Giant benign phyllodes tumor with lactating changes in pregnancy: a case report
Introduction
Phyllodes tumor of the breast, previously called cystosarcoma phyllodes, is a rare fibroepithelial neoplasm that accounts for less than 1% (0.3% to 0.5%) of all female breast neoplasms with few cases reported in pregnancy (1-3). Giant phyllodes tumors are those larger than 10 cm in diameter and account for about 20% of all phyllodes tumors (4). Phyllodes tumor typically exhibits an exclusive intracanalicular growth pattern of fibroepithelial cells with classical deep, leaf-like stromal projections into dilated lumens from which its name is derived (5). Base on the histologic features of presence or absence stromal overgrowth, degree of stromal cellularity and cellular atypia, mitotic activity per 10 high power fields (HPFs) and the nature of tumor borders, phyllodes tumor can generally be classified as benign, borderline and malignant (5-7). Heterogeneous stromal components are common in phyllodes tumor but is commonly associated with large or malignant phyllodes tumor (7-9).
Presentation during pregnancy is extremely rare and may grow rapidly and its size is relatively larger (10). In this case we present a pregnant patient who presented with a giant benign phyllodes tumor with the heterogeneity in stromal components as lactational changes from microscopic examination. It is occasionally difficult to distinguish between benign phyllodes tumors with lactating changes and lactating adenomas that are the most prevalent breast masses seen in pregnant women (11). We discuss and emphasize some of the issues in the diagnosis and complexities of management of phyllodes tumor with lactating changes.
Case presentation
A married Thai female, 36 years of age, gravida 1 para 0, at 32 weeks according to her last menstrual period, presented with a large left breast mass with rapidly growth for 5-6 months. The mass was associated with pain off and on. There was no history of trauma, weight loss or loss of appetite. She had no history of previous breast disease or family history of breast cancer.
Physical examination revealed a massively enlarged left breast with palpable firm mass measuring about 20 cm in maximum diameter occupying the whole breast (Figure 1). There was an area of cutaneous necrosis at upper outer quadrant region without axillary lymph node enlargement. The contralateral breast examination was unremarkable.
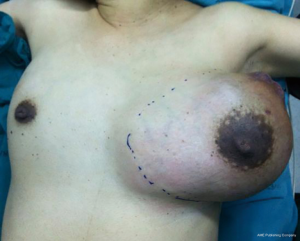
A mammogram was not performed because of the size of the lesion. Ultrasonographic examination showed large heterogenous well-defined margin mass with multiple lobulations and cystic spaces also presented.
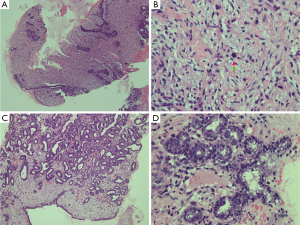
Ultrasound-guided core needle biopsy was performed at our breast surgery out-patient clinic. Microscopic examination revealed a fibroepithelial proliferative lesion. The epithelial part was benign and arranged in intracanalicular pattern. Stromal part showed hypercellularity without atypia or stromal overgrowth. Mitosis was 1-2 mitotic figures per 10 HPFs. In addition there were also some benign ducts that were lined by vacuolated secretory cells and contained secretions in the lumens (Figure 2).
The options for the treatment were discussed, at the multidisciplinary team tumor conference and eventually with the patient, regarding the operative procedure, timing for the removal of the tumor and the skin coverage problem. We concluded that this tumor was likely to be benign, so we would perform a definitive surgery after birth delivery. Due to the size of the lesion, mastectomy was the procedure of choice. As this pregnant patient had reduced abdominal subcutaneous tissue for a transverse rectus abdominis myocutaneous (TRAM) flap, we chose a latissimus dorsi myocutaneous (LD) flap for skin flap coverage. The patient and obstetrician decided that her baby should be delivered by elective caesarean section at the 37th gestational week so the operation for the tumor could be fastened.
Three weeks after birth delivery, she was scheduled for mastectomy. In addition to the mastectomy, our patient also underwent a level I axillary clearance because of multiple intraoperative enlarged lymph nodes. During the operation, the skin flaps could be approximately sutured without wound tension after undermining skin, so the planned LD flap was omitted. The plastic surgeon team planned to perform the delayed reconstruction later because the size of the contralateral breast in postpartum period or during lactation is bigger than normal. Gross examination of the resected specimen showed large well defined heterogeneous red tan and dark tan mass, measuring 13 cm × 11 cm × 10 cm. Cystic cavity containing milky substance about 300 mL was found in the medial part of the mass which caused the mass to be larger than its actual size on physical examination.
Microscopic examination revealed tumor mass with extensive hemorrhagic necrosis and small foci of viable tissue that consisted of predominant solid proliferative mammary lobules lined by cuboidal epithelium with vacuolated cytoplasm, outer thin layer of myoepithelial cells and contained eosinophilic secretions within the lumens. What mentioned above were the characteristics of lactating adenoma. However those findings were admixed with proliferation of benign ductal epithelium in intracanalicular pattern of leaf-like projections. Stromal cellularity was mild increased without atypia and mitosis was less than one per 10 HPFs. No stromal growth was identified (Figure 3). Combination of the previous core needle biopsy tissue and the recent mastectomy specimen favored phyllodes tumor with lactational changes as final diagnosis instead of lactating adenoma or the coexistence between two benign lesions (lactating adenoma and phyllodes tumor) in the same mass.
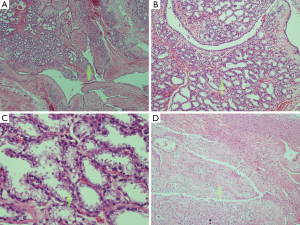
There was no evidence of invasion at borders and all resected margins were free of the tumor. Ten enlarged lymph nodes were removed with the specimen and all were negative for malignancy.
The patient made a good recovery and her wound was well healed. She did not receive any adjuvant therapy (postoperative radiotherapy or chemotherapy) because of the absence of the malignant features. She was planned to follow up by physical examinations and breast imaging studies twice per year for the first two years because chances of recurrence are maximum in the first two years, then on an annual basis. We also instructed her to do self-breast examination regularly.
Discussion
Phyllodes tumor is a rare fibroepithelial neoplasm but is extremely rare occuring during pregnancy (5,12,13). There are only nine cases reported to this date, all classified as malignant except one borderline lesion (14). To the best of our knowledge, we report here the first case of a benign phyllodes tumor with lactating changes. It is unknown if the rapidly growing mass in pregnant patient is hormone-dependent (15). The goal of the treatment of all phyllodes tumors is wide local excision to achieve at least a 1-cm or greater clear surgical margin (16,17). More than a half of previous reported cases presented in the third trimester as large masses, like our patient, required mastectomy to achieve adequate margins (5). Many prospective studies did not support the use of adjuvant therapy for patients with adequately resected disease (18). So the adjuvant treatment for this benign phyllodes tumor is not administered. However, some have considered adjuvant therapy when the primary tumor is greater than 5 cm, has stromal overgrowth, a high mitotic index, or infiltrative border (18).
In the future radiation therapy may be considered appropriate treatment for our patient if phyllodes tumor recurs following mastectomy (19). At present there is no evidence to support chemotherapy or endocrine therapy for phyllodes tumors (16,20).
This case emphasizes the heterogeneity of the phyllodes tumor that is commonly found in large or malignant phyllodes tumor (6,9,10). Not only necrosis and hemorrhage can occur, but also lactational changes too, especially in this large mass in postpartum period. Even though most parts of the tumor are typically look liked lactating adenoma. They admix with areas of hypercellular stroma with epithelial proliferation in tongue-like or leaf-like pattern. So we finally diagnosed and treated as a phyllodes tumor with lactating changes.
It is difficult to distinguish between benign phyllodes tumors with lactating changes and lactating adenomas (11). We need to differentiate these two diseases because the treatments for each are different. For lactating adenoma, although the tumor may spontaneously involute, surgical excision or enucleation may be necessary because of the mass effect it produces and this tumor does not tend to recur locally (21).
Conclusions
Phyllodes tumor can occur in pregnancy and is fast growing. With early detection a rare lesion like benign phyllodes tumor at smaller size then breast conserving surgery, recommends for benign lesion with adequate resection margin, is possible (5).
The final diagnosis can differ from the tissue biopsy diagnosis due to the heterogeneity of phyllodes tumors especially in the large or giant one (6,9,10). So it is important to thoroughly examine the resected specimen for possible additional stromal or epithelial components. Differentiation of phyllodes tumors from lactating adenomas, fibroadenomas and other benign breast lesions is pivotal to avoid inappropriate surgical treatment which may lead to local recurrence (22).
Although the classification for phyllodes tumor is subjective based on which criteria is used, the key point is that even in benign lesion with lactating changes, an adequate, clear surgical margin must be achieved to reduce local recurrence rate (23). The role of adjuvant therapy with radiation therapy and/or chemotherapy for benign phyllodes tumors has not been clearly defined (16,18,20).
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Rowell MD, Perry RR, Hsiu JG, et al. Phyllodes tumors. Am J Surg 1993;165:376-9. [PubMed]
- Donegan WL. Sarcoma of the breast. Major Probl Clin Surg 1979;5:504-42. [PubMed]
- Rosen PP. Fibroepithelial neoplasms. In: Weinberg RW, Donnellan K, Palumbo R, editors. Rosen’s Breast Pathology, 2nd ed. Philadelphia: Lippincott Williams & Wilkins, 2001:176-200.
- Liang MI, Ramaswamy B, Patterson CC, et al. Giant breast tumors: surgical management of phyllodes tumors, potential for reconstructive surgery and a review of literature. World J Surg Oncol 2008;6:117. [PubMed]
- Telli ML, Horst KC, Guardino AE, et al. Phyllodes tumors of the breast: natural history, diagnosis, and treatment. J Natl Compr Canc Netw 2007;5:324-30. [PubMed]
- Treves N, Sunderland DA. Cystosarcoma phyllodes of the breast: a malignant and a benign tumor; a clinicopathological study of seventy-seven cases. Cancer 1951;4:1286-332. [PubMed]
- Hart WR, Bauer RC, Oberman HA. Cystosarcoma phyllodes. A clinicopathologic study of twenty-six hypercellular periductal stromal tumors of the breast. Am J Clin Pathol 1978;70:211-6. [PubMed]
- Lester J, Stout AP. Cystosarcoma phyllodes. Cancer 1954;7:335-53. [PubMed]
- Treves N. A study of cystosarcoma phyllodes. Ann N Y Acad Sci 1964;114:922-36.
- Blaker KM, Sahoo S, Schweichler MR, et al. Malignant phylloides tumor in pregnancy. Am Surg 2010;76:302-5. [PubMed]
- Baker TP, Lenert JT, Parker J, et al. Lactating adenoma: a diagnosis of exclusion. Breast J 2001;7:354-7. [PubMed]
- Lenhard MS, Kahlert S, Himsl I, et al. Phyllodes tumour of the breast: clinical follow-up of 33 cases of this rare disease. Eur J Obstet Gynecol Reprod Biol 2008;138:217-21. [PubMed]
- Way JC, Culham BA. Phyllodes tumour in pregnancy: a case report. Can J Surg 1998;41:407-9. [PubMed]
- Andreola J, Damin A, Cruz J, et al. Giant borderline phyllodes breast tumor in pregnancy. J Senologic Int Society 2012;1:3.
- Aranda C, Sotelo M, Torres A, et al. Phyllodes tumor and pregnancy. A report of a case. Ginecol Obstet Mex 2005;73:387-92. [PubMed]
- Reinfuss M, Mituś J, Duda K, et al. The treatment and prognosis of patients with phyllodes tumor of the breast: an analysis of 170 cases. Cancer 1996;77:910-6. [PubMed]
- Mangi AA, Smith BL, Gadd MA, et al. Surgical management of phyllodes tumors. Arch Surg 1999;134:487-92; discussion 492-3. [PubMed]
- Chaney AW, Pollack A, McNeese MD, et al. Primary treatment of cystosarcoma phyllodes of the breast. Cancer 2000;89:1502-11. [PubMed]
- Pandey M, Mathew A, Kattoor J, et al. Malignant phyllodes tumor. Breast J 2001;7:411-6. [PubMed]
- Joshi NP, Mallick S, Pandit S, et al. Phyllodes tumor of the breast treated with a multimodality approach: a single-institution retrospective analysis. J Clin Oncol 2011;29:abstr e11552.
- Reeves ME, Tabuenca A. Lactating adenoma presenting as a giant breast mass. Surgery 2000;127:586-8. [PubMed]
- Nabi J, Akhter SM, Authoy FN. A case of large phyllodes tumor causing “rupture” of the breast: a unique presentation. Case Rep Oncol Med 2013;2013:871292.
- White DS, Irvine TE. Rapidly progressive multifocal phyllodes tumour of the breast: A case report and review of the literature. Int J Surg Case Rep 2013;4:901-3. [PubMed]


