Breast cancer in Wales: time trends and geographical distribution
Introduction
Breast cancer is the second commonest malignancy in the world, accounting for approximately 25% of all cancers (1). In 2012, approximately 522,000 women died of breast cancer across the world, with more than 60% of these deaths in less developed countries (1). In the United States of America, aside skin malignancies, breast cancer is the commonest cancer diagnosed among women, accounting for approximately 1 in 3 cancers (2). Breast cancer has been the commonest cancer in the United Kingdom since 1997 and it accounts for almost one-third of all new cases of cancer in women (3). There is a significant difference in the epidemiology of breast cancer across the world, with a higher incidence among European women compared to women in Africa, although African women tend to present at an earlier age and the disease appears to be more aggressive than in their European counterparts (4). Risk factors that are associated with breast cancer include use of hormone replacement therapy (HRT), nulliparity, late menopause, family history of breast cancer in a first-degree relative, personal history of benign breast disease and alcohol (5).
There is no study that has addressed the trends of breast cancer in Wales especially since the introduction of the screening programme in 1988. The aim of this study is therefore to provide an up-to-date analysis of time trends in incidence, geographical distribution, survival and mortality from breast cancer in Wales using data from the national cancer registry for Wales.
Materials and methods
The Welsh Cancer Intelligence and Surveillance Unit (WCISU) is the national cancer registry for Wales, which holds records of all cancers since 1972 with an electronic database of approximately 686,000 records. The registry covers the entire resident population of Wales of approximately three million people. The population of Wales is predominantly Caucasian ethnicity, with only 2.9% ethnic minorities. WCISU collects data from multiple sources, including inpatient activity through the Patient Episode Database for Wales (PEDW), pathology laboratory reports and death certificate registration. WCISU also obtains information from other United Kingdom cancer registries for Welsh residents treated outside Wales. Cases of breast cancer registered between 1985 and 2012 were identified from the database.
Data analysis
European age-adjusted standardised rates (EASR) were determined according to the standardised European population for the period 1985-2012. Incidence rates were calculated using mid-year population estimates. A Poisson regression model was fitted to assess temporal trends and rate ratios (RR) and 95% confidence intervals (CI) were determined and compared in relation to age, geographical distribution and mortality across the study period. Data were grouped by the 22 local authority areas in Wales. The data were analysed using IBM SPSS Statistics 20. The level of statistical significance at which null hypothesis was rejected was chosen as 0.05.
Results
Incidence
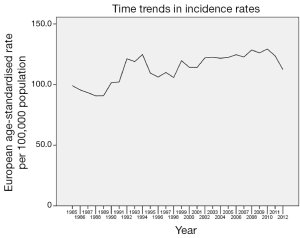
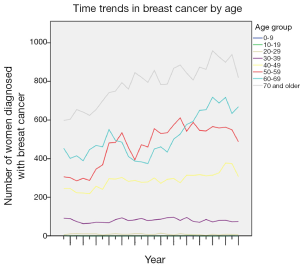
A total of 60,227 women diagnosed with breast malignancy were registered with the Welsh cancer registry between 1985 and 2012. The European age-standardised incidence rate of breast cancer was 113.4 per 100,000 populations over the entire study period. There has been a significant increase in the incidence of breast cancer over the study period with a higher rate in 2012 compared to 1985 (RR 1.389, 95% CI: 1.305-1.479, P<0.001). The first significant increase in incidence rate was recorded in 1991 and has remained significant annually till the end of the study period when compared to the initial study period. However, incidence appears be fluctuating between 105.8 and 129.3 per 100,000 populations from 1993 to 2012, rather than a linear increase in incidence. Figure 1 shows time trends in incidence rates. The number of women diagnosed with breast cancer remains fairly steady in the 20-29, 30-39 and 40-49 age groups throughout the study period. Among the 50-59 age group, a significant increase was recorded in 1991, but this has remained fairly stable since the late 1990s till the end of the study period. Following an increase in the number of women diagnosed with breast cancer in 1991, the 60-69 age group recorded a reduction in the number of breast cancer cases throughout the 1990s before a progressive increase since 2001. Women over the age of 70 years recorded their first significant increase in 1990 and unlike their younger counterparts, have not recorded any major decline in incidence throughout the study period. Figure 2 shows time trends in breast cancer by age.
Geographical distribution
On a broad geographical division of North and South Wales, there is no significant difference in the incidence of breast cancer (RR 0.767, 95% CI: 0.488-1.208, P=0.253). However, there appears to be a significant regional variation (local authority) in the incidence of breast cancer over the study period, with a higher incidence rate in the rural areas compared to urban areas (P<0.001) (Table 1). Overall, the highest rates are seen in Denbighshire, Gwynedd, Conwy, Monmouthshire and Ceredigion. The lowest rates are in Newport, Blaenau Gwent, Caerphilly and Merthyr Tydfil.
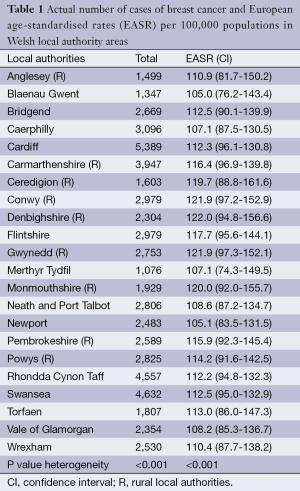
Full table
Survival
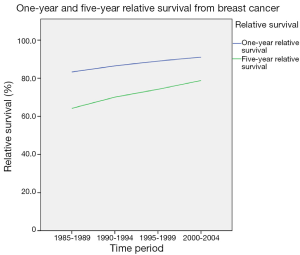
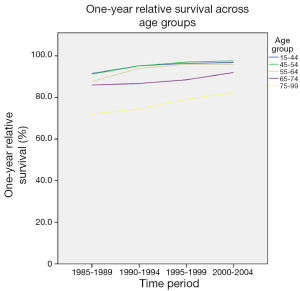
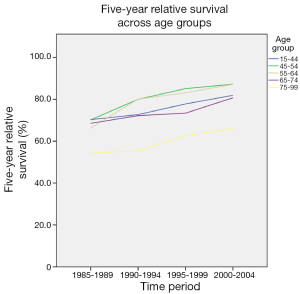
Survival was determined for 5-year diagnosis periods for all-cause and breast cancer-specific mortality. One- and five-year relative survival from all-cause mortality improved from 59.7 and 42.8 respectively in 1985-1989 to 67.3 and 52.1 respectively in 2000-2004. Overall, there has been a significant improvement in both 1- and 5-year relative survival over the study period (Figure 3). There has also been a considerable improvement in relative survival across all age groups (Figures 4,5). However, women over the age of 65 years continue to have lower 1-year relative survival (P<0.001) and 5-year relative survival (P=0.004) compared to their younger counterparts over the time period.
Mortality
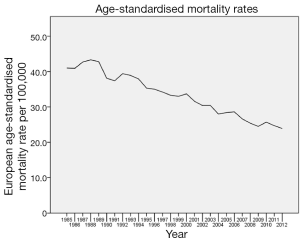
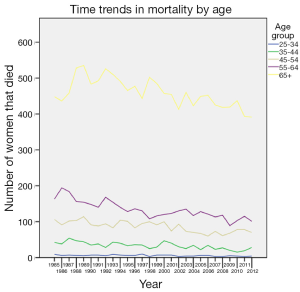
There was a steady decline in mortality from breast cancer over the entire study period (Figure 6). However, this was not statistically significant until around 2002 and it continued to be significant until the end of the study period in 2012. All age groups, except women over the age of 65 years, recorded a considerable decline in mortality from around 2000-2003. Figure 7 shows time trends in mortality according to age. The mortality rate among the 25-34 age group remained comparably low throughout the study period (P<0.001). The most dramatic decline was seen in the age groups 45-54 and 55-64 years (P<0.001). Women above the age of 65 years continued to fare worse compared to other age groups across the study period: 25-34 (RR 0.012, 95% CI: 0.011-0.014, P<0.001), 35-44 (RR 0.072, 95% CI: 0.068-0.077, P<0.001), 45-54 (RR 0.187, 95% CI: 0.179-0.196, P<0.001), 55-64 (RR 0.290, 95% CI: 0.280-0.301, P<0.001).
Discussion
This is the first study to highlight the trends of breast cancer in Wales over a period of almost three decades. The age-standardised incidence rate of breast cancer was 113.4 per 100,000 populations over the entire study period. This is higher than the incidence rate in China (6), Bahrain (7), Iran (8) and India (9), but significantly lower than the incidence rate in the United States (10), Canada (11) and Sweden (12).
Overall, there has been a significant increase in the incidence of breast cancer in Wales over the last 30 years. The first significant increase in incidence rate was recorded in 1991 and has remained significant annually till the end of the study period when compared to the initial study period. This coincides with the introduction of the National Health Service Breast Screening Programme in 1988 for women between the ages of 50 and 64 years every three years across the United Kingdom. Today, women aged 50-70 years are invited for screening every 3 years in Wales. While a significant increase was recorded following the introduction of mammography screening, incidence appears be fluctuating from 1993 till the end of the study period, rather than a progressive annual linear increase. The reason for this is unclear but it could be due to fluctuations in mammography rates. The incidence rate among women below the age of 50 years remained fairly steady over the study period. However, the incidence rate among women between the ages of 50 and 69 years was fluctuating over the study period since the first significant increase in incidence in the early 1990s following the introduction of the screening programme. This pattern may be because of changes to the breast screening programme during the study period. When the screening programme started in 1988, women between the ages of 50 and 64 years were invited, however this was extended to women aged 65-69 years in 2002 (13).
Many studies have reported a drastic reduction in the incidence of breast cancer as a result of decline in the use of HRT following the publication of the results of a major clinical trial that concluded that the long-term risks of HRT outweighed the health benefits for postmenopausal women in 2002 (11,14,15). However, this trend was not recorded in Wales. In fact, incidence rate increased significantly among older women in recent years despite a decline in the use of HRT in the UK. Although it may be argued that there may be a cohort effect reflecting previous exposure to HRT for older women, it is also well established that there is no increased risk in women beyond 5 years after stopping HRT (16).
There was no significant overall difference in the incidence of breast cancer between North and South Wales. However, incidence was higher in rural local authorities of Denbighshire, Gwynedd, Conwy, Monmouthshire and Ceredigion. Lower incidence rates were seen in more urban areas of Newport, Blaenau Gwent, Caerphilly and Merthyr Tydfil. This is contrary to most studies in the literature that have suggested that breast cancer is more prevalent in the urban areas than rural areas (17,18). Lower incidence of breast cancer was also seen in more deprived areas of Merthyr Tydfil, Blaenau Gwent and Caerphilly. It may be that uptake of screening is lower in urban and deprived areas of Wales possibly due to busy work life in urban areas and less education in deprived areas. There may therefore be a need to improve on awareness campaign in these areas.
Overall, there has been a significant improvement in both 1- and 5-year relative survival over the study period, which is similar to the trend in other European countries (19,20). It is highly likely that this is largely due to the Breast Screening Programme, resulting in early detection of breast malignancy, together with improved surgical intervention, advances in chemotherapy and more targeted radiation therapy. There has also been a considerable improvement in relative survival across all age groups. However, women over the age of 65 years continue to have lower 1- and 5-year relative survival rates compared to their younger counterparts over the time period.
There was a steady reduction in mortality from breast cancer over the entire study period. However, this was not statistically significant until around 2002 and it continued to be significant until the end of the study period in 2012. The decline in mortality rate is similar to the trend across other European countries (21). There was a considerable decline in mortality rate across almost all age groups, especially in recent years. However, women over the age of 65 years had poorer outcome throughout the study period.
This study has limitations. The findings rely on the quality of the coded data in the database. Detailed histopathology coding was not available, which could have resulted in better insights into the trends of breast cancer in Wales. Nevertheless, this study has been facilitated by the availability of a robust cancer registry in Wales that has systematically obtained records over the past four decades.
Conclusions
In conclusion, there has been a significant increase in the incidence of breast cancer in Wales over the last three decades, which is likely to be partly due to the introduction of the National Health Service Breast Screening Programme. While some studies in developed countries have reported a reduction in the incidence of breast cancer following a decline in the use of HRT, the incidence rate actually increased significantly among older women in recent years in Wales. Breast cancer incidence is higher in rural areas than urban areas and lower incidence was seen in more deprived areas. There has been a significant improvement in survival, although outcome remains poor for older women. Continued monitoring of the incidence of breast cancer will be crucial in fully understanding these trends.
Acknowledgements
The author thanks the Welsh Cancer Intelligence and Surveillance Unit (WCISU) for making the data available for the study.
Disclosure: The authors declare no conflict of interest.
References
- Ferlay J, Soerjomataram I, Dikshit R, et al. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer 2014. [Epub ahead of print]. [PubMed]
- DeSantis C, Ma J, Bryan L, et al. Breast cancer statistics, 2013. CA Cancer J Clin 2014;64:52-62. [PubMed]
- Cancer Research UK. Breast cancer incidence statistics. Available online: http://www.cancerresearchuk.org/cancer-info/cancerstats/types/breast/incidence/uk-breast-cancer-incidence-statistics. Accessed: 8th June 2014.
- Abdulrahman GO Jr, Rahman GA. Epidemiology of breast cancer in europe and Africa. J Cancer Epidemiol 2012;2012:915610.
- Schonfeld SJ, Pfeiffer RM, Lacey JV Jr, et al. Hormone-related risk factors and postmenopausal breast cancer among nulliparous versus parous women: An aggregated study. Am J Epidemiol 2011;173:509-17. [PubMed]
- Chen WQ, Zheng RS, Zeng HM, et al. Incidence and mortality of breast cancer in China, 2008. Thoracic Cancer 2013;4:59-65.
- Hamadeh RR, Abulfatih NM, Fekri MA, et al. Epidemiology of Breast Cancer among Bahraini Women: Data from the Bahrain Cancer Registry. Sultan Qaboos Univ Med J 2014;14:e176-82. [PubMed]
- Mohagheghi MA, Mosavi-Jarrahi A, Malekzadeh R, et al. Cancer incidence in Tehran metropolis: the first report from the Tehran Population-based Cancer Registry, 1998-2001. Arch Iran Med 2009;12:15-23. [PubMed]
- Manoharan N, Tyagi BB, Raina V. Cancer incidences in urban Delhi - 2001-05. Asian Pac J Cancer Prev 2009;10:799-806. [PubMed]
- Anderson WF, Katki HA, Rosenberg PS. Incidence of breast cancer in the United States: current and future trends. J Natl Cancer Inst 2011;103:1397-402. [PubMed]
- De P, Neutel CI, Olivotto I, et al. Breast cancer incidence and hormone replacement therapy in Canada. J Natl Cancer Inst 2010;102:1489-95. [PubMed]
- Möller T, Anderson H, Aareleid T, et al. Cancer prevalence in Northern Europe: the EUROPREVAL study. Ann Oncol 2003;14:946-57. [PubMed]
- Gompel A, Plu-Bureau G. Is the decrease in breast cancer incidence related to a decrease in postmenopausal hormone therapy? Ann N Y Acad Sci 2010;1205:268-76. [PubMed]
- Jemal A, Ward E, Thun MJ. Recent trends in breast cancer incidence rates by age and tumor characteristics among U.S. women. Breast Cancer Res 2007;9:R28. [PubMed]
- Glass AG, Lacey JV Jr, Carreon JD, et al. Breast cancer incidence, 1980-2006: combined roles of menopausal hormone therapy, screening mammography, and estrogen receptor status. J Natl Cancer Inst 2007;99:1152-61. [PubMed]
- Olsson H, Bladström A, Ingvar C, et al. A population-based cohort study of HRT use and breast cancer in southern Sweden. Br J Cancer 2001;85:674-7. [PubMed]
- Dey S, Soliman AS, Hablas A, et al. Urban-rural differences in breast cancer incidence in Egypt (1999-2006). Breast 2010;19:417-23. [PubMed]
- Zeng H, Zheng R, Zhang S, et al. Incidence and mortality of female breast cancer in China, 2009. Thoracic Cancer 2013;4:400-4.
- Macià F, Porta M, Murta-Nascimento C, et al. Factors affecting 5- and 10-year survival of women with breast cancer: an analysis based on a public general hospital in Barcelona. Cancer Epidemiol 2012;36:554-9. [PubMed]
- Tryggvadóttir L, Gislum M, Bray F, et al. Trends in the survival of patients diagnosed with breast cancer in the Nordic countries 1964-2003 followed up to the end of 2006. Acta Oncol 2010;49:624-31. [PubMed]
- Autier P, Boniol M, Gavin A, et al. Breast cancer mortality in neighbouring European countries with different levels of screening but similar access to treatment: trend analysis of WHO mortality database. BMJ 2011;343:d4411. [PubMed]