Neck dissection with cervical sensory preservation in thyroid cancer
Introduction
Neck dissections play an essential role in the management of head and neck cancer. Their role in the management of thyroid cancer is somewhat controversial and limited by the absence of prospective clinical trials. The dilemma is further complicated by the indolent history of most well-differentiated thyroid cancers and the common occurrence of both clinically significant and occult cervical lymph node metastases particularly in young patients with papillary carcinoma of the thyroid. A strong indication for modified radical neck dissection (MND) in the setting of well-differentiated thyroid cancer is the finding of cervical lymph-adenopathy by either palpation or an imaging study. Confirmation of metastatic disease can be obtained by a preoperative fine-needle aspiration (FNA) which can be performed under ultrasound guidance. Sentinel lymph node biopsy in the jugulo-carotid chain using methylene blue dye mapping may be a feasible and valuable method for estimating lymph node status in the lateral neck compartment. It may be helpful in the detection of true-positive but non-palpable lymph nodes, and in such cases may support the decision to perform MND in patients with differentiated thyroid carcinoma (1). Prophylactic MND is recommended for cases of papillary carcinoma demonstrating two or more of the following four characteristics; male gender, age 55 years or older, maximal tumor diameter larger than 3 cm, and massive extrathyroid extension (2).
Cervical lymphadenopathy is not uncommon in the setting of well-differentiated thyroid cancer. A recent series reported the frequent occurrence of metastases in both the central (64.1%) and lateral (44.5%) neck (3). In the past many surgeons advocated local “berry picking” resections designed to remove grossly enlarged nodes (4,5). These “berry picking” procedures are associated with a higher local recurrence rate necessitating remedial surgery that is associated with a higher complication rate (6). The complication rate associated with functional neck dissections is no higher than that associated with “berry picking” procedures (6). It is noteworthy that limited modified neck dissections in which the superior extent of surgery is limited to the spinal accessory nerve are also associated with residual and recurrent disease due to skip metastases (7). A carefully performed MND is well-tolerated and results in excellent cosmetic, functional, and oncologic outcomes (8,9).
The technique of radical neck dissection was described by George Crile in 1906 (10). This extirpative procedure was often used in the setting of metastatic head and neck cancers, often of squamous cell origin, with metastases to the cervical lymph nodes. The operation encompassed removal of the cervical nodes and sacrifice of the internal jugular vein, spinal accessory and greater auricular nerves, as well as the sternocleidomastoid, digastric, and stylohyoid muscles. This radical neck dissection was modified to encompass an oncologically equivalent cervical lymphadenectomy while preserving functional structures including the sternocleidomastoid muscle, internal jugular vein, spinal accessory and greater auricular nerves, as well as the digastric and stylohyoid muscles. Accordingly, a MND is also referred to as a functional or Bocca neck dissection (11).
Patients with thyroid cancer are usually treated with a total or near-total thyroidectomy followed by radioactive iodine therapy and life-long thyroid hormone suppression. A subset of patients either present with metastatic nodal disease or develop metachronous nodal disease later in their course. The most frequent sites of metastases are the central cervical nodes (level VI) bounded by the hyoid bone superiorly, the innominate vein inferiorly, and bilaterally by the carotid sheath (12). Small lymph nodes are frequently encountered in this central region during initial thyroidectomy and should be resected when they are suspicious for metastatic disease as evidenced by enlargement, firmness, or irregularity by palpation.
A MND refers to resection of the lymph nodes in levels II through V and often including the central nodes in level VI. This review describes the operation in detail. It can be performed as an isolated procedure or in combination with a total thyroidectomy. It can also be performed bilaterally. It is often performed for well-differentiated thyroid cancers, most commonly papillary carcinoma of the thyroid. Occasionally patients with follicular or Hürthle cell carcinoma also require modified neck dissections. Patients with medullary carcinoma of the thyroid present a unique set of challenges and, due to the absence of effective adjuvant therapy, surgery plays an even more dominant role in their management. Furthermore, medullary cancer of the thyroid has an early predilection for both central and lateral nodal dissemination. Screening for MTC and primary treatment with total thyroidectomy and MND are essential for biochemical cure of MTC (13). The detailed management plan for medullary carcinoma of the thyroid is beyond the scope of this review.
Operative technique
The patient is placed on the operating table with the head extended and the neck and anterior chest are prepared for surgery (Figure 1). An inflatable thyroid pillow is placed behind the patient’s back. A linear incision is made that extends from the mid-neck as a continuation of a Kocher incision extending superiorly to approximately 1 inch below the left earlobe. If a bilateral neck dissection is required this incision is extended bilaterally and an apron flap is raised superiorly. Because the spinal accessory nerve is superficial it is important for the surgeon to note the surface landmarks depicting the course of the accessory nerve. If one were to connect a line between the angle of the mandible and the mastoid process and transect this line at a right angle in its mid-portion, the inferior course of that line would be a close approximation of the course of the spinal accessory nerve. It is well worth drawing this on the patient’s skin at the time of surgery. The marginal mandibular nerve is protected superiorly but is rarely seen in this exposure. Once the incision is made skin hooks are utilized to develop the anterior flap which is raised with an electrocautery (14).
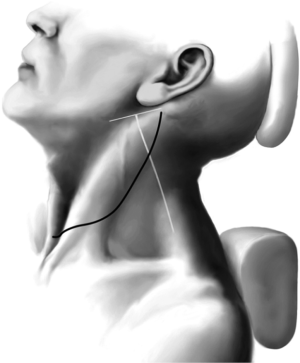
In Figure 2 the anterior and posterior flaps are completed and the underlying anatomy is demonstrated. The anterior flap is created in the subplatysmal layer and developed as the skin is pulled toward the medial neck. Once the anterior flap has been developed, the posterior flap is developed. The sternocleidomastoid muscle, external jugular vein, and greater auricular nerve are shown. The greater auricular nerve anatomy is extremely important serving as a landmark as the nerve emerges from the lateral aspect of the sternocleidomastoid muscle at Erb’s point. The nerve then traverses upward over the muscle going toward the earlobe. Preservation of this nerve is readily accomplished thereby preserving sensation to the earlobe. As the posterior flap is developed, great care and attention must be focused on protecting the spinal accessory nerve which is superior to the greater auricular nerve. The trapezius muscle is identified posterolaterally and the accessory nerve will course from behind the sternocleidomastoid muscle eventually innervating the trapezius muscle. A nerve stimulator is quite useful in locating the accessory nerve. Once the flaps have been completed, a self-retaining retractor is used to hold the skin and platysma in place. Frequently the skin is sewn to the retractor for added exposure. At this point, the external jugular vein is identified superiorly and ligated to form the apex of the tissues that will be unwrapped from the sternocleidomastoid muscle (14).

The fascial sheath covering the sternocleidomastoid muscle is shown being unwrapped in Figure 3. The flap, once developed, is mobilized from superior to inferior (Figure 4). Once this has been performed (Figure 4) a Penrose drain is placed around the sternocleidomastoid muscle (note there are two heads, a sternal head and a clavicular head) and the muscle is pulled medially. If the patient is particularly muscular, it is useful to place separate drains around each head facilitating enhanced exposure. The omohyoid muscle is identified and deep to it the carotid sheath is located. A thin Penrose drain is placed around the omohyoid and in the majority of cases this muscle is preserved. The three structures of the sheath, the common carotid artery, vagus nerve, and internal jugular vein, are carefully protected. Frequently vessel loops are placed around these structures to assist with mobilization. One must be very careful not to injure the sympathetic trunk that lies deep to the common carotid artery as injury to this structure will result in Horner’s syndrome. Occasionally, it is necessary to sacrifice the internal jugular vein due to tumor invasion; however, this is unusual in the setting of well-differentiated thyroid cancer. Maintaining the integrity of the anterior jugular veins is important as they become an effective collateral drainage system when the internal jugular vein is sacrificed (14).
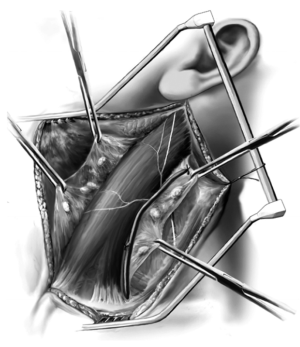
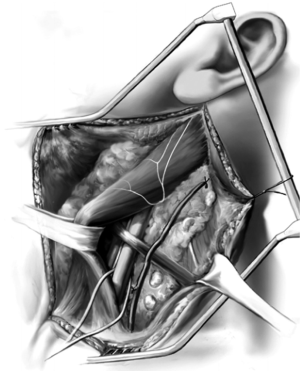
At this point the cervical fat pad extending below the clavicle is mobilized from inferior to superior. The carotid sheath structures and particularly the thoracic duct are protected. An unrecognized injury to the thoracic duct can cause a troublesome postoperative lymphatic leak which often leads to a lymphocele and infection. It is prudent to tie the lymphatic tissues inferiorly to minimize lymphatic leaks.
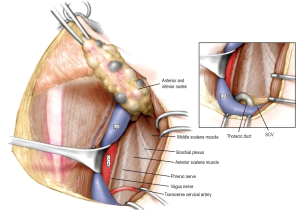
The next step is to identify the phrenic nerve which lies lateral to the vagus nerve (Figure 5). This is an important landmark to identify as it innervates the diaphragm. It also represents the deep margin of the specimen lying superficial to the anterior scalene muscle. All superficial fat and lymph node-bearing tissues are resected anterior to the phrenic nerve as this block of tissue is swept from inferior to superior. The specimen is then passed beneath the omohyoid muscle as the dissection continues as shown and the specimen is pulled superiorly. The omohyoid muscle and phrenic nerve are shown, the brachial plexus is identified laterally, and superiorly the accessory nerve is seen. Branches of the transverse cervical nerves are usually sacrificed. However, it is not difficult to preserve one or more of these branches and thereby maintain sensation to the skin of the ipsilateral shoulder. The soft tissues underneath the accessory nerve are mobilized and included with the specimen.
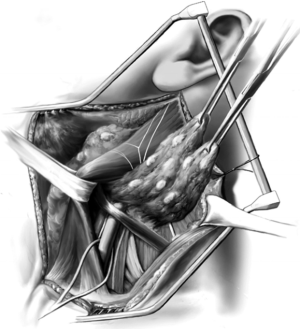
Figure 6 shows the specimen has now been mobilized off the sternocleidomastoid muscle and has been passed from the lateral aspect of the muscle underneath the muscle belly anteriorly. At this point, a thyroid retractor is placed underneath the mandible and the mandible is pulled superiorly. The parotid and submandibular glands are preserved and the digastric muscle is identified. Inferior to the digastric muscle is the hypoglossal nerve which must be identified. A useful trick to help locate the hypoglossal nerve is to follow the branch of the ansa cervicalis nerve as it courses superiorly along the anterior surface of the carotid artery until it joins the hypoglossal nerve at a right angle. Stimulation of the hypoglossal nerve will result in a movement of the tongue. In addition, the proximal accessory nerve must be identified at the medial aspect of the sternocleidomastoid muscle as soft tissues along the accessory nerve are included with the specimen. The nerve can be felt like a violin string if one distracts the sternocleidomastoid muscle posteriorly. It is extremely important to protect this nerve. All tissues inferior to the digastric muscle and hypoglossal nerve are resected in continuity with the specimen as the final tissues are removed from the great vessels and vagus nerve (14).
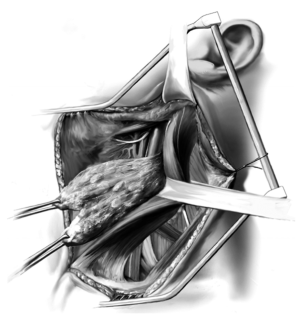
At the completion of the operation, a Penrose drain has been placed around the sternocleidomastoid muscle and the omohyoid muscle; the common carotid artery, vagus nerve, internal jugular vein, phrenic nerve, brachial plexus, accessory nerve, and greater auricular nerve are all preserved. Closure is obtained by reapproximating the platysma muscle with interrupted sutures and a subcuticular closure of the skin. A drain is always placed due to the extensive lymphadenectomy (14).
The operation is well-tolerated and the vast majority of patients are extubated in the operating room.
Since the introduction of endoscopic techniques in thyroid surgery, several trials of endoscopic lateral neck dissection have been conducted with the aim of avoiding a long cervical scar, but these endoscopic procedures require more effort than open surgery, mainly because of the relatively non-sophisticated instruments used. However, the recent introduction of surgical robotic systems has simplified the operations and increased the precision of endoscopic techniques. Some people have described their initial experience with robot-assisted MND in thyroid cancer using the da Vinci S system. Robot-assisted MND is technically feasible, safe, and produces excellent cosmetic results. Based on their initial experience, robot-assisted MND should be viewed as an acceptable alternative method in patients with low-risk, well-differentiated thyroid cancer with lateral neck node metastasis (15).
Cervical sensory preservation
Cervical plexus consist of four cervical spinal nerves, namely form occipital nerve, great auricular nerve, neck cutaneous nerve, supraclavicular nerve and phrenic nerve. Sensory branches of the cervical roots provide sensation to skin of the neck and shoulder. As the skin flaps are elevated in subplatysmal plane, several branches of the cervical plexus are immediately encountered overlying the SCM. Sacrifice of these branches will results in a sensory deficit that extends from pinna to the chest wall below the clavicle (16). In another study, Saffold et al. found that preservation of the cervical root branches resulted in a small, limited, and uniform area of the neck rendered permanently anesthetic. Conversely, sacrifice of the nerve branches led to a pattern of anesthesia involving the entire neck (17). Most of this sensory deficit will spontaneously resolve postoperatively over a period of months. Branches of the cervical plexus can also form neuromas, which present as firm, painful masses in the lateral neck that are exquisitely tender to palpation (18).
When performing modified neck dissection, we can find the superficial cutaneous branches of the cervical plexus at the superficial fascia midpoint of the trailing edge of the sternocleidomastoid muscle: (I) auricular nerve along the sternocleidomastoid surface upward, dominate the ear and the parotid gland skin feel; (II) the transverse cervical nerve along the front line of muscle to the anterior surface area; (III) occipital nerve piercing point is higher along the posterior border of sternocleidomastoid backward and upward to occiput; (IV) supraclavicular nerve run out below the midpoint of the trailing edge of the muscle slightly, depending on the starting position is deeper, so we can look for the chest and shoulder branch sections in the clavicle 2/3 above the superficial fascia, then up to track trunk.
During neck dissection including level V, the cervical sensory branches were identified and preserved after careful separation from fibrofatty tissue of the posterior cervical triangle. The nerves were isolated and traced into the SCM, and cervical nerve rootlets were found after the posterior border of the SCM was incised and elevated (19).
The cervical nerve rootlets were identified and preserved after mobilizing the anterior aspect of the SCM along its full extent. Dissection was carried underneath this muscle so that it could be fully mobilized and elevated as a bipedicle flap. The dissection was carried forward along the deep cervical fascial plane. The other cervical root branches were preserved after careful separation from fibrofatty tissue of the neck if the tumors or lymph nodes had not directly invaded the nerves. During selective neck dissection excluding level V, the cervical root branches were usually accessed after mobilizing the anterior aspect of the SCM, reflecting off the muscles, and dissecting back along its medial surface. The SCM was then reflected off the underlying soft tissues, dissecting back along its medial surface until the cervical rootlets were identified wrapping around its posterior border. The cervical root branches were preserved after deep plane dissection and careful separation from fibrofatty tissue of the bloc neck (19) (Figure 7).
Because the neck skin are widely separated, if cutaneous sensory nerves are damaged, postoperative patients’ skin often feel wood swelling, sluggish or formication, paresthesia etc. This feeling usually began to recover in a few weeks after surgery, disappear in three months. The surgeon should explain to the patient before surgery that this feeling does not need special treatment. Before the return to normal sensation, facial skin should be protected because it feels relatively slow and is vulnerable to trauma. At the time, the connection between separated skin and basal blood is not perfect, it is easy to become injured scarring.
Sensory nerve trunk injury can cause permanent feeling disappear in its disposal zone. And it can not be rebuilt. Therefore, surgery should be taken to avoid damage sensory nerve trunk, as in the separation of the ear flap, pay attention to not cut auricular nerve. Once damaged, nerve anastomosis or transplantation should be considered to be repaired.
The practice of neck dissection has greatly advanced from radical to function-preserving surgery (20). To avoid postoperative neck and shoulder morbidity, current techniques of neck dissection tend to be more selective (21,22). When performing modified neck dissection, we recommend to protect more reserved cervical plexus. The purpose is to better protect patients’ neck skin feeling.
Acknowledgements
Disclosure: The authors declare no conflict of interest.
References
- Dzodic R, Markovic I, Inic M, et al. Sentinel lymph node biopsy may be used to support the decision to perform modified radical neck dissection in differentiated thyroid carcinoma. World J Surg 2006;30:841-6. [PubMed]
- Ito Y, Higashiyama T, Takamura Y, et al. Risk factors for recurrence to the lymph node in papillary thyroid carcinoma patients without preoperatively detectable lateral node metastasis: validity of prophylactic modified radical neck dissection. World J Surg 2007;31:2085-91. [PubMed]
- Wada N, Duh QY, Sugino K, et al. Lymph node metastasis from 259 papillary thyroid microcarcinomas: frequency, pattern of occurrence and recurrence, and optimal strategy for neck dissection. Ann Surg 2003;237:399-407. [PubMed]
- Raina S, Rocko JM, Swaminathan AP, et al. Current attitudes in the management of thyroid cancer. Am Surg 1983;49:110-2. [PubMed]
- Nicolosi A, Malloci A, Esu S, et al. The role of node-picking lymphadenectomy in the treatment of differentiated carcinoma of the thyroid. Minerva Chir 1993;48:459-63. [PubMed]
- Musacchio MJ, Kim AW, Vijungco JD,et al. Greater local recurrence occurs with “berry picking” than neck dissection in thyroid cancer. Am Surg 2003;69:191-6; discussion 196-7. [PubMed]
- Pingpank JF Jr, Sasson AR, Hanlon AL, et al. Tumor above the spinal accessory nerve in papillary thyroid cancer that involves lateral neck nodes: a common occurrence. Arch Otolaryngol Head Neck Surg 2002;128:1275-8. [PubMed]
- Gemsenjäger E, Perren A, Seifert B, et al. Lymph node surgery in papillary thyroid carcinoma. J Am Coll Surg 2003;197:182-90. [PubMed]
- Kupferman ME, Patterson DM, Mandel SJ, et al. Safety of modified radical neck dissection for differentiated thyroid carcinoma. Laryngoscope 2004;114:403-6. [PubMed]
- Crile G.. Landmark article Dec 1, 1906: excision of cancer of the head and neck. With special reference to the plan of dissection based on one hundred and thirty-two operations. By George Crile. JAMA 1987;258:3286-93. [PubMed]
- Bocca E, Pignataro O, Sasaki CT. Functional neck dissection. A description of operative technique. Arch Otolaryngol 1980;106:524-7. [PubMed]
- Greene FL, Page DL, Fleming ID, et al. eds. AJCC cancer staging manual, 6th edition. New York: Springer, 2002.
- Weber T, Schilling T, Frank-Raue K, et al. Impact of modified radical neck dissection on biochemical cure in medullary thyroid carcinomas. Surgery 2001;130:1044-9. [PubMed]
- Daniel O, Robert U. eds. Surgery of the thyroid and parathyroid glands. Berlin: Springer-Verlag, 2007.
- Kang SW, Lee SH, Ryu HR, et al. Initial experience with robot-assisted modified radical neck dissection for the management of thyroid carcinoma with lateral neck node metastasis. Surgery 2010;148:1214-21. [PubMed]
- Saki N, Nikakhlagh S. Complications of neck dissection. Rijeka: In Tech, 2012.
- Saffold SH, Wax MK, Nguyen A, et al. Sensory changes associated with selective neck dissection. Arch Otolaryngol Head Neck Surg 2000;126:425-8. [PubMed]
- Weiss KL, Wax MK, Haydon RC 3rd, et al. Intracranial pressure changes during bilateral radical neck dissections. Head Neck 1993;15:546-52. [PubMed]
- Roh JL, Yoon YH, Kim SY, et al. Cervical sensory preservation during neck dissection. Oral Oncol 2007;43:491-8. [PubMed]
- Harish K.. Neck dissections: radical to conservative. World J Surg Oncol 2005;3:21. [PubMed]
- Spiro RH, Gallo O, Shah JP. Selective jugular node dissection in patients with squamous carcinoma of the larynx or pharynx. Am J Surg 1993;166:399-402. [PubMed]
- Traynor SJ, Cohen JI, Gray J, et al. Selective neck dissection and the management of the node-positive neck. Am J Surg 1996;172:654-7. [PubMed]


