Surgical approaches for liver metastases in carcinoid tumors
Introduction
Neuroendocrine tumors (NETs) are a broad family of tumors which originate from cells of nervous and endocrine systems. Most of these tumors arise from small bowel, large bowel, or the pancreas. More than half of Gastro-entero-pancreatic (GEP) NETs are non-functioning tumors which secret no hormones or biologically inactive hormones, and remain symptomatically silent until a large tumor volume leads to non-specific symptoms such as jaundice, intestinal obstruction, abdominal pain etc. In these tumors morbidity and mortality mainly result from tumor mass or distant metastases. On the other hand, Some NETs are functioning tumors which can distinctively secret several biologically active hormones or peptides. Primary tumor location in the duodenum or pancreas, the excess hormone production, the multitude of hepatic lesions, and ultimate liver disease were noted to be an indicators of poor prognosis (1). These tumors morbidity and mortality are the results of hormonal or hormone-related symptoms, but can also be the consequence of problems caused by tumor expansion. Plasma Chromgranin A (CgA) is a common tumor marker and it should be measured in every patient with a suspected NETs. Sensitivity and specificity of CgA for the detection of NET is 68% and 100%, respectively (2,3).
The liver is a common site for NETs metastasis. Surgical treatment of NETs is strictly related to the localization of NET, the grade of tumor, and the stage of disease. NETs can be classified by extent of disease as having either local, regional, or distant involvement. The grade of a tumor depends on the rate of proliferation, which is measured by the percentage of tumor cells that are positive for the Ki-67 antigen. Low and intermediate grade tumors (G1 and G2) are characterized by low proliferative indices (Ki-67 <20%) and are thus indolent in nature. High-grade tumors (G3) have high proliferative indices (Ki-67 >20%), and are considered very aggressive (4). The European Neuroendocrine Tumor Society (ENETS) and the American Joint Committee on Cancer classify NETs by primary tumor (T), lymph node (N) involvement, and distant metastasis (M). Staging relies mainly on the size of the tumor and the extent of invasion into anatomical structures (5). Furthermore, The North American Neuroendocrine Tumor Society (NANETS) recommends reporting the TNM stage of the tumor in the pathology report, which helps physicians to select the best of the available treatment options.
A multidisciplinary approach involves a liver surgeon, hepatobiliary radiologist, and a medical oncologist is usually recommended to validate the management strategy. Different treatment options including surgery, regional and systemic biological therapy, and ablation with a variety of interventional radiology procedures have been proposed. Recently, radiofrequency ablation (RFA) of liver metastases has developed into a valuable treatment option. It can be performed using open, percutaneous, and laparoscopic approaches. Percutaneous RFA is the least invasive technique, however, intrahepatic and neighboring structures damage can occur. Laparoscopy is thought to be the best of the aforesaid techniques. It offers a less invasive method and a full visualization of the access of the RFA device at the surface of the liver, allowing the surgeon to hold away adjacent structures and organs (6).
In spite of diverse complex management strategies for neuroendocrine liver metastases, surgery remains the only potentially curative option. This discussion aims to critically review the existing and available information on surgical management of NETs liver metastases.
Open vs. laparoscopic surgical resection
Aggressive surgical resection is often the treatment of choice for most of NETs liver metastases. Although unilobar metastases have a better prognosis when compared to bilobar metastases, different aspects of the disease including primary tumor site, histological grade, and other metastatic sites play a role in the overall survival. Surgical resectability is a primary concern, as neuroendocrine liver metastases are frequently numerous and bulky, and it can be confirmed based on anatomical feasibility and volumetric tolerance. Furthermore, saving a liver remnant with sufficient function for life maintenance, low perioperative morbidity and mortality, and acceptable long-term survival is one of the major challenges facing surgeons. Non-pathological liver has an excellent regeneration capacity after resection, and a remnant functional amount of 20% to 25% normal liver tissue may be adequate. Postoperative liver failure has been demonstrated when the remaining functional liver ratio drops to less than 0.5% of total body weight. Liver volumes are usually measured using multidetector computed tomography (CT) of the liver. The majority of surgeons will not proceed with surgery if the expected volume of effective remnant liver is either less than 20-25% of the total liver or less than 0.5% of total body weight. In unilobar metastases, Portal embolization technique can minimize the risk of postoperative liver failure by raising the mean gain in liver volume to over 40%. The principle lies in compensatory hypertrophy of the opposite functional lobe expanding the volumetric feasibility for resection of hepatic metastases (7,8). In patients with bilobar liver metastases, the two-stage hepatectomy technique is considered an alternative to portal embolization. The left lobar metastases are first resected followed by those in the right lobe. The right portal vein is ligated in the initial stage of the surgery to induce hypertrophy of the left lobe. In the second stage, a right hepatectomy is completed with the hypertrophied left lobe supporting postoperative liver function (9). Furthermore, major hepatic transection and placement of endovascular staplers should be preceded by Glisson’s capsule incision using ultrasonic dissector (Figure 1).
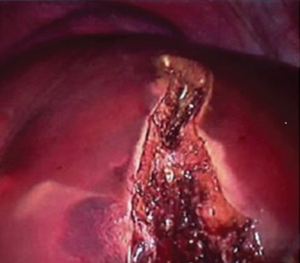
Resection of the primary tumor, liver metastases, and local mesenteric lymph node metastases is thought to strictly promote long-term survival and quality of life. Recently, Mayo et al. reported a multi-institutional experience of surgical management of NET liver metastases. Out of 399 patients included in this study, 77.6% underwent surgical resection of the NET liver metastases, 19.5% underwent resection plus ablation, and in 2.9%, underwent ablation only. A curative resection (R0 status) was successfully completed in 53.7% of the patients. A multivariate analysis of the same study reveals that concomitant extrahepatic disease, functional NETs, and presence of synchronous disease are all negative prognostic factors. The overall 5- and 10-year survivals were 74% and 51%, respectively (10). Additionally, if preoperative imaging shows that the primary and regional diseases are controlled and 90% or more of all hepatic metastases can be resected, cytoreductive hepatic surgery is recommended before resection for palliative causes. Significant reductions in biomarkers are associated with symptoms relief and disease control, nevertheless, local recurrence is evident in up to 94% at 5 years after (11).
Operative management with somatostatin analogues (SSA) and prophylactic cholecystectomy are recommended to avoid carcinoid crisis and SSA induced gallstone disease (12).
Recently, the emergence of minimally invasive surgery has had a massive effect on surgical practice. In Laparoscopic hepatectomy, Patient is positioned supine with the surgeons standing on opposite sides. Typically, the primary surgeon is on the patient’s left for right sided lesions, to the patient’s right for left sided lesions. These positions facilitate the inferior retraction of the liver for incision and division of the coronary attachments. Optimal mobilization and dissection of the liver is achieved by placing a 30° laparoscope in the 12-mm periumbilical port and placing other ports superiorly in the abdomen. An intraperitoneal pressure of 12 to 14 mmHg and a differential pressure with the central venous pressure of 5 to 8 mmHg are used to maintain Pneumoperitoneum. Using laparoscopic liver ultrasonography, we evaluate the extent of intraparenchymal lesions, rule out the presence of unknown lesions, and recognize major intrahepatic vascular and biliary structures. The gastrohepatic ligament and the liver are lifted up by holding the falciform ligament and gallbladder fundus to obtain an access to the foramen of Winslow. Through the right and left lateral ports, atraumatic graspers are introduced to hold a 5-mm nylon tape which is applied around the portal triad and then passed through an 8- to 10-cm-long 14-French plastic tube, with the ends brought out next to the right lateral port. Parenchymal transection is achieved by means of harmonic scalpel (Harmonic ACE; Ethicon Endo-Surgery) in combination with a Cavitron Ultrasonic Surgical Aspirator (Valleylab). These allow safe dissection of the biliovascular structures, which are then divided using Hem-o-Lock clips (Weck Closure Systems), or endoscopic staplers as required. Resection margins are evaluated vigilantly for hemorrhage or bile loss. Any bile leakage or bleeding is controlled using monopolar or bipolar diathermy for small bleeding points and with clips and Prolene sutures for any substantive bleeding or bile leakage. The Pringle maneuver is used intermittently (10-15 minutes on and 5 minutes off), usually when more than two segments are excised. As an end point, the central venous pressure and blood pressure are restored to normal physiological parameters and a Valsalva maneuver is performed to confirm hemostasis. The specimen is removed in impermeable bags (Endocatch; Ethicon Endo-Surgery) introduced through a 10 cm suprapubic incision (13).
Open liver resection requires a 20-40 cm incision in the upper abdomen, whereas Laparoscopic hepatectomy is technically promising with new instruments that permit resection through a transverse 10 cm supra-pubic incision. However, pneumoperitoneum due to cardiopulmonary status and adhesions that cannot be lysed are an absolute contraindication for laparoscopic approach. Relative contraindications include lesions adjacent to the inferior vena cava or major vessels, and cases in which there is a hilar lesion or a need for biliary or vascular reconstruction (13). Laparoscopic approach is superior to open approach in terms of magnification, which allows meticulous exposure of the small vessels under pneumoperitoneum without damaging them, total operative time, and hospital stays (14). In open hepatectomy, the bleeding point can be ligated and sutured directly to stop the bleeding. Laparoscopic ligation and suturing, however, are not simple. Dissection around the blood vessels should be managed carefully to avoid hemorrhage from major vessels. One potential drawback for laparoscopic hepatectomy is restrictive manipulation. Blind manipulation must never be challenged. Surgeons usually overcome this problem with the assistance of energy devices and laparoscopic magnification which permit clearer access to the liver and prevent major vessels injury (14,15).
Nevertheless, it is impossible to reach a precise conclusion regarding superiority of laparoscopic hepatectomy over open hepatectomy. Laparoscopic hepatectomy may be superior in such short-term factors as the complication rate, duration of hospitalization, amount of hemorrhage, blood transfusion rate. Long-term factors such as overall survival and disease-free survival may not be inferior. Randomized Clinical Trials are warranted to further evaluate the benefits and limitations of both approaches.
Liver transplantation
The development of new alternative medical and surgical treatment options is the main obstacle facing the adoption of liver transplant in the treatment of unresectable metastases from NETs. It has been suggested that liver transplant should be an option when other treatments fails to eradicate the disease (16). Intractable carcinoid syndrome and hepatic failure Symptoms are all indications for liver transplantation. However, the selection criteria for transplantation should assure that the clinical improvement in patient’s quality of life must exceed the considerable risks of both the liver transplant and the immunosuppression (17,18).
Liver transplantation for NETs is one of few indications for transplant in the setting of metastatic disease. In 1998, Lehnert analyzed a total of 103 patients transplanted for metastatic neuroendocrine carcinoma. The overall 5-year survival was 47%, and disease-free survival was 24%. On 2007, Mazzaferro et al. has reported an overall survival of 80% with the strict “Milan criteria” for selection of patients who can undergo liver transplant. These guidelines considered young patients (<55 years old) who had well-differentiated endocrine tumors with low-grade malignancy based on the basis of mitotic and proliferation indices as eligible candidates for liver transplantation (Table 1) (19). However, a more recent multicenter French study of 85 patients who underwent liver transplant for NETs liver metastases, including symptomatic patients with extensive liver involvement, reported an overall survival of 47% and a recurrence free survival of 20% at 5 years interval (17). Finally, according to ENETS 2012 guidelines, minimal requirements for consideration for liver transplant should include well-differentiated NET with a Ki-67 index of <10%, age <55 years, diffuse unresectable disease confined to the liver with absence of extrahepatic disease, primary tumor removed before transplantation (at least 6 months), stable disease for at least 6 months before Liver Transplant, and <50% liver involvement (<75% for patients with refractory hormonal symptoms) (Table 2) (20).

Full table

Full table
Advances in surgical techniques and improved perioperative care have made liver transplantation safer recently. However, according to the United Network for Organ Sharing database, only 185 liver transplants were performed for metastatic NETs in the USA, between November 1988 and March 2011, with an overall 5-year survival of 57.8%. Recurrent liver disease, shortage of donor organs, and poor selection criteria all contribute to the controversy about effectiveness of liver transplant in patients with unresectable NETs liver metastases.
Conclusions
Surgical treatment of NETs liver metastases plays a central role in the multimodal management of these tumors. Before planning surgical approach, tumor characteristic symptoms, distribution of the metastases, and the histological features should be always considered along with patients’ co-morbidities and expectations. An aggressive surgical approach can lead to long-term survival with a good quality of life. Nevertheless, the laparoscopic option by experienced laparoscopic liver surgeons can be safe, feasible and provides earlier recovery and fewer complications. Liver transplant might be considered in carefully selected patients who fail to respond to other treatment options. Additional future randomized clinical trials are warranted to identify the group of patients who will most benefit from each approach.
Acknowledgements
This research and work was fully supported by Tulane University Medical Center.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Anthony LB, Strosberg JR, Klimstra DS, et al. The NANETS consensus guidelines for the diagnosis and management of gastrointestinal neuroendocrine tumors (nets): well-differentiated nets of the distal colon and rectum. Pancreas 2010;39:767-74. [PubMed]
- O'Connor DT, Pandlan MR, Carlton E, et al. Rapid radioimmunoassay of circulating chromogranin A: in vitro stability, exploration of the neuroendocrine character of neoplasia, and assessment of the effects of organ failure. Clin Chem 1989;35:1631-7. [PubMed]
- Eriksson B, Arnberg H, Oberg K, et al. A polyclonal antiserum against chromogranin A and B--a new sensitive marker for neuroendocrine tumours. Acta Endocrinol (Copenh) 1990;122:145-55. [PubMed]
- Klimstra DS, Modlin IR, Coppola D, et al. The pathologic classification of neuroendocrine tumors: a review of nomenclature, grading, and staging systems. Pancreas 2010;39:707-12. [PubMed]
- Rindi G, Arnold R, Bosman FT, et al. Nomenclature and classification of neuroendocrine neoplasms of the digestive system. In: Bosman FT, Carneiro F, Hruban RH, et al. editors. WHO Classification of Tumours of the Digestive System. Lyon, France: International Agency for Research on cancer (IARC), 2010:S1-S14.
- Mulier S, Mulier P, Ni Y, et al. Complications of radiofrequency coagulation of liver tumours. Br J Surg 2002;89:1206-22. [PubMed]
- Kianmanesh R, Farges O, Abdalla EK, et al. Right portal vein ligation: a new planned two-step all-surgical approach for complete resection of primary gastrointestinal tumors with multiple bilateral liver metastases. J Am Coll Surg 2003;197:164-70. [PubMed]
- Boleslawski E, Dharancy S, Truant S, et al. Surgical management of liver metastases from gastrointestinal endocrine tumors. Gastroenterol Clin Biol 2010;34:274-82. [PubMed]
- Kianmanesh R, Sauvanet A, Hentic O, et al. Two-step surgery for synchronous bilobar liver metastases from digestive endocrine tumors: a safe approach for radical resection. Ann Surg 2008;247:659-65. [PubMed]
- Mayo SC, de Jong MC, Bloomston M, et al. Surgery versus intra-arterial therapy for neuroendocrine liver metastasis: a multicenter international analysis. Ann Surg Oncol 2011;18:3657-65. [PubMed]
- Steinmüller T, Kianmanesh R, Falconi M, et al. Consensus guidelines for the management of patients with liver metastases from digestive (neuro)endocrine tumors: foregut, midgut, hindgut, and unknown primary. Neuroendocrinology 2008;87:47-62. [PubMed]
- Benkel M, Brasch F, Neumann JD, et al. Poorly differentiated neuroendocrine small-cell carcinoma of the gallbladder. Zentralbl Chir 2012;137:71-2. [PubMed]
- Buell JF, Cherqui D, Geller DA, et al. The international position on laparoscopic liver surgery: The Louisville Statement, 2008. Ann Surg 2009;250:825-30. [PubMed]
- Kandil E, Noureldine SI, Koffron A, et al. Outcomes of laparoscopic and open resection for neuroendocrine liver metastases. Surgery 2012;152:1225-31. [PubMed]
- Reddy SK, Tsung A, Geller DA. Laparoscopic liver resection. World J Surg 2011;35:1478-86. [PubMed]
- Ramage JK, Ahmed A, Ardill J, et al. Guidelines for the management of gastroenteropancreatic neuroendocrine (including carcinoid) tumours (NETs). Gut 2012;61:6-32. [PubMed]
- Le Treut YP, Grégoire E, Belghiti J, et al. Predictors of long-term survival after liver transplantation for metastatic endocrine tumors: an 85-case French multicentric report. Am J Transplant 2008;8:1205-13. [PubMed]
- Frilling A, Clift AK. Therapeutic strategies for neuroendocrine liver metastases. Cancer 2015;121:1172-86. [PubMed]
- Mazzaferro V, Pulvirenti A, Coppa J. Neuroendocrine tumors metastatic to the liver: how to select patients for liver transplantation? J Hepatol 2007;47:460-6. [PubMed]
- Pavel M, Baudin E, Couvelard A, et al. ENETS Consensus Guidelines for the management of patients with liver and other distant metastases from neuroendocrine neoplasms of foregut, midgut, hindgut, and unknown primary. Neuroendocrinology 2012;95:157-76. [PubMed]


