Robotic adrenalectomy
Introduction
With the increased use of robotics in the operating theatre, robotic adrenalectomy has been adopted by many high-volume tertiary centers as an alternative treatment to conventional laparoscopic adrenalectomy. In adrenal gland surgery, minimally invasive surgery has been proven being effective, compared to open adrenalectomy. It is accepted as the standard surgical method for the majority of adrenal masses. Robotic adrenalectomy is a safe, feasible and effective method, which is positioned as an option, since it is accepted as associated with a three-dimensional perception, a tremor-free surgery, improved moving capacity of the robotic arms with multi-articulation and precise camera control, increasing the ergonomics of this type of surgery (1). These advantages are useful, especially when meticulous dissection is warranted in a deep and narrow field. Overall, this robotic technology allows us to carry out complex tasks in a minimally invasive manner, with an expected faster learning curve than conventional laparoscopy. In adrenal surgery, the application of robotics has not gained that much popularity among surgeons. On the other hand, the “robotic endocrine surgeon” places robot-assisted adrenal surgery as a potential procedure largely supplanting conventional laparoscopic adrenalectomy. The aim of the present review is to give an overview of robotic adrenalectomy and summarize recent outcomes in this field.
Surgical technique
Preoperative preparation
Surgery is performed under general anesthesia. Preoperative preparation and positioning of the patient is the same as for laparoscopic adrenalectomy. The transperitoneal approach provides greater working space, facilitates orientation by providing readily identifiable anatomical landmarks and better visualization of surrounding anatomical structures. The patient is placed in the left or right lateral decubitus position according the tumor location (left decubitus for right side adrenal tumor and right decubitus for left side tumor) and positioned in a slight Trendelenburg position. Extreme care is taken with pressure points and correctly padding them with pillows and foams. Four trocars are used for left adrenalectomy and one additional port used for right adrenalectomy to aid in liver retraction.
Setup of patient
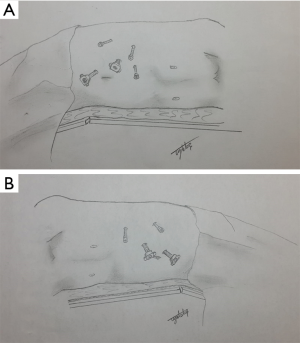
Currently, robotic surgery is performed with the DaVinci Si or Xi robotic surgical systems (Intuitive Surgical Sarl, Aubonne, Switzerland). These systems consist of three-arm robotic manipulator and remote-control surgical console. After positioning the patient, four or five trocars are used for the procedure. After entering the abdomen with open technique, following insufflation, one camera port (12 mm), two ports for the robotic arms (8 mm) and one port for manual assistance (12 mm) (and one additional port for right sided to aid in liver retraction) are placed. First the camera port (12 mm) is inserted above and lateral to the umbilicus, at the lateral border of the abdominal rectus muscle across from the 12th rib. After the first port insertion, the endoscope is inserted and the abdomen is carefully inspected to rule out any accidental injuries or in terms of other intra-abdominal masses.
After exploration, other ports are inserted under direct vision (Figure 1A,B). To avoid clashing between the robotic arms, the ports must be placed about 8–10 cm in distance from each other. The role of the assistant on the surgical table is to change the robotic instruments when necessary, to assist in dissection through the assistant’s port, to attach the clip to the adrenal vein or to seal with the vessel sealing device and to perform the wash & aspiration process. A 30-degree endoscope is used for surgery.
Steps of surgery
Left-sided robotic adrenalectomy
Left-sided robotic transabdominal adrenalectomy is performed with the patient in the right lateral decubitus position. For the perfect gland exposure, a complete medial colonic mobilization may be needed. After dividing the lateral adhesions of the spleen and splenorenal ligaments, the spleen, colon, and the pancreas are mobilized medially until the adrenal gland is clearly visualized. During this mobilization, attention should be paid to the tail of the pancreas. Dissection continues into the periadrenal fat in order to identify the following landmarks: inferiorly the left renal vein, laterally the superior pole of the kidney, medially the tail of the pancreas and splenic vessels and posteriorly the psoas muscle. Then the adrenal vein is carefully dissected and clipped by using the robotic clip applier or standard laparoscopic clips placed by the bedside assistant. A vessel sealer also can be used. For hemostasis control, before the mass is removed from the abdomen, it is advised to wait 3–4 minutes after the intra-abdominal gas is evacuated and re-check the operation site. After the adrenalectomy is complete, the robot is undocked, and the gland is removed using a specimen retrieval bag and delivered through the auxiliary port site. After the operative site is irrigated and suctioned, trocars are removed.
Right-sided robotic adrenalectomy
Right-sided robotic transabdominal adrenalectomy is performed with the patient placed in the left lateral decubitus position. On this, different than the left side, five trocars are used (one additional port to aid in liver retraction). The triangular ligament is divided via a robotic monopolar hook and the liver retracted with a laparoscopic retractor superiorly by the assistant to provide wide exposure of the inferior vena cava. Gerota’s fascia is incised at the level of the upper pole of the kidney. After the precise dissection of the vena cava and identify the major landmarks (laterally the superior pole of the right kidney, posteriorly the psoas muscle), the right adrenal vein is identified and divided after a clip or vessel sealer has been applied by the assistant. After completion of the dissection of the adrenal space with robotic hook, bipolar forceps and/or vessel sealer, hemostasis is controlled as in left adrenalectomy, and the gland is removed using a specimen retrieval bag.
Bilateral robotic adrenalectomy
Surgery of both adrenal glands as a single operation is possible, also for minimally invasive adrenalectomy. This can be carried out either retroperitoneally or peritoneally with the robotic surgical system. Repositioning is warranted in the peritoneal approached cases that will undergo a lateral decubitus positioning. For these cases, two ports (the assistant and most medial ports) can be used in common to avoid redundant trocar insertion. Details regarding trocar positioning and surgical technique for robot-assisted laparoscopic bilateral adrenalectomy have been described by us previously (2).
Postoperative care
After surgery, the patient is kept in the intensive care unit for a few hours. Antibiotics and prophylaxis for deep vein thrombosis are given according to hospital protocol to all patients. Pain control is performed using intravenous non-opioid and intramuscular opioid analgesics. The vital signs and fluid balances of the patients are closely monitored for the first 24 hours. Oral intake of the patients will be started on the first day of surgery with clear liquids and gradually advanced to a normal diet. All patients are mobilized at 8 hours postoperatively. The urethral catheter is generally removed on the first day and—if used—the drain is removed on the first or second day. Patients who do not develop additional complications are discharged on the 2nd or 3rd day after surgery.
Outcomes
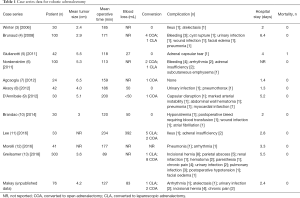
The benefit of robotic adrenalectomy compared to conventional laparoscopic adrenalectomy is still a matter of debate. Although the literature seems to be fruitful especially for the last decade, it is lacking of high quality trials in terms of evidence-based medicine. Table 1 represents perioperative outcomes after robotic transabdominal adrenalectomy in some case studies (3-13). In this section, an overview of comparative assessment of robotic adrenalectomy and conventional laparoscopic adrenalectomy in terms of efficiency and cost will be presented.
Full table
A controversy to be taken into consideration in minimal invasive adrenal gland surgery is whether the technique of posterior retroperitoneal approach or lateral transabdominal approach is a more appropriate method. Although the results of both techniques are similar, in 2013, the minimal surgical approach protocol of the American Society for Gastrointestinal and Endoscopic Surgery (SAGES) has provided recommendations regarding the surgical technique for adrenal gland pathologies for specific clinical conditions (14). These recommendations are:
- Retroperitoneal approach may result in shorter operative time and less complications in the presence of previous abdominal surgery;
- In patients undergoing bilateral adrenalectomy, the posterior retroperitoneal approach may be a more appropriate option because of the advantages of not having to re-position the patient during the operation;
- The lateral transabdominal approach in morbid obese patients (body mass index >35 kg/m2) and for large tumors (>6 cm) is more advantageous in terms of applicability compared to other surgical methods.
Although these recommendations help the surgeons in the surgical procedure, it is important to note that the most appropriate surgical technique is the surgeon’s most experienced technique. Regarding robotic surgery, Kahramangil & Berber showed that both robotic approaches, retroperitoneal and transabdominal are equally safe and efficacious, based on their experience in 200 robotic adrenalectomies (15).
In the literature, there are many studies comparing robotic and conventional laparoscopic adrenalectomy. There have been numbers of systematic reviews and meta-analysis (16-19). Before focusing on the outcomes of these meta-analyses we should mention a few words regarding the learning curve for robotic adrenalectomy. Brunaud et al. defined the learning curve for transabdominal robotic adrenalectomy as 20 cases and found that the previous clinical experience and duration of first assistance are effective factors (4).
Complications
Complications associated with robotic adrenalectomy are hemorrhage, hematoma, wound infection, urinary tract infection, adjacent organ injuries, ileus, complications due to the laparoscopic procedure and atelectasis. The complication rate between the robotic and laparoscopic groups was similar in many studies. Besides, postoperative morbidity and mortality have been shown to be comparable to conventional laparoscopy (1). A recent comprehensive meta-analysis, pooling 1,162 (747 robotic and 415 conventional laparoscopic) cases out of 27 studies, revealed that there was no significant difference between the robotic and the laparoscopic groups for intraoperative complications [odds ratio (OR): 1.20; 95% CI, 0.33–4.38], postoperative complications (OR: 0.69; 95% CI, 0.36–1.31), mortality (OR: 0.42; 95% CI, 0.07–2.72), conversion to laparotomy (OR: 0.51; 95% CI, 0.21–1.23) and conversion to laparotomy or laparoscopy (OR: 0.73; 95% CI, 0.32–1.69) (16). Data regarding complications was also supported by another detailed recent systematic review (19). In a paper carried out by Greilsamer et al., based on an experience of more than 300 robotic cases, independent risk factors for perioperative complications after robotic-assisted unilateral adrenalectomy were described as conversion to laparotomy and patient age. Tumor size >5 cm was the only predictive factor for conversion to laparotomy in that series (13).
Operative time
The publications that have longer operative time are publications in which the robotic technology is being used for the first time and the first experiences of the teams performing the work are reflected. A systematic review published in 2014 by Chai et al. compared robotic and laparoscopic adrenalectomy. There were six studies in accordance with the compilation criteria, and in the first two studies that matched the early stages of the use of robotic surgery, the operation time was longer in the robotic group, but in the other four studies, there was no significant difference between the groups in terms of operative time (20). In the more recent meta-analysis of Economopoulos et al., there was a significantly longer operating time for patients treated with robotic adrenalectomy (16). Another recent meta-analysis, including 1,710 open and minimally invasive adrenalectomies, revealed that operative time was significantly shorter for open adrenalectomy than for the robotic approach and there were no differences were found between laparoscopic and robotic approaches (19).
Blood loss & hospital stay
According to an early systematic review, two studies reported lower blood loss for robotic adrenalectomy. In one of these, it was emphasized that robotic had less pain on the first postoperative day. While the length of hospitalization was similar in both groups, one study reported a shorter hospital stay in the robotic group (20).
In the meta-analysis and systematic reviews comparing the robotic adrenalectomy with laparoscopic adrenalectomy published in 2017, Agrusa et al. reviewed 13 papers that met the criteria and compared 798 patients, 379 of whom underwent robotic surgery and 419 of them underwent laparoscopic surgery. There was no significant difference in age, gender, laterality and tumor size between the two groups, whereas the robotic group had significantly less blood loss and shorter hospital stay (21). This was also the case in the review of Economopoulos et al. for hospital stay, but not for blood loss. In their analysis they report no significant difference between groups in terms of blood loss. It is important to mention that they pooled 1,162 cases (16). The meta-analysis of Heger et al. reports the superiority of robotic adrenalectomy, regarding blood loss and hospital stay, after pooling 1,710 cases out of 26 trials (19).
Cost analysis
In a very recent study conducted by Feng et al., costs of 58 patients undergoing robotic adrenalectomy and 64 patients undergoing laparoscopic adrenalectomy were calculated. According to this study, cost calculations were made on the anesthesia fee, procedure time and consumable fees. Calculated relative costs were $3,527 for the robotic procedure, while it was $3,430 for the conventional laparoscopic procedure (P=0.59). The average duration of anesthesia was 172.4 and 178.3 min (P=0.40) for the robotic and laparoscopic approach respectively, and the average operative time was 124.4 min for robotic surgery and 129.1 min for laparoscopic surgery (P=0.50). The procedure time for the retroperitoneal approach was significantly shorter than the transabdominal approach for both robotic (101.2 vs. 126.6 min, P=0.001) and laparoscopic group (104.4 vs. 135.4 min, P=0.001). Average consumable prices were reported as $1,106 for robotic adrenalectomy and $1,009 for laparoscopic adrenalectomy (P=0.62). This study shows that anesthesia and procedure times for robotic adrenalectomy are similar to those of laparoscopic adrenalectomy and that the cost of robotic surgery can be comparable to that of conventional laparoscopic surgery by limiting the number of robotic instruments and energy devices by an experienced surgical team (22). Bodner et al. reported adrenalectomy to be 1.5 times more expensive compared to laparoscopic adrenalectomy (23), while another study reported no difference in cost outcomes (3).
From a cost perspective, shortening the length of hospital stay, improving ergonomics for the surgeon and the good perioperative outcomes in some difficult cases such as presence of large masses, obesity, and history of abdominal surgery may reduce the additional costs associated with the robotic system.
Summary
Robot-assisted adrenalectomy is a safe and effective treatment for the management of most adrenal masses. This is what comes out of the more than 200 published reports in the current literature. Compared to conventional laparoscopic surgery, cumulative level III evidence indicates similar rates of intraoperative and postoperative complications, similar operative times, less blood loss and decreased duration of stay. Definitive outcomes studies, including randomized controlled trials have yet to establish its benefits and costs relative to the conventional laparoscopic approach.
Acknowledgments
All authors like to thank Tayfun Yoldas, MD for his outstanding drawings.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Teo XL, Lim SK. Robotic assisted adrenalectomy: Is it ready for prime time? Investig Clin Urol 2016;57:S130-46. [Crossref] [PubMed]
- Makay Ö, Uǧuz A, Uǧuz I, et al. Robot-assisted laparoscopic bilateral adrenalectomy: A case report. Turk J Surg 2012;28:225-8. [Crossref]
- Winter JM, Talamini MA, Stanfield CL, et al. Thirty robotic adrenalectomies: a single institution’s experience. Surg Endosc 2006;20:119-24. [Crossref] [PubMed]
- Brunaud L, Ayav A, Zarnegar R, et al. Prospective evaluation of 100 robotic-assisted unilateral adrenalectomies. Surgery 2008;144:995-1001. [Crossref] [PubMed]
- Giulianotti PC, Buchs NC, Addeo P, et al. Robot-assisted adrenalectomy: a technical option for the surgeon? Int J Med Robot 2011;7:27-32. [Crossref] [PubMed]
- Nordenström E, Westerdahl J, Hallgrimson P, et al. A prospective study of 100 robotically assisted laparoscopic adrenalectomies. J Robot Surg 2011;5:127-31. [Crossref] [PubMed]
- Agcaoglu O, Aliyev S, Karabulut K, et al. Robotic versus laparoscopic resection of large adrenal tumors. Ann Surg Oncol 2012;19:2288-94. [Crossref] [PubMed]
- Aksoy E, Taskin HE, Aliyev S, et al. Robotic versus laparoscopic adrenalectomy in obese patients. Surg Endosc 2013;27:1233-6. [Crossref] [PubMed]
- D’Annibale A, Lucandri G, Monsellato I, et al. Robotic adrenalectomy: technical aspects, early results and learning curve. Int J Med Robot 2012;8:483-90. [Crossref] [PubMed]
- Brandao LF, Autorino R, Zargar H, et al. Robot-assisted laparoscopic adrenalectomy: step-by-step technique and comparative outcomes. Eur Urol 2014;66:898-905. [Crossref] [PubMed]
- Lee GS, Arghami A, Dy BM, et al. Robotic single-site adrenalectomy. Surg Endosc 2016;30:3351-6. [Crossref] [PubMed]
- Morelli L, Tartaglia D, Bronzoni J, et al. Robotic assisted versus pure laparoscopic surgery of the adrenal glands: a case-control study comparing surgical techniques. Langenbecks Arch Surg 2016;401:999-1006. [Crossref] [PubMed]
- Greilsamer T, Nomine-Criqui C, Thy M, et al. Robotic-assisted unilateral adrenalectomy: risk factors for perioperative complications in 303 consecutive patients. Surg Endosc 2019;33:802-10. [Crossref] [PubMed]
- Stefanidis D, Goldfarb M, Kercher KW, et al. SAGES Guidelines for minimally invasive treatment of adrenal pathology. Surg Endosc 2013;27:3960-80. [Crossref] [PubMed]
- Kahramangil B, Berber E. Comparison of posterior retroperitoneal and transabdominal lateral approaches in robotic adrenalectomy: an analysis of 200 cases. Surg Endosc 2018;32:1984-9. [Crossref] [PubMed]
- Economopoulos KP, Mylonas KS, Stamou AA, et al. Laparoscopic versus robotic adrenalectomy: A comprehensive meta-analysis. Int J Surg 2017;38:95-104. [Crossref] [PubMed]
- Tang K, Li H, Xia D, et al. Robot-assisted versus laparoscopic adrenalectomy: a systematic review and meta-analysis. J Laparoendosc Adv Surg Tech A 2015;25:187-95. [Crossref] [PubMed]
- Brandao LF, Autorino R, Laydner H, et al. Robotic versus laparoscopic adrenalectomy: a systematic review and meta-analysis. Eur Urol 2014;65:1154-61. [Crossref] [PubMed]
- Heger P, Probst P, Hüttner FJ, et al. Evaluation of open and minimally invasive adrenalectomy: a systematic review and network meta-analysis. World J Surg 2017;41:2746-57. [Crossref] [PubMed]
- Chai YJ, Kwon H, Yu HW, et al. Systematic review of surgical approaches for adrenal tumors: Lateral transperitoneal versus posterior retroperitoneal and laparoscopic versus robotic adrenalectomy. Int J Endocrinol 2014;2014:918346. [Crossref] [PubMed]
- Agrusa A, Romano G, Navarra G, et al. Innovation in endocrine surgery: robotic versus laparoscopic adrenalectomy. Meta-analysis and systematic literature review. Oncotarget 2017;8:102392-400. [Crossref] [PubMed]
- Feng Z, Feng MP, Feng DP, et al. A cost-conscious approach to robotic adrenalectomy. J Robot Surg 2018;12:607-11. [Crossref] [PubMed]
- Bodner J, Augustin F, Wykypiel H, et al. The da Vinci robotic system for general surgical applications: a critical interim appraisal. Swiss Med Wkly 2005;135:674-8. [PubMed]