Management protocol for primary hyperparathyroidism in a single institution: utility of surgeon performed ultrasound
Introduction
Primary hyperparathyroidism (PHPT) is the third most common endocrine disorder and represents the most common cause of hypercalcemia. This disorder largely afflicts individuals in their 50s and 60s and later. In the recent era, PHPT is more frequently asymptomatic (>75%) being discovered in the work up of incidental findings on routine laboratory work (1). The previous defining adage of “stones, bones, moans and groans”, appeared as the constellation of symptoms seen in PHPT patients prior to the advent of routine serum chemistry testing. The foremost etiology of this process is a single parathyroid adenoma ~80% of the time, four gland hyperplasia ~15%, double adenomas 2–4% and parathyroid carcinoma <1% (2,3).
Surgical excision is the definitive therapy, particularly in symptomatic patients or those meeting criteria by the 2014 PHPT guidelines (4). From the 1990s to currently, the tide of treatment has evolved from the routine bilateral four-gland exploration (FGE) to directed exploration (DE) (5). Pre-operative studies such as Sestamibi (SES), radiology performed ultrasound (R-US), and surgeon performed ultrasound (S-US) as well as intraoperative studies like PTH and ultrasound have been integral in this transition (6). DE is a viable option only if there is an equivalent rate of surgical cure with FGE and if there are accurate pre-operative localization studies and surgical adjuncts.
Multiple studies have shown a comparable curative rate ~95–98% with both techniques (4,7,8). However, in terms of localization studies, radiology performed ultrasound was accurate 70–90% of the time (9,10). As a further subset of ultrasound, surgeon performed studies fared better at about 90% (11,12). Sestamibi scans showed varying sensitivity reported from 54–89%, with the largest study showing 68% sensitivity (7). There is clearly a wide array in the literature in terms of accuracies of these diagnostic studies, but presuppositions of their validity is one of the most important components of allowing the surgeon to perform a minimally invasive procedure with confidence regarding surgical cure.
However, no standard method of work up and localization studies exists currently. There are technical differences based on experience and skill of the operator, particularly for ultrasound, but also on community referral patterns to tertiary centers with potentially limited access to imaging modalities. There is no recommended algorithm, which results in non-standardized, provider- and institution-dependent decision-making.
The purpose of this study was to discover preoperative diagnostic studies, intraoperative techniques, and patient factors most predictive of cure within a single hospital system. The secondary aim was to establish practice guidelines for surgical treatment of PHPT to maximize outcomes based on this hospital system’s performance.
Methods
A retrospective chart review was completed of all surgical parathyroid-related procedures from January 1, 2002 to July 31, 2015 undertaken at a single tertiary referral Veterans Affairs (VA) Hospital. Data was gathered from retrospective chart review after obtaining appropriate IRB approval.
Preoperative lab work including parathyroid hormone (PTH), ionized calcium (iCal), serum calcium and vitamin D levels was gathered, along with localization status of pre-operative imaging techniques of SES, R-US, and S-US.
The majority of patients presented with Sestamibi scan already completed, as this was a tertiary referral center. There was variability in the presence of R-US, and S-US. For R-US, it was performed by high-resolution ultrasonography in the radiology department with instructions to localize abnormal parathyroid glands. The S-US was performed at otolaryngology clinic visit prior to surgery with patient lying supine and neck in extension. Presence or absence of hypoechoic lesion and sidedness was recorded. The senior investigator reviewed all imaging studies.
Intra-operative usage of specific adjunct modalities such as intraoperative PTH (IOPTH), frozen section (FS), and intraoperative ultrasound were recorded as well as associated overall operative time of the procedures and surgical localization determined by operative reports.
Post-operative lab work of PTH, iCal, serum calcium and vitamin D were documented to identify evidence of surgical cure, which is determined as a normal calcium level 6 months after surgery (13).
All statistical analysis was completed in Microsoft Excel (Redmond, WA). ANOVA and Tukey range testing was completed for comparing imaging modality data sets, along with calculation of accuracy, sensitivity and positive predictive value. As this was a tertiary referral center, no true negative values were obtained, limiting the ability to calculate specificity and negative predictive values and, therefore, are not reported herein. Otherwise, mean of data was compared and statistical significance was determined by P value <0.05.
Results
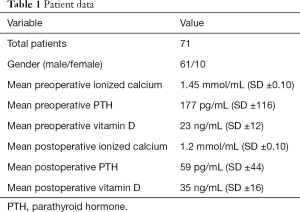
The study includes 71 patients, 61 males and 10 females, whose charts were available and eligible for analysis from the Memphis VA hospital who underwent surgical management of parathyroid disease (Table 1). The mean preoperative PTH was 177±116 pg/mL (range of 56–794 pg/mL, reference range 10–70 pg/mL). Average iCal level was 1.45±0.10 mmol/mL (range, 1.26–1.80 mmol/mL). Mean pre-operative vitamin D levels were 23±12 ng/mL (range, 7.2–50 ng/mL). Post-operative mean levels of iCal fell to 1.2±0.10 mmol/mL, while postoperative mean PTH was 59±44 pg/mL. Mean vitamin D levels postoperatively were 35±16 ng/mL.
Full table
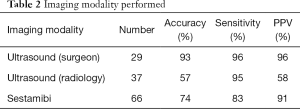
Patients underwent a series of pre-surgical localization studies of SES, S-US, R-US. As there was no agreed upon algorithm, different patients got a different variety of diagnostic studies. As Table 2 shows, the large majority of patients had SES (n=66), followed by R-US (n=37) and S-US (n=29). S-US showed the best accuracy (93%), sensitivity (96%), and positive predictive value (96%). The R-US was found to be least helpful in accurately localizing lesions (57%). The S-US was found to be better than the R-US and SES (ANOVA P<0.0001, Table 3). Additionally, some cases showed discordance between the SES and S-US. In 7/8 cases the S-US was found to be correct in comparison to the operative localization site. In 7/7 cases where there was difference between S-US and R-US, surgeon performed ultrasound was found to be correct.
Full table

Full table
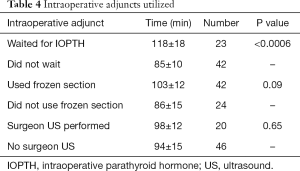
Mean operative time increased while sending off and waiting for IOPTH results versus not waiting, 118±18 vs. 85±10 min, respectively (P<0.0006, Table 4). There was on average a 33-minute increase in operative times with IOPTH. In contrast, cases utilizing FS or intraoperative, pre-incision S-US, did not show an increase in surgical times as compared to those that did not. FS had comparison times 103±12 vs. 86±15 min (P=0.09), while cases involving intraoperative, pre-incision S-US took 98±12 vs. 94±15 min (P=0.65).
Full table
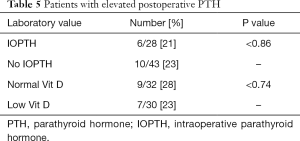
Post operatively, surgical success was measured as normocalcemia 6 months after surgery (13). There was a success rate of 95.8%. Persistent hyperparathyroidism despite a normalized calcium level was seen in 22.5% of our population. In further stratification of this group (Table 5), there was no significant relationship between these persistent hyperparathyroidism patients and either their vitamin D status (P<0.74) or whether or IOPTH (P<0.86, Table 5) was used.
Full table
Discussion
Parathyroid surgery has changed over the decades to a more localization based, minimally invasive surgery with an increasing number of surgical adjuncts. This is evident in our single institution study over the last 15 years at the VA hospital in Memphis, Tennessee.
As this is an analysis of data over an extended period of time, there was an evolution in the diagnostic capabilities of the surgical team as well as the data that was utilized in the pre-surgical work up. S-US became available in 2008. It also, only later, post 2010, was used as an intraoperative, pre-incision adjunct. Additionally, as this is a tertiary care facility, the majority of patients presented with SES already completed. Similarly, IOPTH became available for regular use in 2008 and later.
The majority of our procedures were directed exploration with pre-operative and intraoperative localization leading to equivalent rates of surgical success as compared to FGE in the literature, at greater than 95% biochemical cure. Directed exploration has shown to have equal efficacy, particularly in the setting of adequate adjunctive studies and diagnostic tools (6-8,10).
In examining our population, looking at preoperative and postoperative groups, there was a decline in PTH, iCal and an improvement in vitamin D. 95.8% of population had normalized iCal levels. There were 16/71 or 22.5% of patients that had persistently elevated PTH despite a normal calcium, which was a level that was consistent with the literature, at about ~20% (14). This persistent level was not dependent on whether IOPTH was used or a patient’s associated vitamin D level.
As patients over the course of the study had different degrees of preoperative workup we were able to compare various aspects in terms of their benefit in the overall management of the disease. The pre-operative imaging modality of S-US was more consistently accurate and better at localizing an adenoma than either SES or R-US with significantly better accuracy and positive predictive value. However, SES and R-US were not statistically significant from each other.
Surgeon performed ultrasound, though a more recent endeavor by our group, had a more accurate profile compared to the other two modalities in determining the surgical location of the lesion, particularly in settings where there was discordance between studies. Seven out of eight cases where there was difference between S-US and SES, surgeon ultrasound was correct and similarly it was correct in 7/7 cases where there was disagreement with R-US. There is some consistency to this fact in the literature particularly in reference to R-US, as ultrasound is known to be technician dependent (3,11). When performed directly by the endocrine surgeon with the surgeon’s specialized knowledge of anatomy and operative experience it can be seen how results can be better than a radiologist or technician who may have more expertise in other regions. The question then arises as to the utility of the SES if the S-US is consistently better in identifying operative location. It ends up coming down to its role for lesion verification and assistance with localization of more ectopic lesions. Additionally, as a tertiary referral center, patients frequently presented with this study completed, as it was an available diagnostic study from an outside provider, as opposed to the S-US.
For further evaluation of diagnostic data, the benefit of intraoperative PTH (IOPTH) served as validating evidence coupled with surgical findings and preoperative localization, but at our institution there was a disadvantage in terms of an average increased operative time of 33 minutes that was not present with FS or intraoperative US. There is a noted drawback in operative time with the use of IOPTH, but it clearly has a well-established role in providing supportive evidence (15).
On review of our cases, there was a trend towards having at least two pieces of corroborating data, be it a SES agreeing with a FGE with a dominant nodule or S-US with a directed exploration and IOPTH. In determining an appropriate working algorithm with this idea in mind, the two best pre-operative studies S-US and SES can be used to stratify options, which is displayed in Figure 1. The one arm to focus on is in those situations when there is a concordant S-US and SES scan. Directed exploration can be completed, and the adenoma can be removed with confidence without time spent in the OR waiting for the return of IOPTH. There should very much still be a role for IOPTH where there is disagreement between pre-operative localization studies, but in a system where there is an effective S-US and a congruent SES there is opportunity to forego the study.
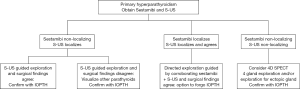
In discussion of the limitations of the project, it is a retrospective study with an assortment of practice patterns that developed over the long time course that were not standardized. As a tertiary referral center, we were unable to determine specificity or negative predictive value of localization studies as there were no true negatives, since referred patients were known positives. Any dialogue about surgeon performed ultrasound needs to take into account operator dependence and that without adequate experience there may be difficulty in replicating accuracy.
Conclusions
As the overall goal of the study was to review pre-operative localization tests and to determine their efficacy, it was shown that surgeon-performed ultrasound was overall more accurate and had better positive predictive value than the sestamibi or radiology performed ultrasound. For a management protocol to be developed, we propose an option to forego intraoperative PTH in the setting in which SES and S-US are concordant with one another and correctly agree with operative findings (Figure 1), as there is a temporal cost to the study as it adds an average of 33 minutes to operating time at our institution.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: Data was gathered from retrospective chart review after obtaining appropriate IRB approval (approval number: 13-02525-XM).
References
- Fraser WD. Hyperparathyroidism. Lancet 2009;374:145-58. [Crossref] [PubMed]
- Randolph GW. Surgery of the Thyroid and Parathyroid Glands. 2nd ed. Saunders, 2013.
- Mohebati A, Shaha AR. Imaging techniques in parathyroid surgery for primary hyperparathyroidism. Am J Otolaryngol 2012;33:457-68. [Crossref] [PubMed]
- Udelsman R, Åkerström G, Biagini C, et al. The surgical management of asymptomatic primary hyperparathyroidism: proceedings of the Fourth International Workshop. J Clin Endocrinol Metab 2014;99:3595-606. [Crossref] [PubMed]
- Greene AB, Butler RS, McIntyre S, et al. National trends in parathyroid surgery from 1998 to 2008: a decade of change. J Am Coll Surg 2009;209:332-43. [Crossref] [PubMed]
- Ryan JA Jr, Eisenberg B, Pado KM, et al. Efficacy of selective unilateral exploration in hyperparathyroidism based on localization tests. Arch Surg 1997;132:886-90; discussion 890-1. [Crossref] [PubMed]
- Siperstein A, Berber E, Barbosa GF, et al. Predicting the success of limited exploration for primary hyperparathyroidism using ultrasound, sestamibi, and intraoperative parathyroid hormone: analysis of 1158 cases. Ann Surg 2008;248:420-8. [PubMed]
- Gracie D, Hussain SS. Use of minimally invasive parathyroidectomy techniques in sporadic primary hyperparathyroidism: systematic review. J Laryngol Otol 2012;126:221-7. [Crossref] [PubMed]
- Berber E, Parikh RT, Ballem N, et al. Factors contributing to negative parathyroid localization: an analysis of 1000 patients. Surgery 2008;144:74-9. [Crossref] [PubMed]
- Terris DJ, Stack BC Jr, Gourin CG. Contemporary parathyroidectomy: exploiting technology. Am J Otolaryngol 2007;28:408-14. [Crossref] [PubMed]
- Van Husen R, Kim LT. Accuracy of surgeon-performed ultrasound in parathyroid localization. World J Surg 2004;28:1122-6. [Crossref] [PubMed]
- Gurney TA, Orloff LA. Otolaryngologist-head and neck surgeon-performed ultrasonography for parathyroid adenoma localization. Laryngoscope 2008;118:243-6. [Crossref] [PubMed]
- Wang TS, Pasieka JL, Carty SE. Techniques of parathyroid exploration at North American endocrine surgery fellowship programs: what the next generation is being taught. Am J Surg 2014;207:527-32. [Crossref] [PubMed]
- Carsello CB, Yen TW, Wang TS. Persistent elevation in serum parathyroid hormone levels in normocalcemic patients after parathyroidectomy: does it matter? Surgery 2012;152:575-81; discussion 581-3. [Crossref] [PubMed]
- Lew JI, Solorzano CC, Montano RE, et al. Role of intraoperative parathormone monitoring during parathyroidectomy in patients with discordant localization studies. Surgery 2008;144:299-306. [Crossref] [PubMed]


