
Alteration in skeletal posture between breast reconstruction with latissimus dorsi flap and mastectomy: a prospective comparison study
Introduction
Breasts are important anatomical structures that symbolize femininity for women. Symmetry, natural shape, and proper size are biologically and esthetically important. An extremely large breast size may lead to difficulties in daily life and studies have demonstrated that physical weight and psychological factors may cause an altered skeletal posture (1-3). Moreover, many studies have reported that having both breasts is important in maintaining a center of gravity and stable posture (4-6). In the treatment of unilateral breast cancer, oncological safety is the primary goal; however, breast reconstruction surgery to esthetically resemble the natural look of the contralateral healthy breast is also essential. In several studies, it was reported that scoliosis of the spine may be induced when only mastectomy is performed unilaterally (7,8). Moreover, recent studies have reported that immediate breast reconstruction leads to decreased postoperative postural change compared to mastectomy-only (6). When selecting the appropriate method of immediate breast reconstruction (autologous tissue transfer, direct-to-implant, tissue expander), various factors such as the patient’s biotype, breast shape, size, other comorbidities, and preference are considered (9). Among the many surgical methods, unilateral autologous tissue transfer of the latissimus dorsi (LD) flap, which is the most commonly used autologous tissue, is a common breast reconstruction method in patients with a small-to-moderate breast size in Asian. Studies have reported the functional outcomes of this method (10); however, there are only a few reports on how reconstruction correlates with alteration in skeletal posture. The purpose of this study was to examine postural and functional changes after unilateral LD flap. If postural change occurred after autologous LD breast reconstruction, the degree of posture change between LD flap and mastectomy-only was compared. We hypothesized that transfer of the unilateral LD flap to the breast, and consequently, the absence of the LD among the back muscles, may affect the skeleton and cause changes in posture. Therefore, the alteration in skeletal posture was assessed after unilateral breast reconstruction using a LD flap and was compared to that of a mastectomy-only patient group. We present the following article in accordance with the STROBE reporting checklist (available at http://dx.doi.org/10.21037/gs-21-31).
Methods
Ethical statement
The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The Institutional Review Board of Kyungpook National University Chilgok Hospital (No. 2018-02-007-003) approved this prospective study and all patients provided informed consent to have their data recorded, analyzed, and published for research purposes.
Study design
From January 2018 to February 2020, a control group of 23 patients who underwent a mastectomy only and an experimental group of 31 patients who underwent breast reconstruction using a LD flap immediately after mastectomy were enrolled. Patients’ characteristics [age, body mass index (BMI), excised mass weight, etc.] were collected preoperatively.
Patient selection
This prospective randomized study included patients (I) with diagnosed breast cancer, (II) who underwent immediate reconstruction after a breast cancer operation or received a mastectomy only, and (III) were aged 30–60 years at the time of the operation. This study excluded patients (I) diagnosed with advanced-stage III or IV breast cancer, (II) who were unable to answer the self-questionnaire due to cognitive impairment, (III) with a history of neurologic disorders or musculoskeletal problems on the trunk and the upper extremity, and (IV) with a history of alcohol or drug abuse.
Operative technique for mastectomy-only (control group)
The patient was placed supine with ipsilateral arm abducted. The skin incision was designed as elliptically including the nipple-areolar complex. The subcutaneous flap dissection was performed with electrocautery (BovieR) removing remaining breast parenchyma from the inframammary line as the inferior boundary of the breast, the mid-axillary line as the lateral boundary, the clavicle as the superior boundary, and the sternum as the medial boundary. After the whole breast was removed, a 400 mL of drainage tube was inserted into the mastectomy bed. The superior and inferior skin flap were closed with a subcutaneous suture technique.
Breast reconstruction operative technique using the LD flap (experimental group)
Patients diagnosed with unilateral breast cancer underwent mastectomy in the supine position by a breast surgeon for unilateral breast reconstruction. The patients were then changed to a decubitus position to elevate the extended LD flap and humoral detachment was performed, followed by axillary tunneling to transfer the flap to the defective breast area. Afterward, patients were returned to the supine position. Where breast volume was insufficient, a small implant was added to reconstruct a breast with a shape similar to the healthy breast.
Biomechanical measurement
In the experimental and control groups, we assessed the Cobb’s angles in the spine X-ray, computed tomography (CT) parameters, and 3D scan parameters preoperatively (T0), 6 months after surgery (T1), and 1 year after surgery (T2).
Cobb’s angle
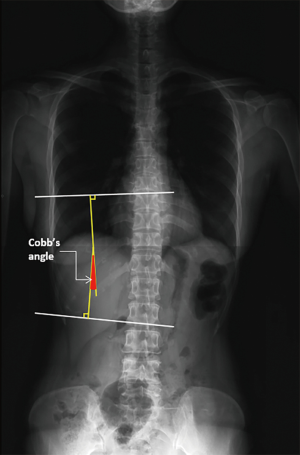
We assessed spine X-rays and measured the Cobb’s angle in the thoracic spine curve by two independent physiatrists using the Picture Archiving and Communication System (PACS, INFINITT®). Two parallel lines were drawn as follows: the upper margin of the cranial-end vertebrae and the lower margin of the caudal-end vertebrae in the coronal alignment of the vertebral body. The angle between the upper lower lines of the thoracic spine curve was calculated as the Cobb’s angle in PACS (Figure 1).
Gross photometry
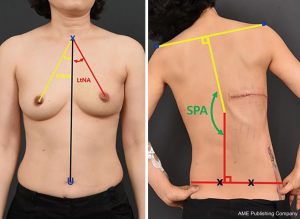
In the frontal view, the line angles of each nipple relative to the midline connecting the SN and the umbilicus were measured using Photoshop. In the back view, bending views were obtained for both sides and the shoulder, pelvic angle (SPA), where the lines connecting both shoulder edge points and the vertical line connecting the posterior superior iliac spine (PSIS) were measured using Photoshop (Figure 2).
CT parameters
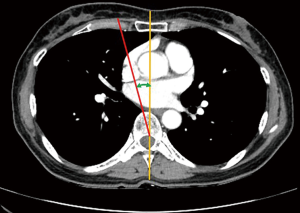
In the axial view where the healthy nipple was visible, the angle between a line connecting the sternum and spinous process and a line extending along the long axis of the spine column was measured in PACS (Figure 3).
3D scan parameters
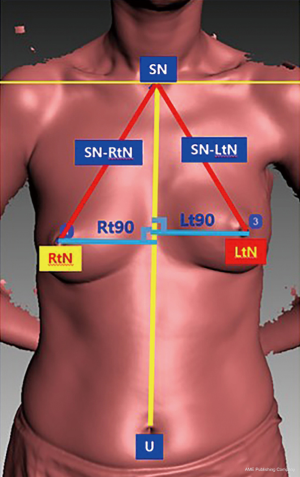
Using an Artec 3D scanner, the midline connecting the sternal notch (SN) and umbilicus in the frontal view, the line connecting both nipples to the SN, and the vertical distance from both nipples to the midline was measured using PACS (Figure 4).
Functional assessments
In the experimental group, we assessed pain intensity, disability of the upper extremities, and quality of life at T0, T1, and T2.
Intensity of pain
The visual analog scale (VAS) was used to assess pain intensity during shoulder abduction motion, which was scored from 0 to 10.
Disability of the upper extremities
Disabilities in shoulder function were assessed using the Disabilities of the Arm, Shoulder and Hand (DASH) questionnaire, which consists of 30 items, each with five response choices; 21 items assessed difficulty in performing different physical activities, six items assessed symptoms, and the remaining three items assessed psychosocial effects. A score of 0 indicates ‘no disability’ and a score of 100 indicates ‘complete disability’. In this study, we used the Korean version of the questionnaire, which has proven reliable for measuring upper-extremity dysfunction (11).
Quality of life
The 36-Item Short-Form Health Survey (SF-36) is a widely used, generic, and patient-reported measure of health status. It comprises four physical domain scales: physical functioning, physical role functioning, bodily pain, and general health, which are combined into a physical component summary scale. The survey also comprises four mental domain scales: vitality, social functioning, emotional functioning, and mental health, which are combined into a mental component summary scale. We used the Korean version of the SF-36 (12).
Statistical analysis
All statistical analyses were performed using SPSS 23.0 (SPSS Inc., Chicago, IL, USA). Assessments were normally distributed according to the Shapiro-Wilk test. A t-test was performed to assess differences in clinical characteristics, Cobb’s angle, and CT parameters between the control and experimental groups at T0. Repeated measures analysis of variance (ANOVA) with a Bonferroni post-hoc test was performed to evaluate the interaction effects of time (T0, T1, and T2) and group (control and experimental groups) on Cobb’s angle and CT parameters, which changed over time. In addition, we assessed the relationship between the Cobb’s angle and CT parameters at each time point using Spearman’s correlation. Finally, a paired t-test was performed to assess the difference in VAS, DASH, and SF-36 scores between T0 and T1, T0 and T2, and T1 and T2 in the experimental group.
Results
In the control and experimental groups, the average age, BMI, and mass weight were 58.7/46.2 years, 24.9/22.5 kg/m2, 386.8/259.1 g, respectively. There were no significant differences in patient characteristics between groups at T0 (Table 1). Additionally, there were three cases in which a small implant was added due to the lack of LD flap volume, and the mean size of the implant was 133.3 mL.
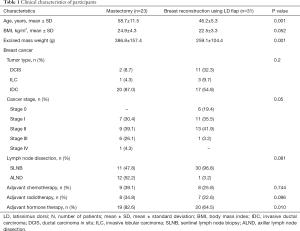
Full table
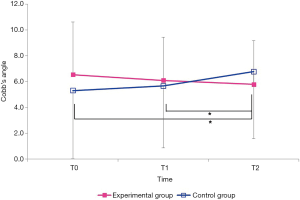
The mean values of the Cobb’s angle and CT parameters in both groups are shown in Table 2. There were changes in the mean values of the Cobb’s angle over time between the groups (Figure 5). Interaction effects of time and groups had a significant effect on the Cobb’s angle (F=10.536, P=0.000). Bonferroni post-hoc analysis revealed a significant increase in the Cobb’s angle from T0 to T2 (95% CI: –2.465 to –0.483, P=0.002) and from T1 to T2 (95% CI: –2.016 to –0.233, P=0.010) in the control group. However, there were no changes in the mean values of CT parameters over time between groups (F=0.429, P=0.653).

Full table
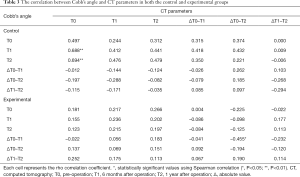
The relationship between Cobb’s angle and CT parameters in each group is shown in Table 3. In the control group, the Cobb angle values at T1 and T2 were positively correlated with the CT parameter values at T0 (r=0.688, P=0.003; r=0.694, P=0.003, respectively). In the experimental group, changes in the Cobb’s angle between T0 and T1 were negatively correlated with changes in CT parameters between T0 and T2 (r=–0.455, P=0.029).
Full table
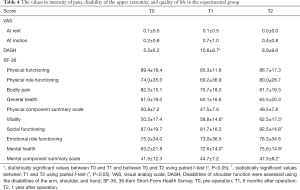
Table 4 shows the values of pain intensity, DASH score, and SF-36 score in the experimental group at T0, T1, and T2. In the experimental group, pain intensity at rest and with shoulder motion did not change, whereas there was a significant increase in DASH score between T0 and T1 (t=–2.249, P=0.035). In addition, there were significant increases in the vitality scale (t=–2.828, P=0.010) and mental health scale (t=–2.100, P=0.048) of SF-36 scores between T0 and T1. From T0 to T2, there were significant increases in the scores of the vitality scale (t=–2.218, P=0.042), mental health scale (t=–2.776, P=0.014), and mental component summary scale (t=–2.455, P=0.027) in the SF-36. In addition, there was a significant increase in the social functioning scale between T1 and T2 (t=–2.276, P=0.035).
Full table
In Table 5, changes in the 3D scan parameters were analyzed for correlation with changes in the Cobb’s angle at T0 and T1. The absolute value of change (ΔT0–T1) in the Cobb’s angle at T0 and T1 was positively correlated with the change in vertical distance from the midline connecting the SN and umbilicus to the right nipple in the 3D scanner (P=0.009). The ΔT0–T2 in the Cobb angle and the vertical distance from midline to the left nipple (Lt90) from the 3D scans were negatively correlated (P=0.038).

Full table
Discussion
In 1978, Fredricks first reported that skeletal posture may be affected by the size, weight, and difference between the two breasts due to their bilateral and anterior location. The study reported that a large difference between the breasts may affect the pelvis, shoulder, and spinal posture due to the wagon-wheel effect (13). Therefore, unilateral breast reconstruction using the LD muscle for symmetrical volume to the healthy breast after mastectomy may also affect the spine. In addition, there have been interesting reports on the differences between patients who underwent mastectomy-alone and those who underwent mastectomy with immediate breast reconstruction. In a study by Jeong et al., it was reported that the amount of postoperative change was less in the reconstruction group than in the mastectomy-only group when using chest X-rays to measure the Cobb’s angles (14). Moreover, studies have compared immediate breast reconstruction patients of various methods and mastectomy-only patients, which showed that there were no significant differences in trunk rotation (15). In a study on upper-limb movement in patients who underwent a mastectomy-only, changes were observed; however, the changes were not considered as harmful (15). Another study reported the possibility of postural change after surgery and that physical therapy should be performed as well (15). Furthermore, a previous study reported that the possibility of scoliosis may increase if the difference in breast volume between the two breasts is large following augmentation mammaplasty, an esthetic breast surgery (16).
Based on these previous reports, the current study compared the Cobb’s angle between the mastectomy-only control group and the reconstruction experimental group by assessing CT findings taken during cancer evaluation. In the control group, the Cobb’s angle increased at both T1 and T2, which was significantly increased compared to that of the experimental group. The results here show that unilateral transfer of LD muscle had significantly fewer effects on spine posture in the experimental group compared to the control group. Changes in the thoracic spine angle had no significant correlation with the Cobb’s angle (Figure 3, Tables 2,3). Based on these comparative outcomes, the change in the Cobb’s angle between the control and experimental group was statistically significantly different between preoperative and 1-year postoperative time points, and between 6-month postoperative and 1-year postoperative time points, confirming that there was an impact. Although the change in the Cobb’s angle between the two groups did not accompany pathological severity that requires spine operation, the difference was slightly increased in the control group (Figure 5), whereas the difference was improved over time in the experimental group. Although it is difficult to measure the absolute length using gross photometry, a bending view was acquired and angles of various parameters of the healthy and affected sides showed no significant correlation with the Cobb’s angle (Figure 2). Our results showed the relationship between Cobb’s angle in 2D X-ray images and CT parameters in axial images. Tauchi et al. reported that Cobb’s angle measurements using X-ray in the frontal plane could be reproduced with measurements using 3D-CT images (17). These findings are in accordance with a previous study. Additionally, in our study, each group showed the differential relationship between Cobb’s angle in frontal spine X-ray and CT parameters in axial CT views. In the control group, the value of Cobb’s angle at T1 and T2 were positively correlated with the value of CT parameters at pre-operation, respectively. In the experimental group, the changes in the Cobb’s angle from T0 to T1 were negatively correlated with the changes in CT parameter from T0 to T2. These findings suggest that the degree of 3D spine alignments at pre-operation would be closely related to the degree of 2D spine alignments after T1 and T2 in patients with only mastectomy. However, the changes of 2D spine alignments after operation would not be related to 3D spine alignments. In the experimental group, the changes of 2D spine alignment from T0 to T1 would be conversely related with the change of 3D spine alignments from T0 to T2. LD flap operation may prevent further aggravation of changes of spine alignments after a skin sparing mastectomy. For parameters measured using a 3D scanner, the absolute value of the change in the Cobb’s angle at T0 and T1 was positively correlated with a change in the vertical distance from the midline connecting the SN and umbilicus to the right nipple (P=0.009) (Figure 4). This finding suggests that a greater change in the Cobb’s angle indicates that the spine posture is tilted to the affected side, increasing the vertical distance from the midline to the nipple. Moreover, ΔT0–T2 of the Cobb’s angle and the Lt90 of the 3D scanner were negatively correlated (P=0.038), which further supports the idea that the vertical distance to the nipple on the healthy side is shortened. To analyze spine posture changes related to scoliosis caused by breast reconstruction, X-rays need to be performed, with the results evaluated by a specialist to measure the Cobb’s angle. Our findings suggest that gross care using a 3D scanner may be sufficient to allow easy and reliable evaluation and follow-up observations. The pathological severity and post-treatment recovery could be confirmed by measuring the Cobb’s angle required for the diagnostic criteria of scoliosis through X-ray imaging, along with a small amount of irradiation. However, parameters that can predict the spine posture based on the vertical distance from the canonical line of the body to the nipple through a 3D scanner were derived in this study. Prior to the analysis for this association, the change in skeletal posture was shown to be significantly lower in the experimental group than in the control group, in agreement with other reports. In addition, physiotherapy was not conducted in either group, as the possibility of physiotherapy affecting skeletal posture—the main aim of the study—could not be excluded (18).
Postoperative outcomes in the experimental group were evaluated using the DASH and SF36 questionnaires, which showed statistically significant values, suggesting that reliable outcomes were obtained in functional or mental aspects (11,12). This study is meaningful as a basic study showing parameters related to postural alteration resulting from breast reconstruction using a LD flap. However, this study was limited by its small patient pool and the inability to perform functional evaluations and QOL in the control group. Additionally, this study derived the amount of change by comparing the preoperative and postoperative measurements in each patient, as opposed to making comparisons between patients, because of the relatively early cancer stage of the experimental group with breast reconstruction, and the likelihood of variance in recovery owing to the young age of the experimental group, which could have led to a bias. Further, while breast volume had a minimal effect on the operation in the cases of mastectomy-only, it was identified that breast volume of the experimental group could be lower than that of the control group, as volume enhancement with a small implant is limited when breast reconstruction using a LD flap is being performed. A subsequent long-term follow-up study is being planned for detailed analysis as a function of age and breast volume and comparative analysis between partial breast reconstruction and total breast reconstruction.
Conclusions
The experimental group that underwent immediate breast reconstruction with a LD flap after total or partial mastectomy showed recovery in the skeletal posture up to 1-year post-surgery compared to the control group that underwent mastectomy-only. In the group that underwent LD flap reconstruction, the functional outcome was satisfactory and the change in the Cobb’s angle was significantly less than in the mastectomy-only group, suggesting that immediate breast reconstruction using a LD flap is a reliable method to prevent skeletal alteration. Moreover, it is thought that measured parameters using a 3D scanner are sufficient to show the effects of reconstruction on spine posture.
Acknowledgments
Funding: This work was supported by Biomedical Research Institute grant, Kyungpook National University Hospital (2018).
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at http://dx.doi.org/10.21037/gs-21-31
Data Sharing Statement: Available at http://dx.doi.org/10.21037/gs-21-31
Peer Review File: Available at http://dx.doi.org/10.21037/gs-21-31
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/gs-21-31). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by Institutional Review Board of Kyungpook National University Chilgok Hospital (No. 2018-02-007-003) and informed consent was taken from all individual participants.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Hanuszkiewicz J, Malicka I, Stefańska M, et al. Body posture and trunk muscle activity in women following treatment of breast cancer. Ortop Traumatol Rehabil 2011;13:45-57. [Crossref] [PubMed]
- Malicka I, Barczyk K, Hanuszkiewicz J, et al. Body posture of women after breast cancer treatment. Ortop Traumatol Rehabil 2010;12:353-61. [PubMed]
- Barbosa AF, Lavoura PH, Boffino CC, et al. The impact of surgical breast reduction on the postural control of women with breast hypertrophy. Aesthetic Plast Surg 2013;37:321-6. [Crossref] [PubMed]
- Barbosa Jde A, Amorim MH, Zandonade E, et al. Evalution of body posture in women with breast cancer. Rev Bras Ginecol Obstet 2013;35:215-20. [PubMed]
- Lapid O, de Groof EJ, Corion LU, et al. The effect of breast hypertrophy on patient posture. Arch Plast Surg 2013;40:559-63. [Crossref] [PubMed]
- Nicoletti G, Mandrini S, Finotti V, et al. Objective clinical assessment of posture patterns after implant breast augmentation. Plast Reconstr Surg 2015;136:162e-70e. [Crossref] [PubMed]
- Rostkowska E, Bak M, Samborski W. Body posture in women after mastectomy and its changes as a result of rehabilitation. Adv Med Sci 2006;51:287-97. [PubMed]
- Serel S, Tuzlalı ZY, Akkaya Z, et al. Physical effects of unilateral mastectomy on spine deformity. Clin Breast Cancer 2017;17:29-33. [Crossref] [PubMed]
- Atanes Mendes Peres AC, Dias de Oliveira Latorre MD, Yugo Maesaka J, et al. Body posture after mastectomy: comparison between immediate breast reconstruction versus mastectomy alone. Physiother Res Int 2017; [Crossref] [PubMed]
- Yang JD, Huh JS, Min YS, et al. Physical and functional ability recovery patterns and quality of life after immediate autologous latissimus dorsi breast reconstruction: a 1-year prospective observational study. Plast Reconstr Surg 2015;136:1146-54. [Crossref] [PubMed]
- Lee JY, Lim JY, Oh JH, et al. Cross-cultural adaptation and clinical evaluation of a Korean version of the disabilities of arm, shoulder, and hand outcome questionnaire (K-DASH). J Shoulder Elbow Surg 2008;17:570-4. [Crossref] [PubMed]
- Han CW, Lee EJ, Iwaya T, et al. Development of the Korean version of Short-Form 36-Item Health Survey: health related QOL of healthy elderly people and elderly patients in Korea. Tohoku J Exp Med 2004;203:189-94. [Crossref] [PubMed]
- Fredricks S. Skeletal and postural relations in augmentation mammaplasty. Ann Plast Surg 1978;1:44-7. [Crossref] [PubMed]
- Jeong JH, Choi B, Chang SY, et al. The effect of immediate breast reconstruction on thoracic spine alignment after unilateral mastectomy. Clin Breast Cancer 2018;18:214-9. [Crossref] [PubMed]
- Crosbie J, Kilbreath SL, Dylke E, et al. Effects of mastectomy on shoulder and spinal kinematics during bilateral upper-limb movement. Phys Ther 2010;90:679-92. [Crossref] [PubMed]
- Tsai FC, Hsieh MS, Liao CK, et al. Correlation between scoliosis and breast asymmetries in women undergoing augmentation mammaplasty. Aesthetic Plast Surg 2010;34:374-80. [Crossref] [PubMed]
- Tauchi R, Tsuji T, Cahill PJ, et al. Reliability analysis of Cobb angle measurements of congenital scoliosis using X-ray and 3D-CT images. Eur J Orthop Surg Traumatol 2016;26:53-7. [Crossref] [PubMed]
- Lohana P, Button J, Young D, et al. Functional recovery after bilateral extended autologous latissimus dorsi breast reconstruction: a prospective observational study. J Plast Reconstr Aesthet Surg 2019;72:1060-6. [Crossref] [PubMed]


