to firmly secure the device. Anterior coverage of the device can be achieved using the pectoralis major muscle completely or partially. In the setting of partial muscle coverage, an acellular dermal matrix is usually used and sutured first to the inferior edge of the pectoralis major muscle and then to the fascia along the desired inframammary fold. The tissue expander is usually filled to 40-60% of capacity to minimize pressure on the mastectomy skin flaps and the nipple areolar complex. The periprosthetic space is copiously irrigated with an antibiotic solution. One or two closed suction drains are inserted. The mastectomy skin flap is carefully redraped over the reconstructed breast mound to minimize malposition of the nipple areola complex. The incisions are closed with resorbable sutures. Patients are seen in the office weekly for expansion until complete.
The second stage usually occurs 3 months later; however, this may be extended depending on the timing of chemotherapy and radiation therapy (24). The prior incision is usually used for access. The tissue expander is removed and a capsulotomy is usually performed along the upper pole and sometimes medially or inferiorly as needed for optimal positioning of the permanent implant. In the event of lateral or inferior device migration, a capsulorrhaphy is performed to compartmentalize the permanent implant. Device sizers are usually used to determine the optimal shape and volume of the permanent implant. A shaped or round silicone gel implant can be selected based on the breast parameters. Closed suction drains are rarely used at the time of device exchange unless there is a specific indication for them. Figures 5-10 highlight a woman that had immediate 2-stage prosthetic reconstruction following conservative mastectomy.
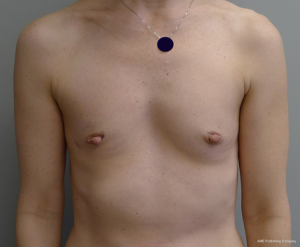
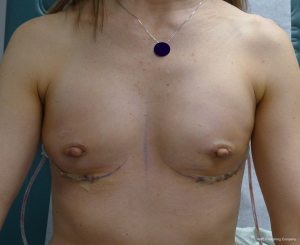

Outcomes following 2-stage reconstruction in the setting of conservative mastectomy have also been favorable. Sbitany et al. reviewed 122 patients and 202 breasts following 2-stage prosthetic reconstruction in the setting of total skin sparing mastectomy (25). Total pectoralis major coverage was used in 113 breasts and partial pectoral coverage with an ADM was used in 89 breasts. Intraoperative fill volume was greater in the partial muscle coverage group (205 vs. 52 cc). The postoperative complication profile with regard to delayed healing, seroma, and infection was similar for the two cohorts. Final nipple position was better controlled with the partial muscle coverage technique. Chen et al. reviewed a series of 115 nipple-sparing mastectomies performed in 66 patients that had immediate 2-stage reconstruction using a total muscle coverage technique (26). The most common incisional pattern was periareolar and radial (n=61) followed by inframammary (n=25), omega (n=14), prior incision (n=10), and trans areolar (n=5). Of the 115 conservative mastectomies, six were removed because of cancer (5.2%) and four were removed because of delayed healing (3.5%).
Delayed 2-stage reconstruction following conservative mastectomy
A delayed approach to prosthetic reconstruction is sometimes considered following conservative mastectomy. This is usually in the setting of thin mastectomy skin flaps, questionable viability for the nipple areolar complex, or to minimize the incidence of adverse events in patients at increased risk due to tobacco use or poorly controlled diabetes mellitus (9,27). The rationale is that placement of a tissue expander or implant in this setting would pose an unnecessary risk for reconstructive failure.
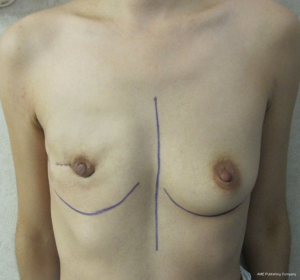
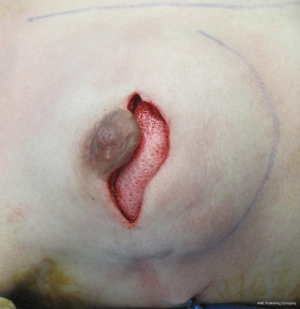
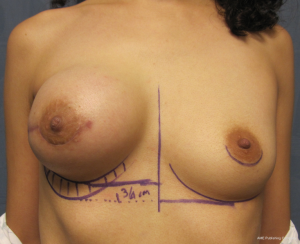
The technique of delayed reconstruction following conservative mastectomy requires special considerations in order to ensure optimal positioning of the nipple areolar complex. This is especially true in the setting of unilateral mastectomy because achieving breast symmetry will be more challenging. The timing can be as soon as 4 weeks following the mastectomy or years later. It is usually not considered when there has been prior radiation therapy of the natural breast or the mastectomy defect. The 2-stage technique is preferred in the setting of delayed reconstruction because a moderate to severe degree of skin contracture has usually occurred that will require reexpansion. Reelevation of the mastectomy skin flaps can sometimes recreate the mastectomy defect and allow for the same 2-stage technique that was described above. An alternative technique in the setting of subpectoral placement of the tissue expander is to leave the upper mastectomy skin flap attached to the pectoralis major muscle. Acellular dermal matrices are sometimes considered especially when the mastectomy defect has been recreated. Subpectoral placement of the tissue expander is usually considered although prepectoral placement can be considered in the uncommon scenario of thick mastectomy skin flaps. Tabbed tissue expanders are less important with delayed prosthetic reconstruction because the periprosthetic space has been carefully created. Intraoperative fill volumes are usually less compared to immediate reconstruction because of the skin contraction. The proper positioning of the nipple areolar complex usually requires some degree of mobility of the upper mastectomy skin flap in order to properly drape the skin envelope to match the opposite breast. In unilateral cases, patients are told that contralateral procedures such as mastopexy or reduction mammaplasty may be necessary to achieve symmetry. This is less of a consideration with bilateral cases. Figures 11-14 illustrate a patient having delayed 2-stage prosthetic reconstruction following conservative mastectomy.
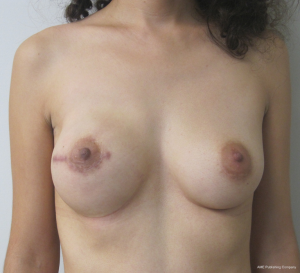
Outcomes following delayed prosthetic reconstruction have been favorable (27,28). Sullivan et al. have demonstrated fewer complications following delayed prosthetic reconstruction compared to immediate (P=0.008) (27). Capsular contracture occurred significantly more often following immediate reconstruction compared to delayed reconstruction (40.4% vs. 17%, P=0.001). This is partially explained because of higher degree of wound contamination during immediate reconstruction, compromised vascularity of the mastectomy skin flaps, and fewer infections following delayed reconstruction compared to immediate (2.4% vs. 5.4%, P=0.26). Alderman et al. have demonstrated that immediate reconstruction is associated with an increased total and major complication rate compared to delayed reconstruction regardless of the type of reconstruction (P=0.011 and 0.005, respectively) (28).
Conclusions
Conservative mastectomy can be safely and effectively performed with a variety of reconstructive techniques using prosthetic devices. The reconstruction can be performed immediately at the time of mastectomy or on a delayed basis. The 1-stage and 2-stage techniques can be used and provide excellent aesthetic and surgical outcomes. Considerations regarding the location of mastectomy incisions, use of ADM, and device location are important and will contribute to the outcome. Adverse events can occur and often related to skin and nipple vascularity, infection, and symmetry.
Acknowledgements
None.
Footnote
Conflicts of Interest: Dr. MY Nahabedian is a consultant for LifeCell, Allergan, and Sientra.
References
- Spear SL, Willey SC, Feldman ED, et al. Nipple-sparing mastectomy for prophylactic and therapeutic indications. Plast Reconstr Surg 2011;128:1005-14. [PubMed]
- Endara M, Chen D, Verma K, et al. Breast reconstruction following nipple-sparing mastectomy: a systematic review of the literature with pooled analysis. Plast Reconstr Surg 2013;132:1043-54. [PubMed]
- Albornoz CR, Bach PB, Mehrara BJ, et al. A paradigm shift in U.S. Breast reconstruction: increasing implant rates. Plast Reconstr Surg 2013;131:15-23. [PubMed]
- Cemal Y, Albornoz CR, Disa JJ, et al. A paradigm shift in U.S. breast reconstruction: Part 2. The influence of changing mastectomy patterns on reconstructive rate and method. Plast Reconstr Surg 2013;131:320e-6e. [PubMed]
- Salzberg CA, Ashikari AY, Koch RM, et al. An 8-year experience of direct-to-implant immediate breast reconstruction using human acellular dermal matrix (AlloDerm). Plast Reconstr Surg 2011;127:514-24. [PubMed]
- Colwell AS, Damjanovic B, Zahedi B, et al. Retrospective review of 331 consecutive immediate single-stage implant reconstructions with acellular dermal matrix: indications, complications, trends, and costs. Plast Reconstr Surg 2011;128:1170-8. [PubMed]
- Colwell AS. Direct-to-implant breast reconstruction. Gland Surg 2012;1:139-41. [PubMed]
- Colwell AS. Current strategies with 1-stage prosthetic breast reconstruction. Gland Surg 2015;4:111-5. [PubMed]
- Pusic AL, Cordeiro PG. Breast reconstruction with tissue expanders and implants: a practical guide to immediate and delayed reconstruction. Semin Plast Surg 2004;18:71-7. [PubMed]
- Spear SL, Seruya M, Rao SS, et al. Two-stage prosthetic breast reconstruction using AlloDerm including outcomes of different timings of radiotherapy. Plast Reconstr Surg 2012;130:1-9. [PubMed]
- Davila AA, Mioton LM, Chow G, et al. Immediate two-stage tissue expander breast reconstruction compared with one-stage permanent implant breast reconstruction: a multi-institutional comparison of short-term complications. J Plast Surg Hand Surg 2013;47:344-9. [PubMed]
- Roostaeian J, Sanchez I, Vardanian A, et al. Comparison of immediate implant placement versus the staged tissue expander technique in breast reconstruction. Plast Reconstr Surg 2012;129:909e-18e. [PubMed]
- Gould DJ, Hunt KK, Liu J, et al. Impact of surgical techniques, biomaterials, and patient variables on rate of nipple necrosis after nipple-sparing mastectomy. Plast Reconstr Surg 2013;132:330e-8e. [PubMed]
- Nahabedian MY. Breast reconstruction: a review and rationale for patient selection. Plast Reconstr Surg 2009;124:55-62. [PubMed]
- Nahabedian MY. Symmetrical breast reconstruction: analysis of secondary procedures after reconstruction with implants and autologous tissue. Plast Reconstr Surg 2005;115:257-60. [PubMed]
- Gdalevitch P, Ho A, Genoway K, et al. Direct-to-implant single-stage immediate breast reconstruction with acellular dermal matrix: predictors of failure. Plast Reconstr Surg 2014;133:738e-747e. [PubMed]
- Salzberg CA. Focus on technique: one-stage implant-based breast reconstruction. Plast Reconstr Surg 2012;130:95S-103S. [PubMed]
- Munhoz AM, Aldrighi CM, Montag E, et al. Clinical outcomes following nipple-areola-sparing mastectomy with immediate implant-based breast reconstruction: a 12-year experience with an analysis of patient and breast-related factors for complications. Breast Cancer Res Treat 2013;140:545-55. [PubMed]
- Nahabedian MY, Tsangaris TN. Breast reconstruction following subcutaneous mastectomy for cancer: a critical appraisal of the nipple-areola complex. Plast Reconstr Surg 2006;117:1083-90. [PubMed]
- Nahabedian MY. Shaped versus Round Implants for Breast Reconstruction: Indications and Outcomes. Plast Reconstr Surg Glob Open 2014;2:e116. [PubMed]
- Dent BL, Small K, Swistel A, et al. Nipple-areolar complex ischemia after nipple-sparing mastectomy with immediate implant-based reconstruction: risk factors and the success of conservative treatment. Aesthet Surg J 2014;34:560-70. [PubMed]
- Nahabedian MY. Breast reconstruction with tissue expanders and implants. In: Cosmetic and Reconstructive Surgery. Nahabedian MY. ed. Elsevier: UK, 2009.
- Spear SL, Economides JM, Shuck J, et al. Analyzing implant movement with tabbed and nontabbed expanders through the process of two-stage breast reconstruction. Plast Reconstr Surg 2014;133:256e-260e. [PubMed]
- Cordeiro PG, Albornoz CR, McCormick B, et al. What is the Optimum Timing of Post-mastectomy Radiotherapy in Two-stage Prosthetic Reconstruction: Radiation to the Tissue Expander or Permanent Implant? Plast Reconstr Surg 2015;135:1509-17. [PubMed]
- Sbitany H, Wang F, Peled AW, et al. Tissue Expander Reconstruction After Total Skin-Sparing Mastectomy: Defining the Effects of Coverage Technique on Nipple/Areola Preservation. Ann Plast Surg 2014. [Epub ahead of print]. [PubMed]
- Chen CM, Disa JJ, Sacchini V, et al. Nipple-sparing mastectomy and immediate tissue expander/implant breast reconstruction. Plast Reconstr Surg 2009;124:1772-80. [PubMed]
- Sullivan SR, Fletcher DR, Isom CD, et al. True incidence of all complications following immediate and delayed breast reconstruction. Plast Reconstr Surg 2008;122:19-28. [PubMed]
- Alderman AK, Wilkins EG, Kim HM, et al. Complications in postmastectomy breast reconstruction: two-year results of the Michigan Breast Reconstruction Outcome Study. Plast Reconstr Surg 2002;109:2265-74. [PubMed]