
Patient reported outcome measures (PROMs) following mastectomy with breast reconstruction or without reconstruction: a systematic review
Introduction
Breast cancer is the second most common cause of cancer related death in females worldwide. It is estimated that over one million women are diagnosed globally each year with over 450,000 cancer related deaths (1). Mastectomy still has its consolidated role in breast surgery, although the refinement in screening and surgical techniques has led to the increase use of breast conserving surgery (2). Irrespective of the type of breast surgery chosen, a great proportion of women who undergo oncologic breast surgery perceive a negative self-image and experience negative changes in their sexuality and well-being following treatment (3).
The cornerstone of reconstructive surgery following mastectomy is to restore cosmesis and improve physical and psychological health. Because of such, it has become essential for instruments that measure surgical outcomes to include the direct perspective of the patient on reconstruction results. This change in standpoint to a patient centred approach has led to the development of patient reported outcome measures (PROM) specific to breast surgery, gaining significant popularity and validity over the past decade (4). Prior to the implementation of quality-of-life (QoL) evaluating tools, the majority of studies focused on surgical based outcomes such as morbidity and mortality. Moreover, only limited numbers of studies published prior to the year 2000 had direct patient input, with the majority designed for research and seldon included patient reports directly (5).
Previous reviews have failed to show favourable patient reported outcomes for breast reconstruction following mastectomy compared to patients undergoing mastectomy alone (6). This can likely be attributed to the relatively modern development of validated PROM tools used in breast reconstruction, which previous studies have lacked, and advances in surgical techniques. Given these limitations, the purpose of this systematic review is to evaluate the modern literature comparing PROMs of patients undergoing breast reconstruction following mastectomy compared to patient undergoing mastectomy as a single procedure.
Method
Search strategy
A systematic literature review was performed using the CINAHL, Cochrane Library and Medline (using PubMed) adhering to the PRISMA statement (7). The keywords selected were: (patient reported outcome measures OR quality of life OR BREAST-Q OR satisfaction) AND (breast reconstruction OR implant OR DIEP OR TRAM OR latissimus dorsi) AND mastectomy. Articles were searched electronically by the main author with the latest entry added on the 31/5/2018. Reference lists of identified studies and previously published reviews were also explored.
Inclusion criteria
Only articles published in the English or Portuguese language from the year 2000 were included. Portuguese language was added to the review due to the first author being fluent in the language. Eligible articles included patients who underwent mastectomy recommended for invasive breast cancer or carcinoma in situ. Selected studies had to compare patients undergoing mastectomy alone against a cohort of patient undergoing mastectomy followed by breast reconstruction, thus, articles comparing different reconstruction options without a mastectomy alone control group were excluded. Studies were required to include patient reported outcome measures or any quality of life measure reported directly by patients. Articles only including mastectomy for prophylactic purposes were excluded with view that the holistic treatment for breast cancer has a direct influence on quality of life measures.
The process of identifying articles is summarised in Figure 1 as a PRISMA statement diagram.
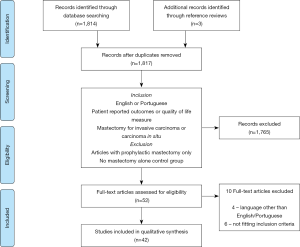
Results
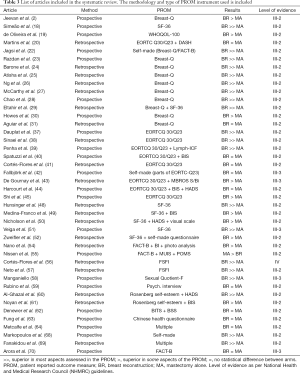
The literature review yielded 52 articles for full text examination. Four were excluded due to being written in a language other than English or Portuguese (8-11) and 6 were excluded for not fitting other elements of the inclusion criteria (12-17). Thus, 42 studies comprised this appraisal, of which three were written in Portuguese (18-20). Twenty one articles were prospective in nature and 28 were published after the year 2010. The countries with the most publications were the USA and Brazil with 7 studies each closely followed by the UK with 4. Geographical differences in results are shown in Table 1. Totalling all articles, over 9,700 patients underwent mastectomy alone and 6,600 patients underwent breast reconstruction.

Full table
Sample size varied significantly when comparing studies, from the smallest with 21 patients (18) to the largest with 6,882 (2). Eleven articles used at least one of the Breast-Q modules as their prime PROM tool, followed by EORTC-Q30/Q23 with ten studies, Short Form 36 with seven studies and FACT-B with four studies. The remainder of articles used PROM tools including self-made questionnaires, psychiatric evaluations, self-esteem assessments and sexual well-being scales.
Patient reported outcome measure instruments
Breast-Q
The PROM Breast-Q (21) was designed in the mid to late 2000s with the aim to measure the quality of life and satisfaction among breast surgery patients undergoing breast reconstruction. Breast-Q contains the following modules: psychosocial well-being, physical well-being, sexual well-being, satisfaction with breasts and satisfaction with care. Segments have further been developed for different surgery types including breast conservation, mastectomy and breast reconstruction, thus allowing for comparisons across divergent surgical groups.
Eleven articles have utilised Breast-Q as their primary outcome measure (2,22-31). Ten of these showed an improvement in Breast-Q score in patients where breast reconstruction was performed. Cancer staging was worst in the mastectomy arm in three studies, potentially confounding results (22,24,30). The single study that failed to show a difference between breast reconstruction compared to mastectomy alone utilised the single psychosocial well-being subscale of Breast-Q and breast implants was the reconstruction of choice (31). The average subscale scores of articles that utilised Breast-Q are displayed in Table 2.

Full table
EORTC-Q30/Q23
The European organization for research and treatment of cancer-Q30 (EORTC-Q30) is a general 30-item questionnaire for health-related quality of life assessment in cancer patients (32). EORTC-Q23 is a complementary breast cancer specific questionnaire that is generally used together with EORTC-Q30 and constitutes of 2 distinct scales (33). Body image, sexual functioning, future perspective and sexual enjoyment forms the functional scale, whereas domains of the symptoms scale include arm symptoms, breast symptoms, side effects of systematic therapy and hair loss (33,34). Despite been validated for the use in breast cancer surgery, both EORTC-Q30 and Q23 were not certified for use in breast reconstruction (35). The development of QLQ-BRR26, a breast reconstruction specific scale, aims to be used in conjunction to EORTC-Q30/Q23 and it is still undergoing phase 3 development (36).
Ten studies used EORTC-Q30/Q23 as their primary PROM instrument (20,37-45). Fallbjörk et al. used an ad hoc questionnaire with inclusion of the EORTC-Q23 and was therefore included in this group (42). Three studies showed an improvement in QoL with breast reconstruction, 5 showed no difference between arms and 2 showed improvement in some PROM domains but not in global QoL. The majority of studies had age disparities between groups, the mastectomy alone cohort being significantly older.
Short form 36 (SF-36)
The SF-36 is a summarised version of 149 validated health-related questions first reported as part of a medical outcomes study of more than 22,000 subjects (46). It entails 36 items concerned with the assessment of QoL and contains eight domains: limitations in physical activities, limitations in social activities, limitations in role activities because of physical problems, limitations in role activities because of emotional problems, bodily pain, general mental health, vitality and general health perceptions (47). Despite its general usability, the SF-36 has not been validated for the use in breast cancer or breast reconstruction patients. In view of such, items such as body image, femininity and sexual well-being, may not be contemplated by the scales of the SF-36 (16). Furthermore, the scores can be distorted by comorbidities other than breast cancer that may affect patient is quality of life.
Seven articles used SF-36 as their primary patient reported outcome measure tool (18,48-52) and one article used both Breast-Q in conjunction with SF-36 (29). All articles showed that breast reconstruction patients had superior PROMs in at least one of the SF-36 domains when compared to mastectomy alone. In three studies, nearly all measures of SF-36 were better in breast reconstruction patients (18,48,51). Medina-Franco found statistically better reported outcomes only in younger females (49). The remaining 3 studies had significant changes only in some of the SF-36 domains and supplemented their methods with the addition of other PROM instruments—ultimately showing an overall increase in patient reported quality of life domains (29,50,52).
FACT-B
A Functional Assessment of Cancer Therapy-Breast (FACT-B) quality of life questionnaire is a patient reported outcome tool utilised for the assessment of QoL after breast surgery. It has been previously validated on 295 breast cancer patients (53). The FACT–B questionnaire includes the domains of physical well-being, social well-being, emotional well-being, functional well-being and doctor relationship. Similar to the EORTC questionnaires, FACT-B has not been validated in the breast reconstruction population.
Four studies utilised FACT-B as their PROM of choice. Two studies found better physical wellbeing and body image in the breast reconstruction group, with other FACT-B measures not having a significant change (22,54). One particular study by Nissen and colleagues found that patients who underwent breast reconstruction scored significantly lower than those who underwent mastectomy alone in the domains of physical, emotional and functional well-being (55).
Other PROM tools
The remaining 14 studies used a myriad of other patient reported outcome tools. Three studies used sexual function scales, with all three finding better patient reported outcomes in the breast reconstruction group (56-58). Three studies used psychometric evaluation tools to assess mainly for anxiety and depression (59-61). Specific body image questionnaires were utilised in eight studies, with or without other PROM instruments used in combination (40,44,49,54,61-64). Examples of PROM instruments used included the Body Image after Breast Cancer (65), Body Image Scale (66) and the Body Satisfaction Scale (67). Three studies used their own questionnaire bases on other available tools (22,42,68).
PROM results
Eleven studies found that PROM were similar between patients that undertook breast reconstruction to that of patients who underwent mastectomy as a single procedure. Thirty studies had statistical significant improvement in the PROM with breast reconstruction, especially in the domains of body image, sexual well-being and breast satisfaction. Only a single study found worst quality of life measures in the breast reconstruction arm (55). Articles included in this review and the PROM results comparing breast reconstruction with mastectomy alone can be found in Table 3.
Full table
Discussion
Undergoing breast reconstruction following mastectomy should optimally be an informed decision by the patient. Thus, all the articles included in this review are limited by the absence of randomisation. The result is an abundant dissimilarity between clinical and demographic characteristics between the breast reconstruction and mastectomy-alone groups, some of these are explored below.
Age difference
Matching the two study arms according to age proved to be a substantial challenge for most studies. Twenty-one studies reported statistically significant (P<0.05) age difference between breast reconstruction and mastectomy groups, reconstruction patients tended to be younger. When translated into PROM results, six of these studies showed breast reconstruction to be equal or worst to mastectomy alone. Two studies subdivided their groups according to age and found contrasting results (48,49).
Efforts of comparison have limitations because of the disparities in age-related quality of life determinants. Generally, younger patients with breast cancer are more concerned with their physical appearance and femininity (19,71). In contrast, older breast surgery patients often view their breast appearance as a less important aspect of their quality of life and choose less involved operations to avoid complications (37). Complex reconstruction is paired with longer operative time, donor site morbidity and slower return to mobility, all of which could have detrimental effects on the elderly (55). Enewold et al. noted that the frequency of patient request for breast reconstruction significantly decreases with increasing age (72). Thus, given differences in age-related priorities, the implications of an older mastectomy-alone cohort would result in significant bias in patient reported outcomes. Similar can be said about a younger breast reconstruction group.
Socioeconomic differences
The impact of breast reconstruction in body image perception and quality of life is influenced by patient socioeconomic background and educational level (49). Fourteen studies had statistically significant disparities when comparing socioeconomic backgrounds. Patients undergoing breast reconstruction were more likely to be employed, live in urban centres, have higher income or have higher level of education when compared to patients undergoing mastectomy alone. The result would be a distortion in PROMs in support for breast reconstruction.
Studies from Turkey and Iran depicted the financial challenges of women who would like breast reconstruction but are unable to afford it (38,61). Having a lower education and socioeconomic background was also associated with poor pre-surgical decision making and increased risk of reporting low satisfaction (61).
Lymphoedema and adjuvant therapy
The majority of studies did not include their rate of lymphedema following the oncological procedures. Data can be extrapolated by taking into account the rates of stage 3 breast cancer and axillary lymph nodes resections although these were also poorly reported. Eight studies narrated the presence of significant disparities in cancer stage, lymphadenectomies or axillary clearance between groups. The majority reported worst staging within the mastectomy-alone arm. Breast cancer related lymphoedema has been shown to have a negative effect on the QoL of breast cancer survivors, possibly negating the positive effects of breast reconstruction (39).
Adjuvant therapies have been shown to negatively affect health-related quality of life (28). Seven articles reported disparities between rates of chemotherapy and radiotherapy. Four studies had higher rates of adjuvant treatment in the breast reconstruction arm (40,44,55,62). None of the 4 studies narrated superior PROMs with breast reconstruction, indicating that higher rates of adjuvant therapy may have negated the effects of the plastic surgery. In contrast, the remaining studies in which adjuvant therapies were more prominent in the mastectomy arms showed significant better outcomes with breast reconstruction (28,37,39).
Time since surgery and timing of reconstruction
With time, women tend to experience changes in the shape and outcomes of their breast reconstruction, and intuitively a changing nature of PROMs. Over time, women who choose to undergo implant reconstruction experience decreased aesthetic satisfaction compared with autologous tissue reconstruction (2). As time passes, reduction in breast satisfaction is seen with all types of reconstruction options, with gluteal and thigh flaps having the worst long-term outlook (25). In contrast, patients who opt to undergo mastectomy alone show an overall increase in reported breast satisfaction over time. Timing of the reconstruction is also an important consideration. Only ten studies explored the impact of both immediate and delayed options and compared it to mastectomy-alone. Six of these showed no difference in PROMs, and 3 supported immediate breast reconstruction (IBR).
The long-term variations in quality of life measures in breast reconstruction are difficult to study because any health change may lead to shifts in health related QoL domains (73). Particular to breast cancer surgery, the first few months following an operation are usually accompanied by adjuvant therapies and surgical recovery. Measuring patient reported outcomes within this period produces lower PROM results. Similarly, studies trying to establish evidence of satisfactory restoration of quality of life after a long-intervals following breast surgery may encounter the confounding effects of other comorbidities. The use of specific PROM tools that target breast surgery and breast reconstruction is therefore recommended to minimise the confounding effects of other diseases.
Conclusions
The current study demonstrated that breast reconstruction following mastectomy has led to better patient reported outcomes compared to mastectomy-alone in the great majority of trials since the introduction of appropriate questionnaire instruments in the beginning of the year 2000. Care must be taken when interpreting the data as the lack of randomised controlled trial limits the control of population characteristics. Women of younger age, higher level of education and good pre-treatment health are more likely to seek breast reconstruction. We encourage the use of prospective trials due to the inability of retrospective studies to account for changes in pre-surgical and post-surgical quality of life as well as to minimise recall bias.
Quality of life embodies broader physical and psychological health, level of independence, social connections, the environment, and spiritual care. Variations can be justified by the influence of comorbidities, dealing with the diagnosis of breast cancer itself and the adjuvant therapies involved in the treatment of the disease. In view of such, the use of validated quality of life questionnaires in breast surgery and breast reconstruction is paramount to minimise the bias of co-existing health issues. Recommendations for breast reconstruction should follow the National Institute for Health and Clinical Excellence (NICE) published guidelines on improving breast cancer outcomes and reconstruction should be available to all women with breast cancer at the initial surgical operation (74).
Acknowledgments
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
References
- Tao Z, Shi A, Lu C, et al. Breast Cancer: Epidemiology and Etiology. Cell Biochem Biophys 2015;72:333-8. [Crossref] [PubMed]
- Jeevan R, Cromwell DA, Browne JP, et al. Findings of a national comparative audit of mastectomy and breast reconstruction surgery in England. J Plast Reconstr Aesthet Surg 2014;67:1333-44. [Crossref] [PubMed]
- McGaughey A. Body image after bilateral prophylactic mastectomy: an integrative literature review. J Midwifery Womens Health 2006;51:e45-9. [Crossref] [PubMed]
- Cano SJ, Klassen A, Pusic AL. The science behind quality-of-life measurement: a primer for plastic surgeons. Plast Reconstr Surg 2009;123:98e-106e. [Crossref] [PubMed]
- Voineskos SH, Nelson JA, Klassen AF, et al. Measuring Patient-Reported Outcomes: Key Metrics in Reconstructive Surgery. Annu Rev Med 2018;69:467-79. [Crossref] [PubMed]
- Lee C, Sunu C, Pignone M. Patient-reported outcomes of breast reconstruction after mastectomy: a systematic review. J Am Coll Surg 2009;209:123-33. [Crossref] [PubMed]
- Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ 2009;339:b2535. [Crossref] [PubMed]
- Giannakou K, Kneknas E. The impact of breast reconstruction in women after mastectomy in relation to quality of life. Nosileftiki 2014;53:133-40.
- Strittmatter HJ, Neises M, Blecken SR. Criteria of life quality after reconstructive breast cancer surgery. Zentralbl Gynakol 2006;128:217-23. [Crossref] [PubMed]
- Tkachenko GA, Arslanov Kh S, Iakovlev VA, et al. Long-term impact of breast reconstruction on quality of life among breast cancer patients. Vopr Onkol 2008;54:724-8. [PubMed]
- Onen Sertöz O, Elbi Mete H, Noyan A, et al. Effects of surgery type on body image, sexuality, self-esteem, and marital adjustment in breast cancer: a controlled study. Turk Psikiyatri Derg 2004;15:264-75. [PubMed]
- Cereijo-Garea C, Pita-Fernandez S, Acea-Nebril B, et al. Predictive factors of satisfaction and quality of life after immediate breast reconstruction using the BREAST-Q((c)). J Clin Nurs 2018;27:1464-74. [Crossref] [PubMed]
- Huguet PR, Morais SS, Osis MJ, et al. Rev Bras Ginecol Obstet 2009;31:61-7. [Quality of life and sexuality of women treated for breast cancer]. [Crossref] [PubMed]
- Galdino AR, Pereira LD, Costa Neto SB, et al. Qualidade de vida de mulheres mastectomizadas matriculadas em um programa de reabilitação Quality of life of mastectomized women enrolled in a rehabilitation program. J Res Fundam Care Online 2017;9:451-8. [Crossref]
- Veiga DF, Veiga-Filho J, Ribeiro LM, et al. Quality-of-life and self-esteem outcomes after oncoplastic breast-conserving surgery. Plast Reconstr Surg 2010;125:811-7. [Crossref] [PubMed]
- Tønseth KA, Hokland BM, Tindholdt TT, et al. Patient-reported outcomes after breast reconstruction with deep inferior epigastric perforator flaps. Scand J Plast Reconstr Surg Hand Surg 2007;41:173-7. [Crossref] [PubMed]
- Wilkins EG, Cederna PS, Lowery JC, et al. Prospective analysis of psychosocial outcomes in breast reconstruction: one-year postoperative results from the Michigan Breast Reconstruction Outcome Study. Plast Reconstr Surg 2000;106:1014-25; discussion 26-7. [Crossref] [PubMed]
- Simeão SF, Landro IC, De Conti MH, et al. Quality of life of groups of women who suffer from breast cancer. Cien Saude Colet 2013;18:779-88. [PubMed]
- de Oliveira RR, Morais SS, Sarian LO. Immediate breast reconstruction effects on quality of life of women with mastectomy. Rev Bras Ginecol Obstet 2010;32:602-8. [PubMed]
- Martins TNO, Santos LF, Petter GD, et al. Immediate breast reconstruction versus non-reconstruction after mastectomy: a study on quality of life, pain and functionality. Fisioter Pesqui 2017;24:412-9. [Crossref]
- Pusic AL, Klassen AF, Scott AM, et al. Development of a new patient-reported outcome measure for breast surgery: the BREAST-Q. Plast Reconstr Surg 2009;124:345-53. [Crossref] [PubMed]
- Jagsi R, Li Y, Morrow M, et al. Patient-reported Quality of Life and Satisfaction With Cosmetic Outcomes After Breast Conservation and Mastectomy With and Without Reconstruction: Results of a Survey of Breast Cancer Survivors. Ann Surg 2015;261:1198-206. [Crossref] [PubMed]
- Razdan SN, Cordeiro PG, Albornoz CR, et al. Cost-Effectiveness Analysis of Breast Reconstruction Options in the Setting of Postmastectomy Radiotherapy Using the BREAST-Q. Plast Reconstr Surg 2016;137:510e-7e. [Crossref] [PubMed]
- Barone M, Cogliandro A, Grasso A, et al. Patient's breast perception after mastectomy: analysis of outcomes in 208 patients with BREAST-Q following mastectomy alone, implant of tissue expander and definitive breast reconstruction. Plast Reconstr Surg 2018. [Epub ahead of print]. [PubMed]
- Atisha DM, Rushing CN, Samsa GP, et al. A national snapshot of satisfaction with breast cancer procedures. Ann Surg Oncol 2015;22:361-9. [Crossref] [PubMed]
- Ng SK, Hare RM, Kuang RJ, et al. Breast Reconstruction Post Mastectomy: Patient Satisfaction and Decision Making. Ann Plast Surg 2016;76:640-4. [Crossref] [PubMed]
- McCarthy CM, Mehrara BJ, Long T, et al. Chest and upper body morbidity following immediate postmastectomy breast reconstruction. Ann Surg Oncol 2014;21:107-12. [Crossref] [PubMed]
- Chao LF, Patel KM, Chen SC, et al. Monitoring patient-centered outcomes through the progression of breast reconstruction: a multicentered prospective longitudinal evaluation. Breast Cancer Res Treat 2014;146:299-308. [Crossref] [PubMed]
- Eltahir Y, Werners LL, Dreise MM, et al. Reply: quality-of-life outcomes between mastectomy alone and breast reconstruction: comparison of patient-reported breast-Q and other health-related quality-of-life measures. Plast Reconstr Surg 2014;133:595e. [Crossref] [PubMed]
- Howes BH, Watson DI, Xu C, et al. Quality of life following total mastectomy with and without reconstruction versus breast-conserving surgery for breast cancer: A case-controlled cohort study. J Plast Reconstr Aesthet Surg 2016;69:1184-91. [Crossref] [PubMed]
- Aguiar IC, Veiga DF, Marques TF, et al. Patient-reported outcomes measured by BREAST-Q after implant-based breast reconstruction: A cross-sectional controlled study in Brazilian patients. Breast 2017;31:22-5. [Crossref] [PubMed]
- Montazeri A, Harirchi I, Vahdani M, et al. The European Organization for Research and Treatment of Cancer Quality of Life Questionnaire (EORTC QLQ-C30): translation and validation study of the Iranian version. Support Care Cancer 1999;7:400-6. [Crossref] [PubMed]
- Montazeri A, Harirchi I, Vahdani M, et al. The EORTC breast cancer-specific quality of life questionnaire (EORTC QLQ-BR23): translation and validation study of the Iranian version. Qual Life Res 2000;9:177-84. [Crossref] [PubMed]
- Sprangers MA, Groenvold M, Arraras JI, et al. The European Organization for Research and Treatment of Cancer breast cancer-specific quality-of-life questionnaire module: first results from a three-country field study. J Clin Oncol 1996;14:2756-68. [Crossref] [PubMed]
- Nguyen J, Popovic M, Chow E, et al. EORTC QLQ-BR23 and FACT-B for the assessment of quality of life in patients with breast cancer: a literature review. J Comp Eff Res 2015;4:157-66. [Crossref] [PubMed]
- Winters ZE, Balta V, Thomson HJ, et al. Phase III development of the European Organization for Research and Treatment of Cancer Quality of Life Questionnaire module for women undergoing breast reconstruction. Br J Surg 2014;101:371-82. [Crossref] [PubMed]
- Dauplat J, Kwiatkowski F, Rouanet P, et al. Quality of life after mastectomy with or without immediate breast reconstruction. Br J Surg 2017;104:1197-206. [Crossref] [PubMed]
- Sinaei F, Zendehdel K, Adili M, et al. Association Between Breast Reconstruction Surgery and Quality of Life in Iranian Breast Cancer Patients. Acta Med Iran 2017;55:35-41. [PubMed]
- Penha TR, Botter B, Heuts EM, et al. Quality of Life in Patients with Breast Cancer-Related Lymphedema and Reconstructive Breast Surgery. J Reconstr Microsurg 2016;32:484-90. [Crossref] [PubMed]
- Spatuzzi R, Vespa A, Lorenzi P, et al. Evaluation of Social Support, Quality of Life, and Body Image in Women with Breast Cancer. Breast Care (Basel) 2016;11:28-32. [Crossref] [PubMed]
- Cortés-Flores AO, Morgan-Villela G, Zuloaga-Fernandez del Valle CJ, et al. Quality of life among women treated for breast cancer: a survey of three procedures in Mexico. Aesthetic Plast Surg 2014;38:887-95. [Crossref] [PubMed]
- Fallbjörk U, Rasmussen BH, Karlsson S, et al. Aspects of body image after mastectomy due to breast cancer - a two-year follow-up study. Eur J Oncol Nurs 2013;17:340-5. [Crossref] [PubMed]
- De Gournay E, Bonnetain F, Tixier H, et al. Evaluation of quality of life after breast reconstruction using an autologous latissimus dorsi myocutaneous flap. Eur J Surg Oncol 2010;36:520-7. [Crossref] [PubMed]
- Harcourt DM, Rumsey NJ, Ambler NR, et al. The psychological effect of mastectomy with or without breast reconstruction: a prospective, multicenter study. Plast Reconstr Surg 2003;111:1060-8. [Crossref] [PubMed]
- Shi HY, Uen YH, Yen LC, et al. Two-year quality of life after breast cancer surgery: a comparison of three surgical procedures. Eur J Surg Oncol 2011;37:695-702. [Crossref] [PubMed]
- Patel AA, Donegan D, Albert T. The 36-item short form. J Am Acad Orthop Surg 2007;15:126-34. [Crossref] [PubMed]
- Ware JE Jr, Sherbourne CD. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med Care 1992;30:473-83. [Crossref] [PubMed]
- Hunsinger V, Hivelin M, Derder M, et al. Long-Term Follow-Up of Quality of Life following DIEP Flap Breast Reconstruction. Plast Reconstr Surg 2016;137:1361-71. [Crossref] [PubMed]
- Medina-Franco H, Garcia-Alvarez MN, Rojas-Garcia P, et al. Body image perception and quality of life in patients who underwent breast surgery. Am Surg 2010;76:1000-5. [PubMed]
- Nicholson RM, Leinster S, Sassoon EM. A comparison of the cosmetic and psychological outcome of breast reconstruction, breast conserving surgery and mastectomy without reconstruction. Breast 2007;16:396-410. [Crossref] [PubMed]
- Veiga DF, Sabino Neto M, Ferreira LM, et al. Quality of life outcomes after pedicled TRAM flap delayed breast reconstruction. Br J Plast Surg 2004;57:252-7. [Crossref] [PubMed]
- Zweifler M, Rodriguez E, Reilly J, et al. Breast reconstruction among inner city women with breast carcinoma. Ann Plast Surg 2001;47:53-9. [Crossref] [PubMed]
- Brady MJ, Cella DF, Mo F, et al. Reliability and validity of the Functional Assessment of Cancer Therapy-Breast quality-of-life instrument. J Clin Oncol 1997;15:974-86. [Crossref] [PubMed]
- Nano MT, Gill PG, Kollias J, et al. Psychological impact and cosmetic outcome of surgical breast cancer strategies. ANZ J Surg 2005;75:940-7. [Crossref] [PubMed]
- Nissen MJ, Swenson KK, Ritz LJ, et al. Quality of life after breast carcinoma surgery: a comparison of three surgical procedures. Cancer 2001;91:1238-46. [Crossref] [PubMed]
- Cortés-Flores AO, Vargas-Meza A, Morgan-Villela G, et al. Sexuality Among Women Treated for Breast Cancer: A Survey of Three Surgical Procedures. Aesthetic Plast Surg 2017;41:1275-9. [Crossref] [PubMed]
- Neto MS, de Aguiar Menezes MV, Moreira JR, et al. Sexuality after breast reconstruction post mastectomy. Aesthetic Plast Surg 2013;37:643-7. [Crossref] [PubMed]
- Manganiello A, Hoga LA, Reberte LM, et al. Sexuality and quality of life of breast cancer patients post mastectomy. Eur J Oncol Nurs 2011;15:167-72. [Crossref] [PubMed]
- Rubino C, Figus A, Lorettu L, et al. Post-mastectomy reconstruction: a comparative analysis on psychosocial and psychopathological outcomes. J Plast Reconstr Aesthet Surg 2007;60:509-18. [Crossref] [PubMed]
- Al-Ghazal SK, Fallowfield L, Blamey RW. Comparison of psychological aspects and patient satisfaction following breast conserving surgery, simple mastectomy and breast reconstruction. Eur J Cancer 2000;36:1938-43. [Crossref] [PubMed]
- Noyan MA, Sertoz OO, Elbi H, et al. Variables affecting patient satisfaction in breast surgery: a cross-sectional sample of Turkish women with breast cancer. Int J Psychiatry Med 2006;36:299-313. [Crossref] [PubMed]
- Denewer A, Farouk O, Kotb S, et al. Quality of life among Egyptian women with breast cancer after sparing mastectomy and immediate autologous breast reconstruction: a comparative study. Breast Cancer Res Treat 2012;133:537-44. [Crossref] [PubMed]
- Fung KW, Lau Y, Fielding R, et al. The impact of mastectomy, breast-conserving treatment and immediate breast reconstruction on the quality of life of Chinese women. ANZ J Surg 2001;71:202-6. [Crossref] [PubMed]
- Metcalfe KA, Semple J, Quan ML, et al. Changes in psychosocial functioning 1 year after mastectomy alone, delayed breast reconstruction, or immediate breast reconstruction. Ann Surg Oncol 2012;19:233-41. [Crossref] [PubMed]
- Baxter NN, Goodwin PJ, McLeod RS, et al. Reliability and validity of the body image after breast cancer questionnaire. Breast J 2006;12:221-32. [Crossref] [PubMed]
- Hopwood P, Fletcher I, Lee A, et al. A body image scale for use with cancer patients. Eur J Cancer 2001;37:189-97. [Crossref] [PubMed]
- Andersen BL, Legrand J. Body Image for Women: Conceptualization, Assessment, and a Test of its Importance to Sexual Dysfunction and Medical Illness. J Sex Res 1991;28:457-77. [Crossref] [PubMed]
- Markopoulos C, Tsaroucha AK, Kouskos E, et al. Impact of breast cancer surgery on the self-esteem and sexual life of female patients. J Int Med Res 2009;37:182-8. [Crossref] [PubMed]
- Fanakidou I, Zyga S, Alikari V, et al. Mental health, loneliness, and illness perception outcomes in quality of life among young breast cancer patients after mastectomy: the role of breast reconstruction. Qual Life Res 2018;27:539-43. [Crossref] [PubMed]
- Arora NK, Gustafson DH, Hawkins RP, et al. Impact of surgery and chemotherapy on the quality of life of younger women with breast carcinoma: a prospective study. Cancer 2001;92:1288-98. [Crossref] [PubMed]
- Wenzel LB, Fairclough DL, Brady MJ, et al. Age-related differences in the quality of life of breast carcinoma patients after treatment. Cancer 1999;86:1768-74. [Crossref] [PubMed]
- Enewold LR, McGlynn KA, Zahm SH, et al. Breast reconstruction after mastectomy among Department of Defense beneficiaries by race. Cancer 2014;120:3033-9. [Crossref] [PubMed]
- Hamidou Z, Dabakuyo TS, Bonnetain F. Impact of response shift on longitudinal quality-of-life assessment in cancer clinical trials. Expert Rev Pharmacoecon Outcomes Res 2011;11:549-59. [Crossref] [PubMed]
- National Institute for Clinical Excellence. Guidance on cancer services. Improving outcomes in breast cancer e manual update. London: NICE 2002. Available online: http://www.nice.org.uk/nicemedia/pdf/Improving_outcomes_breastcancer_manual.pdf


