
Abnormality of pl6/p38MAPK/p53/Wipl pathway in papillary thyroid cancer
Introduction
Wild-type p53 induced phosphatase 1 (Wip1) is a member of the PP2C family of evolutionarily conserved protein phosphatases and a novel proto-oncogene (1,2). Originally described as a p53-regulated gene. In recent years, analysis of Wip1 has focused primarily on its role in tumorigenesis because of its overexpression in human tumors. Overexpression or activation of Wip1, as commonly seen in primary human tumors, could have a more general effect on multiple signaling pathways contributing to deregulation of several molecular networks.
p53 and p16 are key anti-oncogenes, whereas p38 mitogen-activated protein kinase (MAPK) is a key cell cycle regulator (3). Wip1 is a major inhibitor of p53 functions. The mutation or missing of p53 gene is the cause of many tumors (4) which is a main carcinogenic mechanism of Wip1. Wip1 has been implicated as a negative regulator of p53 via its ability to attenuate p38 MAPK activity (5,6). p16, also known as mitotic inhibitor or multiple tumor suppressor gene, is one of the cell cycle regulator. p16 is a key anti-oncogene. Moreover, higher Wip1 expression is associated with lower p16 levels in primary human mammary carcinomas (7). And Wip1 can potently down-regulate p16 expression by suppressing p38MAPK (8). However the downregulation of p53 and the DNA damage and DNA repair responses by WIP1 has cancer implications, particularly since the DNA damage response has recently been shown to play a crucial role as an early anti-cancer barrier (9-11). It is known that p38 mitogen-activated protein (MAP) kinase controls cellular pathways for proliferation, differentiation, development of the inflammatory response, and apoptosis. Wip1 also negatively regulates the stress responsive p38 MAPK pathway by directly inactivating p38 through dephosphorylation (12). However phosphorylated p38 MAP kinase phosphorylates and activates p53 to cause cell cycle arrest or apoptosis. And Wip1 can control a feedback loop in the p38 MAP kinase-p53 signaling pathway (12). Thus, pl6/p38MAPK/p53/Wipl signal transduction pathways may play a crucial role in human tumors.
Not surprisingly, Wip1 has been implicated as an oncogene and Wip1 gene amplification and/or protein overexpression have been reported in a variety of human tumors including breast cancer, neuroblastoma, pancreatic cancer, ovarian cancer, and gastric cancer (7,13). However, its expression in papillary thyroid carcinoma (PTC) remains unclear. Up to now few studies have explored the role of pl6/p38MAPK/p53/Wipl pathway in the occurrence and development of PTC. In this study, by detecting the protein expressions of Wipl, p53, p38MAPK, and p16 in 70 cases of PTC tissues and 20 cases of normal thyroid tissues using immunohistochemical staining and analyzing their mutual correlation, we investigated the role of pl6/p38MAPK/p53/Wipl pathway in PTC. To our knowledge, this is the first report establishing the existence of the signaling pathway in PTC and may be a potential target for therapeutic intervention for treatment of PTC.
Materials and methods
Specimens and general information
PTC group
The specimens and clinical data of 70 patients with pathologically confirmed PTC who were treated in our hospital from March 2010 to December 2010 were retrospectively collected. There were 16 males and 54 females aged 15-64 years (mean: 41 years). Lymph node metastasis was noted in 92 patients, while 37 patients had no lymph node metastasis. TNM staging showed 39 cases in stage I, 12 in stage II, 14 in stage III, and 5 in stage IV. All patients were naive to chemotherapy and radiotherapy before the surgery. PTC specimens were obtained from these tissues immediately after surgical resection.
Control group
Pathologically confirmed normal thyroid tissues were obtained from 20 cases. There were 4 males and 16 females aged 20-55 years (mean: 42.3 years).
Main reagents
The main reagents included rabbit anti-human Wip1 polyclonal antibody (Santa Cruze); rabbit anti-human p-p38MAPK monoclonal antibody (Cell Signaling); and mouse anti-human p53/p16 monoclonal antibody and streptavidin-proxidase (SP) reagent kit (Fuzhou Maxim Biotech Inc., China).
Experimental methods
Immunohistochemical staining
Specimens were fixed in 10% neutral buffered formalin, embedded in paraffin, cut into 4 µm sections and stained with hematoxylin and eosin (HE) or SP methods. Known positive sections were used as a positive control and phosphate buffer saline (PBS) was substituted for the primary antibody for a negative control.
Determination of results
The positive protein expression of Wip1 was mostly in nucleus and only a few in cytoplasm. Positive protein expressions of p-p38MAPK, p53, and p16 were found in the nucleus as brown/yellow granules (diamino benzidine (DAB) staining). The numbers of positive cells in five random high-definition fields under the light microscope (×400) were then counted, and their proportions relative to the total cells were calculated. Wip1: a stained cell count =0 or <10% as negative (–), 10-30% as (+), and >30% as (++); negative (–) to (+) was regarded as low expression and (++) as high expression. p53, p38, and p16: a stained cell count =0 or <10% as negative (–) and ≥10% as positive (+).
Statistical analysis
Data were analyzed using SPSS 13.0 software. Rates were compared using Chi square test and correlation was analyzed with Spearman's rank correlation coefficient. P<0.05 was considered significantly different.
Results
Expression of Wip1 in normal thyroid tissues and PTC tissues
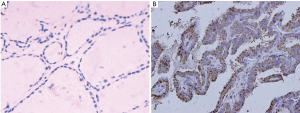
The immunohistochemical staining of Wip1 was negative or weak in the follicular epithelial cells of normal thyroid tissues (Figure 1A), whereas the positive expression of Wip1 in PTC tissues was mostly in nucleus (Figure 1B). The high-expression rate of Wipl protein in the PTC tissue was 64.3% (45/70), which was significantly higher than that in normal thyroid tissue (0/20) (χ2=25.714, P=0.00039).
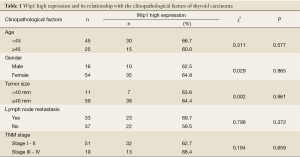
Wip1 high expression and its relationship with the clinopathological factors of thyroid carcinoma
Wip1 high expression showed no correlation with age, gender, tumor size, lymph node metastasis, and pathological stage (all P>0.05). These factors also showed no significant difference among the groups (χ2=0.311, 0.029, 0.002, 0.796, and 0.194, respectively; P=0.577, 0.865, 0.961, 0.372, and 0.659, respectively) (Table 1).
Full table
Protein expressions of p38MAPK, p53, and p16 in PTC tissues their correlations with the Wip1 high expression
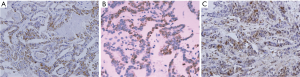
The positive protein expressions of p38, p53, and p16 in PTC tissues were all in nucleus (Figure 2). The Wipl protein high-expression was negatively correlated with p53, p38MAPK, and pl6 protein expressions (Table 2).

Full table
Discussion
Previous study has shown that the occurence and development of human tumors are closely associated with the changes in intracellular genome, among which the activation of proto-oncogenes, the inactivation or missing of anti-oncogenes, and the changes in the functions of many cell cycle regulatory genes play key roles (14). As shown in our current study, the high-expression rate of Wipl protein in the PTC tissue was 64.3% (45/70), which was significantly higher than that in normal thyroid tissue (P<0.01), indicating that Wip1 is involved in the progression of PTC. Correlation analysis showed that Wip1 high expression was not correlated with the clinopathological factors including age, gender, tumor size, lymph node metastasis, and pathologic stage, which are consistent with our previous findings (15). Therefore, Wip1 can not be used to predict the prognosis.
Also in our study, the high protein expression of Wip1 in PTC tissues was negatively correlated with the protein expressions of p38MAPK, p53, and p16; in other words, PTC tissues with Wip1 high protein expression were often associated with the depletion of the protein expression of p38MAPK, p53, or p16, and vice versa. As a stress regulator, Wip1 can inactivate p38MAPK (6,16). p38MAPK, a key cell cycle regulator, plays an important role in controlling the G1/S and G2/M checkpoints; the inactivation of p38MAPK increases the risk of tumorigenesis (3). p53, as one of the most important anti-oncogenes, plays an important role in the apoptosis induced by DNA damage (17,18). p53 has two subtypes: wild type and mutant type. The subtype detected by immunohistochemical methods is mainly mutant type. As shown in our study, by inhibiting the functions of p53, Wip1 reduced the selective mutation of p53 gene.
As a key anti-oncogene, p16 can specifically inhibit CDK4/6 and thus prevent the transition of a cell from G1 to S phase. It adjusts the mitosis via negative feedback and prevents the abnormal proliferation of cells. And its inactivation or missing can dramatically accelerate the growth of cells, therefore play a crucial role in the progression of PTC.
In addition, we found that the pl6/p38MAPK/p53/Wipl pathway is abnormal in PTC, and this abnormality may possibly be associated with the aberrantly up-regulated Wipl, which can induce inhibition of p38MAPK, p53 and p16. It is therefore speculated that Wip1 may be a therapeutic target for PTC; particularly, the inhibition of Wip1 function may enhance the activities of anti-oncogenes and thus exert therapeutic effect on PTC. Research also has shown that arsenic trioxide can inhibit Wip1, during which Wip1 is served as the direct molecular target. Currently, arsenic trioxide is mainly used for the treatment of acute promyelocytic leukemia (19). The prevalence of thyroid cancer has shown an increasing trend in China in recent years (20); unfortunately, this disease is not sensitive to conventional chemotherapy drugs.
In conclusions, the pl6/p38MAPK/p53/Wipl pathway is abnormal in PTC, and this abnormality may possibly be associated with the aberrantly up-regulated Wipl, which can induce inhibition of p38MAPK, p53 and pi6. The arsenic trioxide may exert its anti-tumor efficacy in the targeted therapy for thyroid cancer.
Acknowledgements
Disclosure: The authors declare no conflict of interest.
References
- Bulavin DV, Demidov ON, Saito S, et al. Amplification of PPM1D in human tumors abrogates p53 tumor-suppressor activity. Nat Genet 2002;31:210-5. [PubMed]
- Li J, Yang Y, Peng Y, et al. Oncogenic properties of PPM1D located within a breast cancer amplification epicenter at 17q23. Nat Genet 2002;31:133-4. [PubMed]
- Bulavin DV, Higashimoto Y, Popoff IJ, et al. Initiation of a G2/M checkpoint after ultraviolet radiation requires p38 kinase. Nature 2001;411:102-7. [PubMed]
- Peng Liu, Yan Liu, Hui Sun. Correlation between expression of p53 and bcl-2/bax in breast cancer. Chinese Journal of Experimental Surgery 2007;24:176-8.
- Bulavin DV, Phillips C, Nannenga B, et al. Inactivation of the Wip1 phosphatase inhibits mammary tumorigenesis through p38 MAPK-mediated activation of the p16(Ink4a)-p19(Arf) pathway. Nat Genet 2004;36:343-50. [PubMed]
- Lu X, Nguyen TA, Moon SH, et al. The type 2C phosphatase Wip1: an oncogenic regulator of tumor suppressor and DNA damage response pathways. Cancer Metastasis Rev 2008;27:123-35. [PubMed]
- Yu E, Ahn YS, Jang SJ, et al. Overexpression of the wip1 gene abrogates the p38 MAPK/p53/Wip1 pathway and silences p16 expression in human breast cancers. Breast Cancer Res Treat 2007;101:269-78. [PubMed]
- Lee JS, Lee MO, Moon BH, et al. Senescent growth arrest in mesenchymal stem cells is bypassed by Wip1-mediated downregulation of intrinsic stress signaling pathways. Stem Cells 2009;27:1963-75. [PubMed]
- Halazonetis TD, Gorgoulis VG, Bartek J. An oncogene-induced DNA damage model for cancer development. Science 2008;319:1352-5. [PubMed]
- Bartkova J, Horejsí Z, Koed K, et al. DNA damage response as a candidate anti-cancer barrier in early human tumorigenesis. Nature 2005;434:864-70. [PubMed]
- Nguyen TA, Slattery SD, Moon SH, et al. The oncogenic phosphatase WIP1 negatively regulates nucleotide excision repair. DNA Repair (Amst) 2010;9:813-23. [PubMed]
- Yamaguchi H, Durell SR, Feng H. Development of a substrate-based cyclic phosphopeptide inhibitor of protein phosphatase 2Cdelta, Wip1. Biochemistry 2006;45:13193-202. [PubMed]
- Le Guezennec X, Bulavin DV. WIP1 phosphatase at the crossroads of cancer and aging. Trends Biochem Sci 2010;35:109-14. [PubMed]
- Bo Zhang, Jian-hua Lu, Ke Liu, et al. DBC2 negatively regulates the proliferation of breast cancer cells and induces apoptosis. Chinese Journal of Experimental Surgery 2010;27:345-7.
- Yang DH, He JA, Li J, et al. Expression of proto-oncogene Wipl in breast cancer and its clinical significance. National Medical Journal of China 2010;90:519-22. [PubMed]
- Fuku T, Semba S, Yutori H, et al. Increased wild-type p53-induced phosphatase 1 (Wip1 or PPM1D) expression correlated with downregulation of checkpoint kinase 2 in human gastric carcinoma. Pathol Int 2007;57:566-71. [PubMed]
- Liu G, Chen X. Regulation of the p53 transcriptional activity. J Cell Biochem 2006;97:448-58. [PubMed]
- Gorgoulis VG, Vassiliou LV, Karakaidos P, et al. Activation of the DNA damage checkpoint and genomic instability in human precancerous lesions. Nature 2005;434:907-13. [PubMed]
- Yoda A, Toyoshima K, Watanabe Y, et al. Arsenic trioxide augments Chk2/p53-mediated apoptosis by inhibiting oncogenic Wip1 phosphatase. J Biol Chem 2008;283:18969-79. [PubMed]
- Wang Y, Wang W. Increasing Incidence of Thyroid Cancer in Shanghai, China, 1983-2007. Asia Pac J Public Health 2012; [PubMed]


