Oncoplastic volume replacement technique for the upper inner quadrant using the omental flap
Introduction
Oncoplastic breast-conserving surgery (BCS) can be classified as volume displacement or volume replacement. Many local and distant flaps have been developed for volume replacement. The LD flap plays a major role in volume replacement (1,2), but has the major disadvantages of donor site morbidity and deformity such as postoperative seroma formation and impairment of shoulder function (3,4).
Lateral chest wall perforator flaps have been described to minimize these problems, including use of the thoracodorsal artery perforator (TDAP) flap, and the lateral intercostal artery perforator (LICAP) flap (5). However, it is generally difficult to utilize these perforator flaps in partial breast reconstruction for medial located tumors because adequate mobilization and a longer pedicle are needed to reach and replace the distant defect of the breast tissue (6,7).
Defects in the lower inner quadrant can be addressed using the abdominal adipofascial flap (8,9) and the anterior intercostal artery perforator (AICAP) flap (10). However, it is still difficult to utilize these flaps for the upper inner quadrant (UIQ) which constitutes a major part of a no man’s land of the breast.
Since April 2002, we have performed more than 170 cases of immediate breast reconstruction with a laparoscopically harvested omental flap (OF) (11,12), and previously reported the results of partial reconstruction for the lower inner quadrant (13). Here, we present the oncological and cosmetic outcomes in a series of 30 of partial breast reconstruction with the OF for the most difficult quadrant; the UIQ of the breast.
Materials and methods
Between April 2002 and December 2013, immediate partial breast reconstruction with the OF was performed for 30 patients with a tumor in the UIQ of the breast. All the tumors were evaluated preoperatively using mammography, ultrasound, and magnetic resonance imaging. Wide excisions (>20% of the breast tissue) were planned to achieve negative margins. The procedure was not performed in patients with a history of intraabdominal malignancy or upper abdominal laparotomy. However, patients with a history of laparoscopic surgery such as laparoscopic cholecystectomy or lower abdominal surgery such as Caesarean section were not excluded from the indication. Patients with a body mass index (BMI) ≥35 kg/m2 were also excluded. The study was approved by the hospital ethics committee and all patients provided written informed consent.
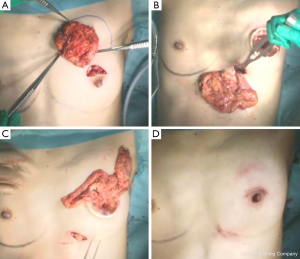
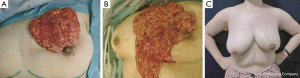
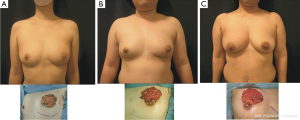
Surgery was performed in a supine position with the ipsilateral arm rested at 90° abduction. A periareolar incision was the first choice (Figures 1,2), however, when the skin over the tumor needed to be resected for an oncological reason, an elliptical radial or a transverse skin incision was chosen (Figure 3). The skin flap around the UIQ was then widely elevated to the lateral wedge of the sternum medially, and to the subclavicle cranially. A partial mastectomy was carried out, excising the tumor with a margin of at least 2 cm of normal breast tissue (Figures 1A,2A,3A). A 2- to 6-cm skin incision was made along the axillary skin crease and sentinel lymph node biopsy or axillary dissection was performed.
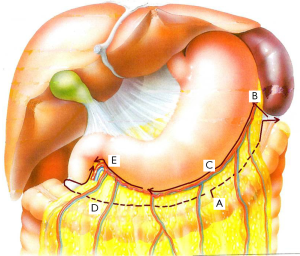
Laparoscopy for harvesting the OF was then performed, as described in detail elsewhere (12). First, the omentum was dissected from the transverse colon, and advanced leftward, and the left gastroepiploic vessels were divided near the spleen. The gastric branches of the gastroepiploic vessels were divided close to the stomach wall. The omentum was dissected till passing the pyloric ring (Figure 4). Roots of the gastroepiploic vessels were preserved as a pedicle and the OF was harvested in about one hour.
An additional 4 cm skin incision was made along the medial inframammary fold, and an approximately 2-finger wide subcutaneous tunnel was prepared toward the xyphoid process. When the tunnel reached the white line, a 2-finger wide longitudinal incision was made to communicate with the abdominal cavity. This was facilitated by intraabdominal resection of the white line. The forceps or fingers were inserted into the abdominal cavity via the tunnel and the pedicled OF was carefully withdrawn with avoidance of twisting (Figures 1B,2B,3B).
A subglandular tunnel which passed under the lower inner quadrant was also created between the inframammary incision and the partial mastectomy defect in the UIQ. After hemostasis was completed, the OF was pulled out and filled the defect (Figure 1C).
It was usually unnecessary to fix the OF to the chest wall. A closed suction drain was inserted over the OF and the incision was closed in two layers (Figure 1D). When the laparotomy incision was wider than 2-finger width, the wound was semi-closed to avoid postoperative ventral hernia. The pedicle of the flap was made as slim as possible by careful defatting, and the subcutaneous fat tissue around the tunnel was excised to avoid postoperative bulging. An entrance of the subcutaneous tunnel was also semi-closed for appropriate recreation of a medial site of the inframammary fold. The reconstructed OF was usually monitored with Doppler sonographic examination for a day or two days postoperatively.
Cosmetic results were evaluated using a cosmetic score (14) assessed by three health professionals and BCCT.core which is 2D computer software as excellent, good, fair, or poor (15).
Results
The characteristics of the 30 patients who underwent immediate volume replacement with the OF after resection of the UIQ of the breast are shown in Table 1. The mean pathological tumor size was 2.8 cm, and two patients had internal lymph node metastasis. The median resected breast tissue volume was 142 g. The OFs were harvested laparoscopically in about 1 hour without conversion to a laparotomy in all cases. The complication rate (including short- and long-term events) was 10.0%. They were two cases of partial flap necrosis and one case of ventral hernia. Partial flap necrosis could be treated conservatively, but caused prolonged necrotic drain discharge which resulted in loss of nearly a half of the volume and lately formed a hard nodule in the reconstructed breast because of calcified fat necrosis. Ventral hernia in infra-xyphoid area occurred in one patient 5 years after surgery, and repaired by laparoscopic herniorrhaphy with a mesh.
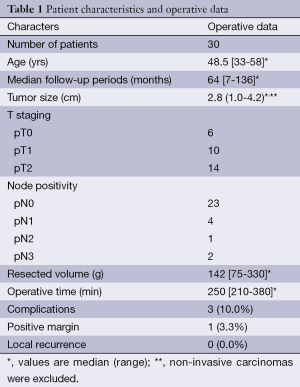
Full table
The surgical margins, defined negative as no ink on tumor were positive in one patient (3.3%) and the patient underwent re-excision (Table 1).
Neither local nor systemic recurrence has occurred to date in any patients during medial follow-up periods of 64 months (Table 1).
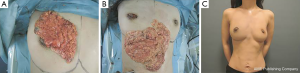
Cosmetic outcomes were mostly satisfactory with negligible donor-site scars (Figures 3,5). The scars on the medial inframammary fold were usually hidden by the breast and became unobvious because of positive effect of postoperative radiation therapy to avoid hypertrophic scar formation. More than 80% of the patients scored excellent or good both with panel assessment and BCCT.core (Table 2). Compared with the panel assessment, fewer patients were scored as excellent. No size reduction of the OF was noted during follow-up except in cases of partial flap necrosis, even after radiation therapy.
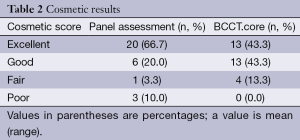
Full table
Discussion
Cosmetic outcome after BCS is negatively influenced by many factors, and medial tumor location is one of the reasons. Grisotti et al. defines the no man’s land that is the area upward to a line drawn on the inferior border of the pectoralis, around the areolar at a superior distance of 16 cm from the sternal notch and 7 cm medially (16), and the no man’s land is mainly constituted with the UIQ. A large skin resection in this area followed by dermoglandular flap advancement including the nipple- areolar complex will shift the nipple in an upward or medial fashion that would look highly unnatural in location (16). Defects in the UIQ are more likely to cause patient dissatisfaction (17,18). Patient outcomes following surgery can be enhanced by restoring volume and minimizing scars in the UIQ. For the patients whose breasts are large, several volume displacement techniques have been developed and well standardized to solve the problem (19). However, for the patients whose breasts are small, volume replacement technique is the only key to solve the problem.
The LD flap has been played a major role in volume replacement (1,2), but has the disadvantages of donor-site scar and morbidities (3,4). Recent advances in chest wall perforator flaps can minimize these problems; however, perforator flaps generally tend to have less range (20), although the TDAP flap replacement of volume is reported in all quadrants (21). The superior epigastric artery perforator (SEAP) flap can also reach and fill the defect in the UIQ, but usually results in very visible donor-site scarring (22).
We have already reported on partial reconstruction using the OF for the lower inner quadrant. The UIQ is also comfortable field for the OF which can easily reach any quadrants of the breasts (13). Although two separate incisions are needed for partial reconstruction for the UIQ, the small incision along the medial inframammary fold to extract the OF is usually invisible. When the volume of the OF is large, the subglangular tunnel should be wider by resecting breast and fat tissue around the tunnel to avoid bulging in the lower inner quadrant.
The big advantages of the OF are minimal donor-site scar and morbidities because the flap can be harvested laparoscopically (12). The scars in the abdomen are just like those of laparoscopic cholecystectomy. The short- and long-term laparoscopy-associated complication rates are very low and acceptable (12). Because the OF is very soft due to abundance of fat and has a long pedicle, it is easy to replace an irregular-shaped defect after BCS (13). A volume replacement procedure can be completed even through a small periareolar incision by which a visible scar in the no man’s land can be avoided. If the surrounding residual breast parenchyma and the inframammary fold are not dissected from the underlying muscles, a fixation of the OF is unnecessary and the OF naturally fits and fills the defect.
The most important disadvantage of the OF is an impossibility of preoperative volume estimation of the flap (11,13). When the resected volume is larger than 100g or the patient is slim, the volume of the OF may be inadequate. On the other hand, the volume of the OF may be very large enough for total reconstruction, however, it is usually difficult to extract an extremely large sized OF through the small subcutaneous tunnel, which might cause a trauma of small vessels of the flap and partial flap necrosis. Then a mid-line abdominal skin incision would be needed in such a case, which negatively affects donor-site cosmesis. Therefore when the volume of the OF is large, a free flap is better choice because a large sized OF can be easily extracted from the umbilical incision (23).
Cosmetic results were mostly satisfactory with natural soft tactile feeling of the reconstructed breast. However, compared with the panel assessment, fewer patients were scored as excellent in this study. One of reasons might be poor quality of our photographs because of lack of adequate lightning and standardization in taking photographs which are the necessary conditions for evaluating cosmetic outcome using BCCT.core (15).
The OF is strong against radiation therapy and less atrophic than the muscle flap, and the LD flap and the lower abdominal wall flap such as the transverse rectus abdominis myocutaneous (TRAM) flap and the deep inferior epigastric artery perforator (DIEP) flap can be completely preserved for total reconstructions in case. In conclusion, the OF is attractive for partial reconstruction after BCS for the UIQ; a part of the no man’s land of the breast.
Acknowledgements
Disclosure: The author declares no conflict of interest.
References
- Noguchi M, Taniya T, Miyazaki I, et al. Immediate transposition of a latissimus dorsi muscle for correcting a postquadrantectomy breast deformity in Japanese patients. Int Surg 1990;75:166-70. [PubMed]
- Munhoz AM, Montag E, Fels KW, et al. Outcome analysis of breast-conservation surgery and immediate latissimus dorsi flap reconstruction in patients with T1 to T2 breast cancer. Plast Reconstr Surg 2005;116:741-52. [PubMed]
- Schwabegger A, Ninković M, Brenner E, et al. Seroma as a common donor site morbidity after harvesting the latissimus dorsi flap: observations on cause and prevention. Ann Plast Surg 1997;38:594-7. [PubMed]
- Hamdi M, Decorte T, Demuynck M, et al. Shoulder function after harvesting a thoracodorsal artery perforator flap. Plast Reconstr Surg 2008;122:1111-7; discussion 1118-9. [PubMed]
- Hamdi M, Van Landuyt K, Monstrey S, et al. Pedicled perforator flaps in breast reconstruction: a new concept. Br J Plast Surg 2004;57:531-9. [PubMed]
- Kronowitz SJ, Feledy JA, Hunt KK, et al. Determining the optimal approach to breast reconstruction after partial mastectomy. Plast Reconstr Surg 2006;117:1-11; discussion 12-4. [PubMed]
- Rainsbury RM, Paramanathan N. Recent Progress with Breast-Conserving Volume Replacement Using Latissimus Dorsi Miniflaps in UK Patients. Breast Cancer 1998;5:139-47. [PubMed]
- Ogawa T, Hanamura N, Yamashita M, et al. Usefulness of breast-volume replacement using an inframammary adipofascial flap after breast-conservation therapy. Am J Surg 2007;193:514-8. [PubMed]
- Kijima Y, Yoshinaka H, Owaki T, et al. Immediate reconstruction using inframammary adipofascial flap of the anterior rectus sheath after partial mastectomy. Am J Surg 2007;193:789-91. [PubMed]
- Hamdi M, Van Landuyt K, de Frene B, et al. The versatility of the inter-costal artery perforator (ICAP) flaps. J Plast Reconstr Aesthet Surg 2006;59:644-52. [PubMed]
- Zaha H, Inamine S, Naito T, et al. Laparoscopically harvested omental flap for immediate breast reconstruction. Am J Surg 2006;192:556-8. [PubMed]
- Zaha H, Inamine S. Laparoscopically harvested omental flap: results for 96 patients. Surg Endosc 2010;24:103-7. [PubMed]
- Zaha H, Sunagawa H, Kawakami K, et al. Partial breast reconstruction for an inferomedial breast carcinoma using an omental flap. World J Surg 2010;34:1782-7. [PubMed]
- Harris JR, Levene MB, Svensson G, et al. Analysis of cosmetic results following primary radiation therapy for stages I and II carcinoma of the breast. Int J Radiat Oncol Biol Phys 1979;5:257-61. [PubMed]
- Cardoso JS, Cardoso MJ. Towards an intelligent medical system for the aesthetic evaluation of breast cancer conservative treatment. Artif Intell Med 2007;40:115-26. [PubMed]
- Grisotti A, Calanbrese C. Conservative treatment of breast cancer: reconstructive problem. In: Spear SL, Willy SC, Robb GL, et al. eds. Surgery of the breast. principles and art. 2nd ed. Philadelphia: Lippincott Williams & Wilkins, 2006:147-78.
- Gill K, Mann R, Brunsworth L, et al. Aesthetic subunit of the breast: an analysis of women's preference and clinical implications. Ann Plast Surg 2014;73:249. [PubMed]
- Kronowitz SJ, Kuerer HM, Buchholz TA, et al. A management algorithm and practical oncoplastic surgical techniques for repairing partial mastectomy defects. Plast Reconstr Surg 2008;122:1631-47. [PubMed]
- Fitzal F, Nehrer G, Hoch D, et al. An oncoplastic procedure for central and medio-cranial breast cancer. Eur J Surg Oncol 2007;33:1158-63. [PubMed]
- Rusby JE, Rainsbury RM. Distant volume flaps for conservative surgery. In: Rietjens M, Urban C. eds. Oncoplastic and reconstructive surgery. Italy: Springer-Verlag Italia, 2013:157-65.
- Hamdi M, Van Landuyt K, Hijjawi JB, et al. Surgical technique in pedicled thoracodorsal artery perforator flaps: a clinical experience with 99 patients. Plast Reconstr Surg 2008;121:1632-41. [PubMed]
- Hamdi M, Craggs B, Stoel AM, et al. Superior epigastric artery perforator flap: anatomy, clinical applications, and review of literature. J Reconstr Microsurg 2014;30:475-82. [PubMed]
- Zaha H, Onomura M, Nomura H, et al. Free omental flap for partial breast reconstruction after breast-conserving surgery. Plast Reconstr Surg 2012;129:583-7. [PubMed]


