Comparative study of software techniques for 3D mapping of perforators in deep inferior epigastric artery perforator flap planning
Introduction
Currently, computed tomographic (CT) angiography (CTA) is widely considered the gold standard perforator imaging technique for preoperative planning an autologous breast reconstruction with deep inferior epigastric artery (DIEA) perforator (DIEP) flap (1,2). The scan data can be 3D reconstructed to produce a “perforator map” that assists surgeons in selecting an appropriate perforator, donor site, and the flap. A plethora of studies have demonstrated a high accuracy of CTA in detecting perforators, reporting a sensitivity and specificity close to 100% (3-12). In comparison to other perforator imaging modalities, such as Doppler ultrasound and magnetic resonance angiography (MRA), CTA has demonstrated superior visualization of the perforators and their subcutaneous course, respectively (7,10). While MRA may be evolving in this role, widespread outcome data is still lacking. The benefits of CTA have translated into improved clinical outcomes, such as increased flap survival, reduced donor site morbidity, and reduced operating time (5,6,10,12-24). To this end, appropriate use of hardware and software is essential to obtain optimal perforator data from CTA.
Through various scanner hardware brands (i.e., Siemens, Toshiba, and Philips), varying number of multi-detector rows (i.e., 4-slice to 320-slice scanners) and differing contrast media and volumes, all scanners and techniques are able to achieve high quality and clinically useful images (1,2). In addition, we have published optimized CTA scanning techniques that enhance perforator visualizations, such as initiating contrast bolus trigger at the common femoral artery, moving the computed tomography table caudo-cranially, and disabling the Siemens Care Dose 4D feature (10).
High cost and limited accessibility of 3D imaging softwares that generate 3D reconstructions suitable for clinical use have been challenging for hospitals with relatively limited resources. Most of the currently available proprietary softwares, such as Siemens Syngo InSpace 4D (Siemens, Erlangen, Germany) (25) and VoNaviX (IVS Technology, Chemnitz, Germany) (26) are expensive. Some are not readily accessible outside the institution where it was originally developed, such as virSSPA (University Hospitals Virgen del Rocio, Sevilla, Spain) (15). Furthermore, many programs available cannot provide adequate images, with some not able to visualize perforators to a clinically useful degree. One particular program that we have found that can achieve optimal images is Siemens Syngo InSpace 4D (10). The program enables users to assign color to various contrast values using color look-up table (CLUT) function, providing superior contrast resolution to the 3D reconstructions. Again however, the cost and availability are significant limitations. Previously, we have demonstrated the application of a free 3D imaging program, Osirix (Pixmeo, Geneva, Switzerland).
Osirix is a free imaging processing software, specifically designed for medical imaging by a radiologist, and is readily downloaded online for use unreservedly (27). It is able to produce the same or better images than the currently available programs on a user-friendly interface. Furthermore, Osirix can be readily operated on a laptop computer, which enables viewing in the operating theatre or at home. Similar to Siemens Syngo InSpace, Osirix enables the user to create 3D volume-rendered reconstructions and assign colors using an appropriate CLUT function to optimize visualization of perforators and their course, as demonstrated in our previous case report (28).
In the current study, we investigate the accuracy of the freely available 3D imaging software, Osirix, by comparing it to the proprietary program, Siemens Syngo InSpace 4D, and also comparing both softwares to the intraoperative findings.
Methods
The study design was a prospective case series. A total of 50 consecutive patients (i.e., 100 hemi-abdominal walls) underwent CTA prior to a DIEP flap breast reconstruction. All patients were aged between 30 and 60 years and spanned a wide range of body habitus. All imaging findings were recorded by a single operator and all intraoperative findings were recorded by the operating surgeon.
CTA technique
All scans were performed at a single institution (Future Medical Imaging Group, Melbourne, Australia) using a standardized protocol that has been modified and improved from the conventional CTA methodology in order to maximize the image quality and minimize radiation exposure (10,21). The computed tomography scanner used was a Siemens SOMATOM Sensation 64 multi-detector row computed tomography scanner (Siemens Medical Solutions, Erlangen, Germany) and the scan parameters are summarized in Table 1.
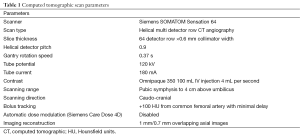
Full table
Patients were scanned in a position matching operative positioning: supine, with no clothing or straps to deform the abdominal contour. The scan range was limited to the tissue used intraoperatively and thus spanned from the pubic symphysis to 4 cm above the level of umbilicus. A bolus of 100 mL of intravenous omnipaque 350 was used for contrast, without oral contrast. We have previously described three major modifications introduced to the standard CTA protocol in order to enhance the arterial phase filling and the resolution of cutaneous vasculature (10). Briefly, the contrast bolus trigger to begin scanning was taken at the common femoral artery; the computed tomography table movement was reversed to scan caudo-cranially from the pubic symphysis to match the filling of DIEA; and the Siemens Care Dose 4D features was disabled, which maximized the abdominal wall signal-to-noise ratio.
Scan analysis
CTA scans were analyzed using both imaging softwares: Siemens Syngo InSpace 4D (Version 2006A; Siemens, Erlangen, Germany) and Osirix (Pixmeo, Geneva, Switzerland). The thin-slice (i.e., 1 mm or less) axial raw data were reformatted into 3D volume-rendered reconstructions and maximum intensity projections (MIPs) to identify the number and location of perforators, and the branching pattern of DIEA (29).
Perforator mapping
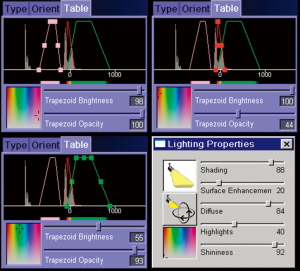
3D-reconstructed images of the abdominal wall perforators are generated using volume-rendering technique (VRT) and MIP techniques. VRT reconstructions required the use of the CLUT function found in both of the image processing softwares. Additionally in Osirix (Pixmeo, Geneva, Switzerland), we applied Gaussian blur to the final 3D reconstruction facilitating the removal of interference within the data (Figures 1,2). All infraumbilical perforators with diameter greater than 0.5 mm were identified and mapped on VRT reconstructions. Arrowheads were placed at the point of emergence of each perforator from the anterior rectus sheath. They were overlaid on to a 2D representation of each patient’s abdominal wall with a grid of 4 mm squares applied to the image centered on the umbilicus as reference point. The transverse distances of each perforator from the midline were recorded to the closest 0.5 cm. The perforators were recorded as found in medial or lateral row. MIP reconstructions were used to illustrate intramuscular course of the perforators.
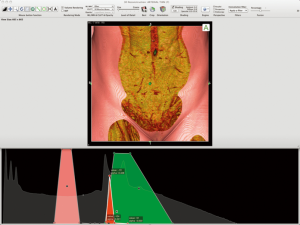
Intraoperative measurements
The perforator locations were compared with operative findings, where they were located on equivalent grids. Intraoperative grids were placed over the lower abdominal wall, with the umbilicus and midline as references, and the location of perforators was documented on it with sterile pens. A 0.5-cm margin of error was given for the location of each perforator. This was a conservative figure given as an estimate of the combined error associated with the calculation of concordance, and included the following factors: CTA error (e.g., patient movement, venous contamination), CTA reporting error (e.g., multiplanar reformatting error, reading error), intraoperative measurement error (e.g., limitation of measurement tool, reading error), and patient error (e.g., umbilical shift, abdominal pannus mobility). For the purpose of comparison, the operative findings were considered the standard.
All perforators were explored bilaterally, including the perforators not included in the flap. All perforators greater than 0.5 mm in diameter were included in the study and recorded in the manner described. As achieved during the CTA scan interpretation, the perforators identified intraoperatively comprised arterial perforators and not adjacent veins.
Statistical analysis
The perforator locations were recorded as exact values and the findings were compared between the two software programs. In addition, the data from each program was compared to the operative findings. The comparative analysis was conducted using SPSS Statistics software package (IBM, Armonk, New York, USA) and the outcomes were analyzed using paired Student’s t-test. A P value of <0.05 was accepted as statistically significant.
Results
A total of 50 CTA scans were performed in 50 consecutive cases (i.e., 100 hemi-abdominal walls) that identified 512 perforators of DIEA at an average of 5.12 perforators per hemi-abdomen. Concordance between Siemens Syngo InSpace 4D (version 2006A; Siemens, Erlangen, Germany) and Osirix (Pixmeo, Geneva, Switzerland) in accurately identifying perforator locations, and comparison between each of the software programs to intraoperative findings were evaluated.
Between Siemens Syngo InSpace 4D and Osirix, 510 out of 512 perforators (99.6%) had concordance. The two discordant perforators between the imaging programs were located in the lateral row and had only 0.5 cm of difference. Mean transverse distance from the midline using both software programs was 3.36 cm, with no statistical difference between them for measuring perforator location (Table 2 and Figure 3).

Full table

Between each of the softwares and the operative findings, there was a mean difference of 0.7 mm per perforator using both programs (Tables 3,4). Although this difference was statistically significant (P<0.01), this was not a clinically significant difference (i.e., less than 1 mm).

Full table

Full table
An analysis of perforators that had a difference between imaging and intraoperative findings was undertaken, with 40 perforators (7.8%) discordant between imaging and operative findings (Table 5). Of 18 perforators that had 0.5 cm difference with operative findings, 7 were located in medial row and 11 in lateral row. Of 12 perforators that had 1 cm difference, 5 were located in medial row and 7 in lateral row. Of 8 perforators that had 1.5 cm difference, 1 was located in medial row and 7 in lateral row. Of 2 perforators that had 2 cm difference, none were located in medial row and 2 in lateral row. Medial row perforators accounted for 13 out of 40 discordant results (32.5%) and lateral row 27 out of 40 (67.5%). Hence, imaging was more accurate when assessing medial row perforators (32.5% vs. 67.5%). Furthermore, when specifically assessing the larger discrepancies (>1 cm), medial row accounted for only 1 out of 10 (10%) and lateral row 9 out of 10 (90%).

Full table
Discussion
An improved understanding of the DIEA and its perforators from CTA has assisted reconstructive surgeons in the selection of the appropriate donor site, perforator, and hemi-abdominal wall of choice for reconstruction, which has translated to significant improvements in clinical outcomes (5,6,10,12-24). To achieve this, the use of appropriate hardware and software is vital. For CTA hardware, CT scanners from various brands using different multi-detector rows with varying IV contrast materials and volumes have demonstrated in the literature to deliver consistently sufficient scan data (1,3,5,10,12,15). In contrast, the high cost and limited accessibility of image processing software that can produce clinically useful 3D volume-rendered reconstructions have limited a wide application of CTA. To this effect, Osirix, a medical imaging program available for free online, have been useful. It is capable of producing the same or superior quality 3D reconstructions than the proprietary softwares and has added advantages of user-friendly interface and portability.
We have previously described the potential utility of Osirix for preoperatively planning a DIEP flap breast reconstruction in a case report (28). In the current case series, we demonstrate that Osirix is as accurate as the commonly used proprietary software, Siemens Syngo InSpace 4D, in identifying perforator number and location (99.6%). Furthermore, the measurements from both programs closely correlated to the operative findings (92.2%). The discordance between imaging and operative findings was most pronounced in assessing lateral row perforators (90% vs. 10%). For the purpose of the current study, we forewent comparing perforator diameters since these measurements can be made on standard axial slices of a CTA, regardless of the software program.
In addition to its accuracy in perforator localization, Osirix has the potential to yield superior quality 3D images than Siemens Syngo InSpace 4D due to its 16-bit CLUT function and the capacity to apply Gaussian blur after the 3D reconstruction to reduce interference. Furthermore, Osirix exhibits an easy-to-navigate user interface that is readily accessible to clinicians without technological background and it is compatible on Mac operating system. As a result, surgeons can access the 3D reconstructed images on their portable computer in the operating theatre or at home.
Of note, although free for the basic version, there is a cost to the fully functional version that allows more images to be processed and your own presets to be used. Even this version offers a widely affordable option for most institutions compared to other options.
One of the limitations of the current study is our relatively small sample size. A larger randomized study with greater sample size will be required to further validate our findings. Moreover, a future study may consider comparing Osirix to a host of other proprietary softwares, such as VoNaviX, and their impact on clinical outcomes. For the purpose of this study, the comparative analysis was performed in cases of autologous breast reconstruction with DIEP flap. However, validating Osirix in assessing other free flap options for autologous breast reconstruction may be of value.
Conclusions
This comparative analysis demonstrates that the accuracy of Osirix, a freely available medical image processing software, is concordant with Siemens Syngo InSpace 4D, a commonly utilized proprietary software, in localizing perforators for autologous breast reconstruction with DIEP flaps. Measurements from both programs correlated equally to the intraoperative findings. Most of the discrepancies arose in the lateral row perforators.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Chae MP, Hunter-Smith DJ, Rozen WM. Comparative analysis of fluorescent angiography, computed tomographic angiography and magnetic resonance angiography for planning autologous breast reconstruction. Gland Surg 2015;4:164-78. [PubMed]
- Rozen WM, Chubb D, Grinsell D, et al. Computed tomographic angiography: clinical applications. Clin Plast Surg 2011;38:229-39. [PubMed]
- Alonso-Burgos A, García-Tutor E, Bastarrika G, et al. Preoperative planning of deep inferior epigastric artery perforator flap reconstruction with multislice-CT angiography: imaging findings and initial experience. J Plast Reconstr Aesthet Surg 2006;59:585-93. [PubMed]
- Cina A, Barone-Adesi L, Rinaldi P, et al. Planning deep inferior epigastric perforator flaps for breast reconstruction: a comparison between multidetector computed tomography and magnetic resonance angiography. Eur Radiol 2013;23:2333-43. [PubMed]
- Masia J, Clavero JA, Larrañaga JR, et al. Multidetector-row computed tomography in the planning of abdominal perforator flaps. J Plast Reconstr Aesthet Surg 2006;59:594-9. [PubMed]
- Masia J, Kosutic D, Clavero JA, et al. Preoperative computed tomographic angiogram for deep inferior epigastric artery perforator flap breast reconstruction. J Reconstr Microsurg 2010;26:21-8. [PubMed]
- Pauchot J, Aubry S, Kastler A, et al. Preoperative imaging for deep inferior epigastric perforator flaps: a comparative study of computed tomographic angiography and magnetic resonance angiography. Eur J Plast Surg 2012;35:795-801.
- Pellegrin A, Stocca T, Belgrano M, et al. Preoperative vascular mapping with multislice CT of deep inferior epigastric artery perforators in planning breast reconstruction after mastectomy. Radiol Med 2013;118:732-43. [PubMed]
- Rosson GD, Williams CG, Fishman EK, et al. 3D CT angiography of abdominal wall vascular perforators to plan DIEAP flaps. Microsurgery 2007;27:641-6. [PubMed]
- Rozen WM, Phillips TJ, Ashton MW, et al. Preoperative imaging for DIEA perforator flaps: a comparative study of computed tomographic angiography and Doppler ultrasound. Plast Reconstr Surg 2008;121:9-16. [PubMed]
- Scott JR, Liu D, Said H, et al. Computed tomographic angiography in planning abdomen-based microsurgical breast reconstruction: a comparison with color duplex ultrasound. Plast Reconstr Surg 2010;125:446-53. [PubMed]
- Tong WM, Dixon R, Ekis H, et al. The impact of preoperative CT angiography on breast reconstruction with abdominal perforator flaps. Ann Plast Surg 2012;68:525-30. [PubMed]
- Casey WJ 3rd, Chew RT, Rebecca AM, et al. Advantages of preoperative computed tomography in deep inferior epigastric artery perforator flap breast reconstruction. Plast Reconstr Surg 2009;123:1148-55. [PubMed]
- Fansa H, Schirmer S, Frerichs O, et al. Significance of abdominal wall CT-angiography in planning DIEA perforator flaps, TRAM flaps and SIEA flaps. Handchir Mikrochir Plast Chir 2011;43:81-7. [PubMed]
- Gacto-Sánchez P, Sicilia-Castro D, Gómez-Cía T, et al. Computed tomographic angiography with VirSSPA three-dimensional software for perforator navigation improves perioperative outcomes in DIEP flap breast reconstruction. Plast Reconstr Surg 2010;125:24-31. [PubMed]
- Ghattaura A, Henton J, Jallali N, et al. One hundred cases of abdominal-based free flaps in breast reconstruction. The impact of preoperative computed tomographic angiography. J Plast Reconstr Aesthet Surg 2010;63:1597-601. [PubMed]
- Malhotra A, Chhaya N, Nsiah-Sarbeng P, et al. CT-guided deep inferior epigastric perforator (DIEP) flap localization -- better for the patient, the surgeon, and the hospital. Clin Radiol 2013;68:131-8. [PubMed]
- Minqiang X, Lanhua M, Jie L, et al. The value of multidetector-row CT angiography for pre-operative planning of breast reconstruction with deep inferior epigastric arterial perforator flaps. Br J Radiol 2010;83:40-3. [PubMed]
- Rozen WM, Anavekar NS, Ashton MW, et al. Does the preoperative imaging of perforators with CT angiography improve operative outcomes in breast reconstruction? Microsurgery 2008;28:516-23. [PubMed]
- Rozen WM, Ashton MW, Grinsell D, et al. Establishing the case for CT angiography in the preoperative imaging of abdominal wall perforators. Microsurgery 2008;28:306-13. [PubMed]
- Rozen WM, Ashton MW, Stella DL, et al. The accuracy of computed tomographic angiography for mapping the perforators of the DIEA: a cadaveric study. Plast Reconstr Surg 2008;122:363-9. [PubMed]
- Schaverien MV, Ludman CN, Neil-Dwyer J, et al. Contrast-enhanced magnetic resonance angiography for preoperative imaging of deep inferior epigastric artery perforator flaps: advantages and disadvantages compared with computed tomography angiography: a United Kingdom perspective. Ann Plast Surg 2011;67:671-4. [PubMed]
- Smit JM, Dimopoulou A, Liss AG, et al. Preoperative CT angiography reduces surgery time in perforator flap reconstruction. J Plast Reconstr Aesthet Surg 2009;62:1112-7. [PubMed]
- Uppal RS, Casaer B, Van Landuyt K, et al. The efficacy of preoperative mapping of perforators in reducing operative times and complications in perforator flap breast reconstruction. J Plast Reconstr Aesthet Surg 2009;62:859-64. [PubMed]
- Rozen WM, Phillips TJ, Stella DL, et al. Preoperative CT angiography for DIEP flaps: ‘must-have’ lessons for the radiologist. J Plast Reconstr Aesthet Surg 2009;62:e650-1. [PubMed]
- Pacifico MD, See MS, Cavale N, et al. Preoperative planning for DIEP breast reconstruction: early experience of the use of computerised tomography angiography with VoNavix 3D software for perforator navigation. J Plast Reconstr Aesthet Surg 2009;62:1464-9. [PubMed]
- Rosset A, Spadola L, Ratib O. OsiriX: an open-source software for navigating in multidimensional DICOM images. J Digit Imaging 2004;17:205-16. [PubMed]
- Rozen WM, Chubb D, Ashton MW, et al. Achieving high quality 3D computed tomographic angiography (CTA) images for preoperative perforator imaging: now easily accessible using freely available software. J Plast Reconstr Aesthet Surg 2011;64:e84-6. [PubMed]
- Moon HK, Taylor GI. The vascular anatomy of rectus abdominis musculocutaneous flaps based on the deep superior epigastric system. Plast Reconstr Surg 1988;82:815-32. [PubMed]


