A systematic review of intraoperative process mapping in surgery
Introduction
With an aging population, there is an increasing demand for elective surgery (1). Having an understanding of how the surgical process works and where improvements need to be made can help improve the quality of surgical care, including efficiency. The World Health Organization has advised that quality improvement (QI) strategies can improve healthcare efficiency (2). Process mapping is a QI technique that breaks down a process, or task, into its individual components, or steps, then analyses it (3,4). The goal is to understand the individual steps as a means to identifying potential areas of improvements (3,5). Surgical care is encompassed in the perioperative period which includes three phases: preoperative, intraoperative, and postoperative. The intraoperative period contains the execution of the surgery; it is of interest as it utilises the operating theatres (OT), and a large proportion of a hospital’s total expenses come from here (6).
We recently described our utilisation of these approaches when applied to breast reconstruction (7). We showed that in surgery as complex as autologous breast reconstruction, QI strategies such as process mapping can improve efficiency, and may improve surgical teaching, education and audit. The intraoperative period specifically is an area that can be applied not only to breast reconstruction, but to a much broader range of surgical procedures.
The OT is a complex environment to optimise, with efficiency influenced by factors such as: case scheduling, cancelled cases, on-time starts, procedure time variation (which is influenced by equipment, patient factors, individual surgeon skill, and case complexity), turnover time, administrative factors (i.e., bed management), and the surgical team (i.e., junior staff who are still training) (6,8,9). Understanding what factors contribute to efficiency in the intraoperative period can allow for improvements to be formulated and implemented; process mapping could help to achieve this. However, the benefits of applying process mapping to the intraoperative period in relation to surgical efficiency remained unclear. A recent review of QI technique effectiveness in surgical care found significant improvement in reducing infection rates, complications, delays, length of stay, and increasing antibiotic compliance (10). However, process mapping was not included as one of the QI techniques. This systematic review thus aimed to assess the application of process mapping within the intraoperative period, to determine its benefits in relation to surgical efficiency.
Methods
A systematic review was performed according to Preferred Reporting Items for Systematic Review and Meta-Analyses (PRISMA) principles (11).
Literature search
Search databases consisted of Ovid MEDLINE (1945–present), Allied and Complementary Medicine Database (AMED, 1985–present), Embase (1980–present), and PsychINFO (1806–present). Manual searching and bibliographic linkage was also performed. Medical Subject Heading (MeSH) terms comprised “Process Assessments”, “Total Quality Management”, “Task Performance and Analysis”, and “Time-motion studies”. Keyword search of process mapping and its related QI strategies included each of the terms: process, map, redesign, assessment, “lean principles”, “lean management”, “lean processes”, “Lean Six Sigma”, “Six Sigma”, and “intraoperative period”. Data items extracted were: study size, country, duration, QI strategy/strategies used, main aims, area of application, study design and methodology, outcomes after interventions, relevant statistical analysis, and follow-up period.
Eligibility criteria
An article was included if it had hospital-based surgical patients only, explicitly stated the use of “process mapping” or used a QI methodology that described a process mapping approach, and applied process mapping to the intraoperative period. All included studies were published in peer-reviewed journals and had available abstracts. Reasons for exclusion were: non-English language, animal studies, studies published over 20 years ago, article was a letter, editorial, audit, review or conference abstract; had a non-surgical population; did not either list the process steps or depict the process as a map; or used an existing evidence-based pathway or a predetermined steps list/checklist (i.e., did not map their centre’s process).
Quality assessment of studies
Risk of bias for studies that implemented improvements/interventions was assessed using the “Risk of Bias Assessment Tool for Nonrandomized Studies” (RoBANS) (12), a validated tool that stratifies risk of bias as “high”, “low” or “unclear” according to six domains: selection of participants, confounding variables, measurement of exposure, blinding of outcome assessments, incomplete outcome data, and selective outcome reporting.
Analysis
Principle summary measures comprised of: process mapping approach described, operations and surgical specialities to which process mapping was applied; intraoperative components that were process mapped; areas of improvement identified. For studies that proceeded to apply improvements, the mean operative time and any patient outcomes were evaluated. Thematic analysis was conducted to identify similarities in applications and outcomes across all studies, then displayed in tabular form and discussed. A descriptive synthesis of the summary measures was made, evaluating the quality and biases of the studies. Due to the lack of randomised control trials and diverse range of study aims and outcomes, meta-analysis was not applicable.
Results
Study characteristics
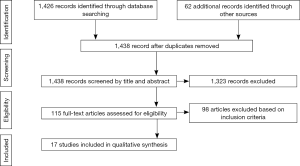
Study selection and citation attrition is demonstrated in Figure 1. Of the 17 studies, eligible for review, 15 did not state the study design explicitly. One study described a prospective longitudinal quasi-experimental study (13), and another study described an observational prospective design (14). All studies were of non-randomised study design; five were purely observational (with no specific improvements/interventions applied) (14-18), one was a controlled before-and-after study (19), and the remainder had before-and-after study designs without controls (7,13,20-28). Table 1 lists more detailed study characteristics.

Full table
Even though the studies named a variety of QI techniques, inspection of their methods revealed that process mapping was used at some stage in the QI process. Four studies named time-motion principles (15,17,18,26); one cited a combination of strategies from Six Sigma, and process and resource analysis (20); two studies specified lean principles (13,22); and three applied Lean Six Sigma principles (21,27,28). One study explicitly stated process mapping as the QI tool (7), and another described a technique similar to process mapping but named it “standardized step-wise technique” (29).
Risk of bias within studies
Nine studies that included real-time process observations were at risk of the Hawthorne effect, an observational bias where study subjects change their behaviour in the knowledge that they are being studied (7,13-15,20,22-24,26). However, only three studies acknowledged this (13,24,28), and only one controlled for this by including an “observer-effect” period, where “fake” observation data collection by research team members was conducted for 3 months at the start of the study (13). Analysis later revealed no differences between baseline database values and values during the “observer-effect” period (13).
Application
The 17 selected studies applied process mapping to achieve a variety of aims, to different surgical specialties, and to different components of the intraoperative period. In doing so, they identified improvement areas which some studies used as the basis to formulate interventions.
The major aims identified were to: identify preventable disruption sources (15); increase OT utilisation (19,20); analyse time spent for each step of a surgical procedure (17,18); increasing intraoperative efficiency (7,13,21,23,27,28); improve educational opportunities (13,14,17,18); redesign intraoperative pathways (24,25); determine limitations of surgical procedure (16); improve costing activities (25); refine surgical practices (26); and determine variance in surgical performance (29). The process mapping approach was applied across a range of surgical specialties, shown in Table 2.
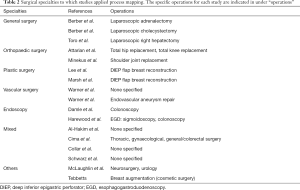
Full table
Intraoperative components
The intraoperative period has several components; each study addressed the processes of some but not all possible components (see Figure 2). Note that while all studies [except Warner et al. (28)] included the operative time, only eight studies applied process mapping to the operation itself (i.e., broke down the surgery into its individual steps) (7,14,16-18,23,24,26).

Process mapping approach
Eight studies used a multidisciplinary team discussion approach to identify the steps in the intraoperative period (13,19-22,24,27,28). This was advantageous as other staff responsibilities (i.e., anaesthetists, nurses and OT technicians) in surgical care could also be included in the process map (19). The process steps were then evaluated by the team and suggestions for improvements were identified. One study reported that this multi-disciplinary team approach allowed a sharing of responsibility and improved communication between surgical team members (24).
In the studies that process mapped the surgical steps (i.e., operative time), steps were identified by individual surgeons (14,17,18,26). Thus the steps pertained to the surgeons themselves, and other surgical staff tasks were not included. Only one study observed the intraoperative pathway in real-time to identify the steps as well as record events that disrupted the natural progression of the patient journey during the operative time (15).
Few studies collected the time taken for each step-in order to generate their process maps, or include in their analyses to aid in the identification of barriers to surgical efficiency (13,15,19,22). In doing so, non-value-added time was able to be assessed. This enabled one study to quantify the length of disruptions within the intraoperative process (15), and another to construct more detailed process maps that included times for each step in the process (22). The remaining studies did not use step timings to aid identification of potential improvements.
Areas of improvement
Numerous areas of improvement within the intraoperative period were identified. Themes that consistently appeared in multiple studies are highlighted in Table 3.
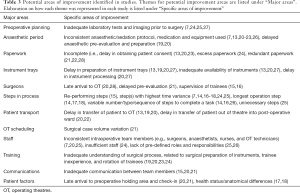
Full table
Improvements implemented
Outcome measures were assessed in ten studies as they implemented interventions based on those identified improvement areas (13,17-22,26-28). The interventions were formulated after discussions with members of the surgical team. Multiple changes to the system were usually implemented at the same time. Some interventions were specific to the department/hospital, however evaluation of the ten studies revealed some common interventions, as discussed below.
Four studies identified the need for parallel processing to occur (13,19-21). Parallel processing is the simultaneous execution of two or more steps in a process (30). One study that implemented this intervention across three surgical specialties found parallel processing significantly improved overall turnover time, allowing more cases per day in the same OT (21). Another also showed this, completing four (occasionally five) total joint replacements in the time needed for three before the intervention (20). Similarly, one study achieved a 30-minute decrease in total intraoperative time (21% decrease, control group vs. optimized process group, t-test, P=0.025), resulting in one additional operation per OT to be added to the surgical list (19). This study was the only controlled before-and-after study thus providing higher quality evidence. Additionally, parallel processing significantly reduced turnover time in one study that found 20.2 minutes saved per case over 1 fiscal year, equated to opportunity costs of approximately $330,000 for one OT used twice a week (13).
Inconsistency in surgical team members was addressed by creating standard operating teams to conduct a particular surgery (18,20). In addition to creating dedicated surgical team members, one study made changes to the OT scheduling and documentation processes, and engaged the surgical registrars in support of the project (20). This increased rate of on-time OT starts from less than 60% to more than 90%, thereby increasing the average number of total joint replacement cases by 29% (20).
Interventions implemented in two of studies included the elimination of redundant steps (21,28); creation of a set communication time when information about the operative procedure status had to be relayed to all OT staff (21,22). the standardisation of operative step sequence, instruments and equipment (7,24). This allowed one study to improve communication and competencies of less experienced staff participating in complex breast reconstruction surgery (24). Some interventions applied to the preoperative period had a knock-on effect on intraoperative time, as seen in two studies (7,21). One employed a standardised preoperative pathway whereby imaging was performed prior to surgery to evaluate the vessels which were to be harvested for a deep inferior epigastric perforator (DIEP) flap breast reconstruction (7). The other study applied a standardised preoperative assessment criteria specific to procedural and patient risk factors to reduce day-of-surgery cancellations, and on-time arrival of the patient in the preoperative holding ward; this resulted in an increase in the rate of on-time arrivals from 52% before the intervention to 81% (P<0.0001) (21).
Risk of bias across studies
The intraoperative processes observed were carried out by more than one surgeon, nurse, anaesthetist, and technician. This was not controlled for except by one study which attempted to do so by assessing the impact of its QI strategy in one surgeon’s OT (13). However, even if the surgeon was consistent, 30 other staff members (nurses, anaesthetists, technicians) were also involved in the OT. All studies with an uncontrolled study design ran the risk of confounding factors. Additionally, there is a lack of consistency in the reporting of QI studies which poses a risk of publication bias that may affect the cumulative evidence.
Discussion
QI studies are vital to increasing surgical efficiency and quality of care. To our knowledge, this is the first systematic review to assess process mapping usage in the intraoperative environment. Process mapping was used in all 17 included studies, albeit with different approaches. Process mapping was used as a tool to identify areas of improvement during the intraoperative period across different surgical specialties, and a variety of improvements that resulted in increased efficiency was applied to that end. These results suggest that process mapping identifies common barriers to surgical efficiency and quality of care. However, the operative steps were not processed mapped in detail, and most of the application of process mapping in the intraoperative period dealt with steps other than the surgery itself. While the published application and outcomes of these process mapping studies have been presented in this report, we did not combine their results as each study had different aims, study designs, and assessment of outcomes. Thus, there were no common outcome measures that could be compared for a cumulative effect estimate.
Majority of studies applied their QI technique to only one type of surgery. Of note, the three studies that applied process mapping to multiple surgical specialties, did not apply it to the surgical component within the intraoperative period. A limitation of the current literature on process mapping is that its application and analysis of the surgical component is usually excluded when investigating surgical efficiency. Studies that include the surgical component mostly do not include the other intraoperative components. Thus, the entire intraoperative process cannot be evaluated thoroughly. However, all studies applying process mapping to surgical process were able to improve pathway performance in some way. This suggests that incorporating process mapping to the entire intraoperative process may enable more improvements.
Most studies identified the intraoperative steps and potential areas of improvement via a multidisciplinary team meeting of staff who engage in the process. This is important as it allows all stakeholders within the intraoperative process to identify how the process flows in relation to an individual’s task (13), and may foster greater interaction between staff members (4). In addition, observation of the intraoperative pathway in real-time should also be carried to fill in any missing steps in the process that might have not occurred to individuals during the team discussion, and allow for a more realistic assessment of the process (4).
The study designs of papers evaluated in this review is similar to the larger body of QI literature where most QI studies have quasi-experimental designs (31). These are either controlled or uncontrolled before-and-after study designs, or a time series design; acknowledging that randomised control trials may not be achievable with characteristics such as randomising and double-blinding being impractical or irrelevant (31). The uncontrolled before-and-after study designs are a limitation to the level of evidence provided. This limitation should be viewed in a gentler light given the difficulty in conducting randomised controlled QI studies. Additionally, controlling for each individual staff member involved in the intraoperative process is ethically and practically difficult. However, perhaps this may be alleviated by designing studies that collect data on potential confounders and employ more vigorous statistical analyses to account for those confounders.
With regards to the Hawthorne effect and how to address it, a systematic review found that it exists in numerous studies, but there is still little understanding of how long the Hawthorne effect lasts, the mechanism of effects, and when and if they occur in a study (32). Thus, it is difficult to control for this bias, but future process mapping studies may want to emulate the study evaluated in this review that used an “observer-effect” period (13), or disregard the first several data points from analysis.
There is a degree of overlap in the approaches of QI strategies, and QI literature lacks explicit and detailed explanations of strategies (33). Our systematic review found that studies did not always use the term “process mapping” but review of the methods reveals that it was used, and some use process mapping as part of more complex QI strategies. This highlights that process mapping, while frequently used in QI studies, is not yet recognised explicitly as a stand-alone QI strategy. The reason for this might be that process mapping is so widely accepted that users do not commonly report its use in the literature, or it is not recognised as a discrete concept. However, the latter seems unlikely, given the National Health Service (NHS) (3) and an article in the British Medical Journal (BMJ) (4) identified process mapping as a concept of its own. The Standards for Quality Improvement Reporting Excellence (SQUIRE) guidelines have been developed to address this inconsistent content and reporting of QI studies (34). but none of the articles that applied process mapping stated its use. More consistent reporting of QI techniques is essential to building up a solid body of evidence to legitimise process mapping as a scientific QI method, and take evidence-based QI to the same level as evidence-based medicine.
Conclusions
Process mapping has thus been clearly identified as a strategy to improve surgical efficiency. It has been used in the intraoperative period but its application to surgical procedure itself remains limited. Process mapping has been used to identify steps that can be modified to improve outcomes, and a range of interventions have been applied to this end. Improved outcomes were achieved largely by decreasing overall duration of surgery. Future research should have more robust study designs and analyses, and focus on using quality improvement reporting guidelines.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Royal Australasian College of Surgeons. Surgical Workforce Projection to 2025 (Australia). 2011. Available online: https://www.surgeons.org/media/437871/rpt_racs_workforce_projection_to_2025.pdf
- WHO. Topic 7: Introduction to quality improvement methods. WHO Patient Safety Curriculum Guide for Medical Schools. 2009. Available online: http://apps.who.int/iris/bitstream/10665/44091/9789241598316_eng.pdf
- NHS. Process Mapping-An Overview. Available online: http://webarchive.nationalarchives.gov.uk/20121108100808/http://www.institute.nhs.uk/quality_and_service_improvement_tools/quality_and_service_improvement_tools/process_mapping_-_an_overview.html
- Trebble TM, Hansi N, Hydes T, et al. Process mapping the patient journey: an introduction. BMJ 2010;341:c4078. [Crossref] [PubMed]
- McLaughlin N, Rodstein J, Burke MA, et al. Demystifying process mapping: a key step in neurosurgical quality improvement initiatives. Neurosurgery 2014;75:99-109. [Crossref] [PubMed]
- Soliman BA, Stanton R, Sowter S, et al. Improving operating theatre efficiency: an intervention to significantly reduce changeover time. ANZ J Surg 2013;83:545-8. [Crossref] [PubMed]
- Marsh D, Patel NG, Rozen WM, et al. Three routine free flaps per day in a single operating theatre: principles of a process mapping approach to improving surgical efficiency. Gland Surg 2016;5:107-14. [PubMed]
- New South Wales Agency for Clinical Innovation. Operating Theatre Efficiency Guideline: A guide to the efficient management of operating theatres in New South Wales hospitals. NSW: Commonwealth of Australia 2014. Available online: http://www.aci.health.nsw.gov.au/resources/surgical-services/efficiency/theatre-efficiency
- Kimbrough CW, McMasters KM, Canary J, et al. Improved Operating Room Efficiency via Constraint Management: Experience of a Tertiary-Care Academic Medical Center. J Am Coll Surg 2015;221:154-62. [Crossref] [PubMed]
- Nicolay CR, Purkayastha S, Greenhalgh A, et al. Systematic review of the application of quality improvement methodologies from the manufacturing industry to surgical healthcare. Br J Surg 2012;99:324-35. [Crossref] [PubMed]
- Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 2009;6:e1000097. [Crossref] [PubMed]
- Kim SY, Park JE, Lee YJ, et al. Testing a tool for assessing the risk of bias for nonrandomized studies showed moderate reliability and promising validity. J Clin Epidemiol 2013;66:408-14. [Crossref] [PubMed]
- Collar RM, Shuman AG, Feiner S, et al. Lean management in academic surgery. J Am Coll Surg 2012;214:928-36. [Crossref] [PubMed]
- Toro JP, Patel AD, Lytle NW, et al. Detecting performance variance in complex surgical procedures: analysis of a step-wise technique for laparoscopic right hepatectomy. Am J Surg 2015;209:418-23. [Crossref] [PubMed]
- Al-Hakim L, Gong XY. On the day of surgery: How long does preventable disruption prolong the patient journey? Int J Health Care Qual Assur 2012;25:322-42. [Crossref] [PubMed]
- Minekus JP, Valstar ER, Rozing PM, et al. Factors influencing the surgical process during shoulder joint replacement: time-action analysis of five different prostheses and three different approaches. Med Sci Monit 2005;11:CR14-20. [PubMed]
- Berber E, Duh QY, Clark OH, et al. A critical analysis of intraoperative time utilization in laparoscopic adrenalectomy. Surg Endosc 2002;16:258-62. [Crossref] [PubMed]
- Berber E, Engle KL, Garland A, et al. A critical analysis of intraoperative time utilization in laparoscopic cholecystectomy. Surg Endosc 2001;15:161-5. [Crossref] [PubMed]
- Schwarz P, Pannes KD, Nathan M, et al. Lean processes for optimizing OR capacity utilization: prospective analysis before and after implementation of value stream mapping (VSM). Langenbecks Arch Surg 2011;396:1047-53. [Crossref] [PubMed]
- Attarian DE, Wahl JE, Wellman SS, et al. Developing a high-efficiency operating room for total joint arthroplasty in an academic setting. Clin Orthop Relat Res 2013;471:1832-6. [Crossref] [PubMed]
- Cima RR, Brown MJ, Hebl JR, et al. Use of lean and six sigma methodology to improve operating room efficiency in a high-volume tertiary-care academic medical center. J Am Coll Surg 2011;213:83-92; discussion 93-4. [Crossref] [PubMed]
- Damle A, Andrew N, Kaur S, et al. Elimination of waste: creation of a successful Lean colonoscopy program at an academic medical center. Surg Endosc 2016;30:3071-6. [Crossref] [PubMed]
- Harewood GC, Chrysostomou K, Himy N, et al. A "time-and-motion" study of endoscopic practice: strategies to enhance efficiency. Gastrointest Endosc 2008;68:1043-50. [Crossref] [PubMed]
- Lee BT, Tobias AM, Yueh JH, et al. Design and impact of an intraoperative pathway: a new operating room model for team-based practice. J Am Coll Surg 2008;207:865-73. [Crossref] [PubMed]
- McLaughlin N, Burke MA, Setlur NP, et al. Time-driven activity-based costing: a driver for provider engagement in costing activities and redesign initiatives. Neurosurg Focus 2014;37:E3. [Crossref] [PubMed]
- Tebbetts JB. Achieving a predictable 24-hour return to normal activities after breast augmentation: part I. Refining practices by using motion and time study principles. Plast Reconstr Surg 2002;109:273-90; discussion 291-2. [Crossref] [PubMed]
- Warner CJ, Horvath AJ, Powell RJ, et al. Endovascular aneurysm repair delivery redesign leads to quality improvement and cost reduction. J Vasc Surg 2015;62:285-9. [Crossref] [PubMed]
- Warner CJ, Walsh DB, Horvath AJ, et al. Lean principles optimize on-time vascular surgery operating room starts and decrease resident work hours. J Vasc Surg 2013;58:1417-22. [Crossref] [PubMed]
- Toro JP, Lytle N, Patel A, et al. Detecting performance variance in complex surgical procedures: Analysis of a step-wise technique for laparoscopic right hepatectomy. Am J Surg 2015;209:418-23. [Crossref] [PubMed]
- Malangoni MA. Assessing operating room efficiency and parallel processing. Ann Surg 2006;243:15-6. [Crossref] [PubMed]
- Portela MC, Pronovost PJ, Woodcock T, et al. How to study improvement interventions: a brief overview of possible study types. BMJ Qual Saf 2015;24:325-36. [Crossref] [PubMed]
- McCambridge J, Witton J, Elbourne DR. Systematic review of the Hawthorne effect: New concepts are needed to study research participation effects. J Clin Epidemiol 2014;67:267-77. [Crossref] [PubMed]
- Jones E, Lees N, Martin G, et al. Describing methods and interventions: a protocol for the systematic analysis of the perioperative quality improvement literature. Syst Rev 2014;3:98. [Crossref] [PubMed]
- Ogrinc G, Mooney SE, Estrada C, et al. The SQUIRE (Standards for QUality Improvement Reporting Excellence) guidelines for quality improvement reporting: explanation and elaboration. Qual Saf Health Care 2008;17 Suppl 1:i13-32. [Crossref] [PubMed]