
Factors associated with disease upgrading in patients with papillary breast lesion in core-needle biopsy
Introduction
Breast papillary lesion is defined as a proliferative lesion characterized architecturally by the presence of finger-like projections composed of a stromal core overlain by a layer of epithelial cells (1). Breast papillary lesions are known to have various clinical presentations and a broad pathological spectrum (2). Papillary lesions may present with nipple discharge or as a palpable mass, or as an asymptomatic lesion detected in imaging study. The incidence of papillary lesion in core-needle biopsy (CNB) specimens of the breast is up to 6% according to previous reports (3-5).
Traditionally, breast papillary lesions are regarded as lesions that require surgical excision because of their malignant potency. The reported upgrading rates from benign papillary lesions diagnosed using CNB to malignancy diagnosed after a subsequent surgical excision ranged from 0% to 29% (6). However, whether all papillary lesions require surgical excision for appropriate management is controversial.
The aims of this study were to identify factors associated with upgrading to malignancy in patients with benign papillary lesions in CNB, and to avoid unnecessary overtreatment.
We present the following article in accordance with the STROBE reporting checklist (available at http://dx.doi.org/10.21037/gs-20-310).
Methods
Medical records of patients were reviewed retrospectively who were diagnosed as having benign papillary lesions in CNB and underwent a subsequent surgical excision between January 2007 and December 2016 at Inha University Hospital and Cheil General Hospital & Women’s Healthcare Center. Surgical excision was recommended to all the patients with benign papillary lesion in CNB. One hundred and seventy-nine patients underwent a subsequent surgical excision among who were diagnosed as having benign papillary lesions in CNB and these 179 patients were included in this study. Patients who had concurrent ductal carcinoma in situ (DCIS) or invasive carcinoma in the ipsilateral breast were excluded.
CNB was performed using the 14-gauge needle gun method with ultrasonography guidance. At least four or five core biopsies were done by lesions. Pathologic evaluation was performed by 3 board-certified pathologists who had received breast subspecialty training. Hematoxylin and eosin-stained slides and if needed, additional immunohistochemical stained slides for cytokeratin 5/6, myoepithelial cell markers, and/or estrogen receptor were evaluated.
The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by the institutional review boards of Inha University Hospital (approval number: INHAUH 2017-10-016) and waived the requirement for informed consent.
The following data were analyzed: age, menopause, symptoms (palpability and nipple discharge), sonographic findings (size, multifocality, and location), pathological result from CNB, and final pathological result after surgical excision.
Upgrading was defined as the identification of DCIS or invasive carcinoma after surgical excision. On ultrasonography, a lesion located within 2 cm from the nipple was defined as a central location, and any location further away from the nipple was defined as a peripheral location.
Statistical analyses were performed using IBM SPSS Statistics Version 19.0 for Windows (IBM Corp., Armonk, NY, USA). To identify the factors associated with upgrading, multiple regression analysis was performed. A P value of <0.05 was considered statistically significant. The results were presented as odds ratios (ORs) with their 95% confidence intervals (CIs).
Results
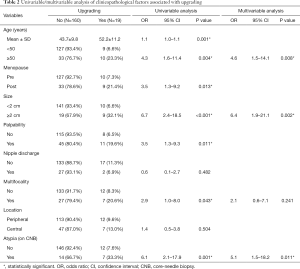
The mean age of the 179 patients was 44.6 years (range, 14–77 years). Among the 179 patients, 43 (24.0%) were aged ≥50 years and 42 (23.5%) were postmenopausal women. Of the patients, 56 (31.3%) presented with a palpable mass, 29 (16.2%) presented with nipple discharge, 28 (15.6%) had a lesion of ≥2 cm on ultrasonography, 34 (19.0%) had multifocal lesions, 54 (30.2%) had a centrally located lesion on ultrasonography, and 21 patients (11.7%) had an atypical papillary lesion in CNB (Table 1).

Full table
In the 179 patients, the upgrade rate to malignancy was 10.6% (7.6% in papillary lesions without atypia vs. 33.3% in papillary lesions with atypia; P=0.001). Concordance rate for atypia between CNB and surgical specimen was 14.3%. The univariable analysis revealed that age at diagnosis (<50 vs. ≥50 years; P=0.004), menopause (P=0.013), lesion size on ultrasonography (<2 vs. ≥2 cm; P<0.001), palpability (P=0.011), multifocality (P=0.043), and atypia in CNB (P=0.001) were associated with upgrading. The number of variables that had a P value of <0.1 in the univariable analysis was six and these were many for multiple regression analysis. Considering the characteristics of the variables, we excluded menopause and palpability from the variables for multiple regression analysis, because there is intrinsic association between menopause and age, and so between palpability and lesion size. The multivariable analysis revealed that age at diagnosis ≥50 years (OR, 4.6; 95% CI, 1.5–14.1; P=0.008), lesion size of ≥2 cm (OR, 6.4; 95% CI, 1.9–21.1; P=0.002), and atypia in CNB (OR, 5.1; 95% CI, 1.5–18.2; P=0.011) were significantly associated with upgrading to malignancy (Table 2).
Full table
Discussion
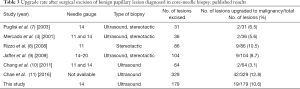
The previously reported upgrading rates from benign papillary lesions diagnosed using CNB to malignancy diagnosed after a subsequent surgical excision ranged from 0% to 29% (6). The Upgrade rate in our study was 10.6%. Table 3 shows the results of several previously published studies. These upgrading rates for papillary lesions were high enough to deserve attention. Thus, the proper management of benign papillary lesions is controversial.
Full table
In our study, age ≥50 years, lesion size of ≥2 cm, and atypia in CNB were significantly associated with upgrading to malignancy. The upgrade rate of atypical papillary lesions in this study was significantly high at 33.3%. In many previous studies, the upgrade rates of benign papillary lesions with atypia were also significantly high, so the need for surgical excision of atypical papillary lesions in CNB was generally accepted (12). However, proper management of benign papillary lesions without atypia diagnosed in CNB is still controversial.
Some authors believe that benign papillary lesions without atypia can be managed safely with clinical and radiological follow-up if the radiological and pathological findings are concordant. However, others recommend excision for these patients to rule out any hidden malignancy. The potential heterogeneity in the distribution of atypia or carcinoma, if present, within a papillary lesion can disrupt accurate diagnosis (6).
In this study, 106 patients were under the age of 50 and had papillary lesions of <2 cm in size without atypia. Among these patients, 5 (4.7%) were upgraded to malignancy. As the value of 4.7% can be acceptable, non-atypia papillary lesions <2 cm in women under the age of 50 can be managed with clinical and radiological follow-up. However, close follow-up is necessary to avoid overlooking 4.7% of cases.
Several previous studies tried to identify the factors associated with the upgrade of papillary breast lesions without atypia (9,10,13-18). These studies reported that palpable lesions (14,15), larger lesion size (cutoff, 1–1.5 cm) (10,16,17), older age (cutoff, 50–54 years), peripheral lesion, higher Breast Imaging Reporting and Data System score, and pathological-radiological discordance (17) were associated with upgrading to malignancy.
Some authors thought the upgrading was associated with the sampling method and reported that vacuum-assisted breast biopsy was a more efficient method than the usual CNB (18-20). Vacuum assisted excision can be a good option for management of papillary lesion without atypia in CNB and can produce better cosmetic outcome (21). However, no long-term prospective studies have been conducted on benign papillary lesions diagnosed using CNB. Thus, prospective long-term studies on benign papillary lesions diagnosed using CNB in a larger number of patients are mandated. This study was a retrospective cross-sectional study with a relatively small number of patients, which is a source of its inherent limitations. Breast imaging and pathologic result were reported according to Breast Imaging Reporting and Data System and World Health Organization Classification of Tumours of the Breast. However there could be interobserver variability, which is also the limitation of this study.
In conclusion, upgrading to malignancy in patients with benign papillary lesions in CNB was associated with age ≥50 years, lesion size ≥2 cm, and atypia. Benign papillary lesions diagnosed using CNB in women under the age of 50, which are small lesions without atypia, can be observed but require close observation with clinical and radiological follow-up.
Acknowledgments
Funding: This work was supported by INHA University Research Grant (INHA-57860).
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at http://dx.doi.org/10.21037/gs-20-310
Data Sharing Statement: Available at http://dx.doi.org/10.21037/gs-20-310
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/gs-20-310). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by the institutional review boards of Inha University Hospital (approval number: INHAUH 2017-10-016) and waived the requirement for informed consent.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Agoumi M, Giambattista J, Hayes MM. Practical Considerations in Breast Papillary Lesions: A Review of the Literature. Arch Pathol Lab Med 2016;140:770-90. [Crossref] [PubMed]
- Nakhlis F, Ahmadiyeh N, Lester S, et al. Papilloma on core biopsy: excision vs. observation. Ann Surg Oncol 2015;22:1479-82. [Crossref] [PubMed]
- Liberman L, Bracero N, Vuolo MA, et al. Percutaneous large-core biopsy of papillary breast lesions. AJR Am J Roentgenol 1999;172:331-7. [Crossref] [PubMed]
- Mercado CL, Hamele-Bena D, Singer C, et al. Papillary lesions of the breast: evaluation with stereotactic directional vacuum-assisted biopsy. Radiology 2001;221:650-5. [Crossref] [PubMed]
- Kim MJ, Kim SI, Youk JH, et al. The diagnosis of non-malignant papillary lesions of the breast: comparison of ultrasound-guided automated gun biopsy and vacuum-assisted removal. Clin Radiol 2011;66:530-5. [Crossref] [PubMed]
- Nayak A, Carkaci S, Gilcrease MZ, et al. Benign papillomas without atypia diagnosed on core needle biopsy: experience from a single institution and proposed criteria for excision. Clin Breast Cancer 2013;13:439-49. [Crossref] [PubMed]
- Puglisi F, Zuiani C, Bazzocchi M, et al. Role of mammography, ultrasound and large core biopsy in the diagnostic evaluation of papillary breast lesions. Oncology 2003;65:311-5. [Crossref] [PubMed]
- Rizzo M, Lund MJ, Oprea G, et al. Surgical follow-up and clinical presentation of 142 breast papillary lesions diagnosed by ultrasound-guided core-needle biopsy. Ann Surg Oncol 2008;15:1040-7. [Crossref] [PubMed]
- Jaffer S, Nagi C, Bleiweiss IJ. Excision is indicated for intraductal papilloma of the breast diagnosed on core needle biopsy. Cancer 2009;115:2837-43. [Crossref] [PubMed]
- Chang JM, Moon WK, Cho N, et al. Management of ultrasonographically detected benign papillomas of the breast at core needle biopsy. AJR Am J Roentgenol 2011;196:723-9. [Crossref] [PubMed]
- Hong YR, Song BJ, Jung SS, et al. Predictive Factors for Upgrading Patients with Benign Breast Papillary Lesions Using a Core Needle Biopsy. J Breast Cancer 2016;19:410-6. [Crossref] [PubMed]
- Richter-Ehrenstein C, Tombokan F, Fallenberg EM, et al. Intraductal papillomas of the breast: diagnosis and management of 151 patients. Breast 2011;20:501-4. [Crossref] [PubMed]
- Liberman L, Tornos C, Huzjan R, et al. Is surgical excision warranted after benign, concordant diagnosis of papilloma at percutaneous breast biopsy? AJR Am J Roentgenol 2006;186:1328-34. [Crossref] [PubMed]
- Jung SY, Kang HS, Kwon Y, et al. Risk factors for malignancy in benign papillomas of the breast on core needle biopsy. World J Surg 2010;34:261-5. [Crossref] [PubMed]
- Shouhed D, Amersi FF, Spurrier R, et al. Intraductal papillary lesions of the breast: clinical and pathological correlation. Am Surg 2012;78:1161-5. [Crossref] [PubMed]
- Chang JM, Moon WK, Cho N, et al. Risk of carcinoma after subsequent excision of benign papilloma initially diagnosed with an ultrasound (US)-guided 14-gauge core needle biopsy: a prospective observational study. Eur Radiol 2010;20:1093-100. [Crossref] [PubMed]
- Youk JH, Kim EK, Kwak JY, et al. Benign papilloma without atypia diagnosed at US-guided 14-gauge core-needle biopsy: clinical and US features predictive of upgrade to malignancy. Radiology 2011;258:81-8. [Crossref] [PubMed]
- Holley SO, Appleton CM, Farria DM, et al. Pathologic outcomes of nonmalignant papillary breast lesions diagnosed at imaging-guided core needle biopsy. Radiology 2012;265:379-84. [Crossref] [PubMed]
- Maxwell AJ. Ultrasound-guided vacuum-assisted excision of breast papillomas: review of 6-years experience. Clin Radiol 2009;64:801-6. [Crossref] [PubMed]
- Kibil W, Hodorowicz-Zaniewska D, Popiela TJ, et al. Vacuum-assisted core biopsy in diagnosis and treatment of intraductal papillomas. Clin Breast Cancer 2013;13:129-32. [Crossref] [PubMed]
- Choi HY, Kim SM, Jang M, et al. Benign Breast Papilloma without Atypia: Outcomes of Surgical Excision versus US-guided Directional Vacuum-assisted Removal or US Follow-up. Radiology 2019;293:72-80. [Crossref] [PubMed]


