The role and timing of parathyroid hormone determination after total thyroidectomy
Introduction
Postoperative hypocalcemia is the most common complication after total thyroidectomy. It occurs as a result of unintentional resection, manipulation, or devascularization of the parathyroid glands during thyroidectomy leading to hypoparathyroidism. Clinical manifestations are primarily neurologic and cardiac in nature, and include perioral and acral paresthesias, muscle cramps, tetany, seizures, prolonged QT syndrome, and congestive heart failure. Postoperative hypocalcemia lasting up to 6 months is considered transient and has been reported to occur in 3–53% of patients. The wide variability in reported rates of hypocalcemia secondary to transient hypoparathyroidism is in part due to differing definitions in the literature of hypocalcemia based on biochemical evaluation, patient symptoms, or both. However, in up to 2% of patients, hypoparathyroidism may last longer than 6 months and is considered permanent (1-11).
Multiple studies have investigated potential predictive factors to better determine which patients are at higher risk for postoperative hypocalcemia. In the majority of studies, demographic factors including age, sex, and other comorbidities play a minimal role (6,7,12-15). Results regarding preoperative factors including 25-OH vitamin D, serum calcium, or parathyroid hormone (PTH) levels, or indications for surgery such as Graves’ disease have been variable with no clear association to the development of postoperative hypocalcemia. Potential intraoperative factors that have been investigated include performance of a central compartment neck dissection as it is associated with increased parathyroid manipulation, as well as potential devascularization of the inferior parathyroid glands requiring autotransplantation; again, results have been mixed (5,16).
Postoperatively, numerous studies have demonstrated that measurement of serum PTH levels can be utilized as a predictor of the development of post-thyroidectomy hypocalcemia. However, there has not been consensus on the most appropriate timing or threshold value for PTH measurement that would optimally guide the management of postoperative hypocalcemia, as there is also not universal consensus as to the routine or selective use of calcium and/or calcitriol supplementation. This review article will focus on the role and timing of PTH measurement in the perioperative period to guide treatment of postoperative hypocalcemia for patients undergoing total thyroidectomy.
Routine versus selective supplementation
Given the ease and low cost of calcium supplements, many have advocated for routine supplementation as the optimal method to prevent postoperative hypocalcemia. Bellantone et al. (17) compared administration of oral calcium alone, oral calcium and vitamin D supplementation, or no supplementation in a study of 79 patients that underwent total thyroidectomy. They defined hypocalcemia as serum calcium <8.0 mg/dL. Compared to no supplementation or calcium alone, the group that received both calcium and vitamin D had a significantly higher serum calcium level on postoperative days 2 and 3 (P<0.05), but there was no difference in the calcium levels among the three groups at 1 week postoperatively. The groups that received calcium alone or calcium with vitamin D had a significant reduction in symptomatic hypocalcemia, compared to the group without routine supplementation (12% vs. 41%, P=0.005). Another study by Roh et al. (18) had a similar study design and compared no supplementation to routine supplementation with oral calcium and vitamin D in a series of 90 patients. Rates of biochemical (serum total calcium <8.0 mg/dL or ionized calcium <1.0 mmol/L) and symptomatic hypocalcemia were significantly lower in the supplementation group (13% vs. 36%, P=0.014; and 7% vs. 24%, P=0.02, respectively). In both studies, addition of intravenous (IV) calcium for persistent symptoms was only needed in the group without routine supplementation, suggesting that routine supplementation could prevent the need for IV calcium and ameliorate postoperative hypocalcemia. However, despite routine supplementation, 12% of patients in the first study and 7% in the second developed symptomatic hypocalcemia, demonstrating that patients should still be counseled regarding the symptoms of hypocalcemia so they can be evaluated and treated promptly should symptoms arise.
There may be certain situations in which routine supplementation is appropriate. Roh et al. (16) examined the role of routine supplementation in patients undergoing total thyroidectomy with central compartment neck dissection, given the higher theoretical risk of devascularization of the inferior glands during this procedure. The authors compared groups who received oral calcium alone (N=49), oral calcium and vitamin D (N=49), or no supplementation (N=50) for the first two weeks postoperatively; a control group of 49 patients who underwent total thyroidectomy without central compartment neck dissection and who did not receive supplementation was included. In comparing groups who did not receive supplementation, the rate of biochemical hypocalcemia (<8.0 mg/dL) was overall higher in patients with central compartment neck dissection compared to total thyroidectomy alone (44% vs. 14%, P=0.002). Routine supplementation with calcium and vitamin D reduced this rate to 8%. It also completely eliminated the need for IV calcium for persistent symptomatic hypocalcemia, which was required for 14% of the group who underwent a central compartment neck dissection and that did not receive any supplementation. Overall, the rate of permanent hypocalcemia was 2.5%, and was similar in all four groups. The authors concluded that the addition of a central compartment neck dissection to total thyroidectomy significantly increased the risk of hypocalcemia and therefore, this group of patients should receive routine supplementation with calcium and vitamin D.
From a cost perspective, a recent analysis (19) demonstrated a reduction in cost per quality-adjusted life-years (QALYs) using a Markov decision model on a hypothetical cohort of patients. Patients were divided into routine supplementation or selective supplementation groups. All patients in the routine supplementation group received a standardized regimen of calcium 1 g 3 times a day and calcitriol 0.5 mcg every 12 hours for 1 week. The selective supplementation group received calcium/calcitriol per the Australian Endocrine Surgeons guidelines (20). For PTH >20 pg/mL, no supplementation was given. For PTH 3–20 pg/mL, calcium carbonate 1,200 mg was given twice daily, and for a PTH ≤3 pg/mL calcium carbonate 1,200 mg and calcitriol 0.5 mcg were each given twice daily. Routine supplementation was associated with a savings of $62 per patient, and a small increase in QALYs. The model was very sensitive to hypocalcemia, which was a factor that varied by surgeon. The effect on quality of life at the individual level was also not well-characterized. The authors concluded that prospective studies are needed to better determine the cost/benefit of routine supplementation on both the individual and societal level.
Although the theoretical cost may be lower and the risk of hypocalcemia despite supplementation is low, this must be balanced against the risks of over supplementation. Patients receiving excessive amounts of calcium unnecessarily can develop hypercalcemia (21), which can increase the risk of developing renal calculi, constipation, and altered absorption of iron and zinc. There is also evidence that parathyroid inhibition can occur as a result of prolonged exposure to calcitriol. These factors may necessitate additional phlebotomies or clinic follow up in the postoperative period which can increase cost and discomfort for the patient (5). All of these factors have led to the examination of the use of selective supplementation following thyroidectomy based on postoperative serum calcium and/or PTH levels.
Defining hypocalcemia and the hypoparathyroid state
Predictive factors for hypocalcemia after thyroidectomy have been extensively investigated, but the definition of hypocalcemia has not been consistent across studies. Many studies on the topic of postoperative hypocalcemia have commented on the presence of symptoms and stratified hypocalcemia as “symptomatic” or “biochemical” if it was only based on laboratory values. The threshold calcium level also varied from 7.2 to 8.5 mg/dL depending on the study. Others have used more complicated algorithms to classify hypocalcemia by combining different thresholds with or without symptoms. As an example, one study (22) defined hypocalcemia as anyone with total calcium less than 7.2 or 7.2–8.6 mg/dL and symptomatic. These definitions were used in many studies to guide calcium supplementation, while others based a supplementation schedule on the postoperative PTH level. Still others only counted patients with symptomatic hypocalcemia as they felt this was most clinically significant. These factors explain why there is such great variability in the incidence of postoperative hypocalcemia across different studies.
Both serum calcium and PTH levels in the perioperative period have been studied to determine their ability to predict postoperative hypocalcemia, with PTH being favored for several reasons. The nadir of serum calcium levels occurs 48–72 hours postoperatively and serum calcium levels obtained on postoperative day 1 (POD1) have been associated with a positive predictive value as low as 4% for determining persistent hypocalcemia. Given this fact, which is in part because the nadir of serum calcium typically occurs after patients have been discharged, there has been a need for an earlier predictor in order to facilitate postoperative management and prevent potential readmissions. In patients who develop hypoparathyroidism, PTH levels decline more rapidly than serum calcium, with significant changes noted within the first 3 hours (23). Postoperative PTH levels have become a more useful predictor of hypocalcemia following total thyroidectomy, as it is associated with the highest accuracy, particularly when compared to measuring postoperative calcium levels. The studies discussed below will outline the utility and timing of postoperative PTH measurements in determining the need for calcium and/or calcitriol supplementation after thyroidectomy (Table 1).
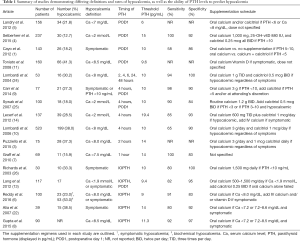
Full table
Timing of PTH measurement to guide selective supplementation
POD1
Multiple studies have examined the role of a single measurement of serum PTH level on POD1. Landry et al. (5) retrospectively reviewed 156 patients who underwent total or completion thyroidectomy and stratified patients into groups based on their risk of developing hypocalcemia. The high-risk group included patients with symptoms of hypocalcemia, and serum PTH <3 pg/mL and/or serum calcium <7 mg/dL on POD1. The low-risk group included all other patients. PTH and calcium levels were checked at 2 hours postoperatively, on the day of surgery at 5 PM, and on POD1 at 5 AM. The high-risk group consisted of 34 (22%) patients, 25 (74%) of whom became symptomatic. The high-risk group was more likely to be women (91% vs. 74%, P=0.04), to have malignant disease (88% vs. 65%, P=0.01), to have a concomitant central compartment neck dissection (73% vs. 43%, P=0.001), or an incidental parathyroidectomy (29% vs. 11%, P=0.013). On further analysis of the high-risk group, the authors found that all symptomatic patients had a PTH value <6 pg/mL and/or a calcium <8 mg/dL. By expanding criteria to capture all symptomatic patients, an additional 32 (26%) patients from the 122 patients in the low-risk group would receive supplementation based on a PTH of 3–6 pg/mL or calcium of 7–8 mg/dL. This left 90 patients in the low-risk group without supplementation, which made up 58% of the total cohort. While this supplementation schedule meant unnecessary supplementation for some patients who would not have become symptomatic, the authors concluded that it could lead to safe early discharge and reduction in postoperative need for medication, follow-up assessments, and phlebotomies for a substantial proportion of patients. They therefore recommended calcium and/or calcitriol supplementation for all patients with PTH <6 pg/mL or calcium <8 mg/dL on the morning after total thyroidectomy.
Selberherr et al. (4) prospectively stratified 237 patients who underwent total thyroidectomy without lymphadenectomy by POD1 PTH values and classified them as having “normal” parathyroid function (PTH >15 pg/mL), “disturbed” function (PTH <10 pg/mL), or “uncertain” function (PTH 10–15 pg/mL). They studied the rate of hypocalcemia, which they defined as serum calcium <2 mmol/L, and the effectiveness of each threshold value of predicting hypocalcemia. The standard supplementation regimen included oral calcium, 25-OH vitamin D, and calcitriol as outlined in the table (Table 1). Threshold PTH values of 10 and 15 pg/mL had a sensitivity of 83% and 100% and specificity of 99% and 92%, respectively. These data suggested that patients with PTH >15 pg/mL were safe for early discharge without supplementation, those with PTH <10 pg/mL were at highest risk and should receive supplementation with calcium, 25-OH vitamin D, and calcitriol, and those with PTH in the 10–15 pg/mL range warrant close observation.
In a prospective, randomized study of 143 patients undergoing total or completion thyroidectomy, Cayo et al. (1) evaluated predictive factors for hypocalcemia and hypoparathyroidism in an effort to appropriately tailor postoperative calcium and vitamin D supplementation for the patients at highest risk of postoperative hypocalcemia. Serum calcium and PTH levels were obtained on the evening of surgery and in the morning of POD1. Patients were stratified by the POD1 PTH level, which was used as a guide for supplementation. In the highest risk group with PTH <5 pg/mL, patients were randomized to supplementation with calcium alone or calcium and calcitriol. With a PTH between 5–10 pg/mL, patients received either calcium alone or no supplementation. No supplements were given to any patient with PTH >10 pg/mL. Calcium supplementation consisted of calcium carbonate 2,500 mg 3 times daily, or calcitriol 0.25–0.50 mcg twice daily. Follow-up serum calcium and PTH levels were obtained at the first postoperative visit. Of the 143 patients, 31 patients (22%) had PTH <10 pg/mL, and 15 of them (48%) developed symptoms of hypocalcemia. Ten of these patients required additional calcitriol or oral calcium, and 5 required prolonged admission for IV calcium. In contrast, only 11 of 112 (10%) patients with PTH >10 pg/mL were symptomatic, none required scheduled supplemental calcium, and PTH levels remained >10 pg/mL in all patients at the time of the postoperative visit (Figure 1). The authors concluded that a POD1 PTH <10 pg/mL had a sensitivity of 86% and negative predictive value of 90% for symptomatic hypocalcemia. On multivariate analysis, younger age and lower postoperative PTH levels were associated with hypocalcemic symptoms. No statistically significant predictors were found for postoperative PTH <10 pg/mL, including performance of a central compartment neck dissection, although the removal and/or autotransplantation of parathyroid glands had a trend toward being predictive (Table 2). The authors acknowledged that sample size was a limitation in finding predictive factors. This limitation notwithstanding, they concluded that PTH <10 pg/mL on the first postoperative day is highly predictive of patients at highest risk for developing symptomatic hypocalcemia after total thyroidectomy and these patients should receive supplementation. In contrast, patients with PTH >10 pg/mL are at low risk for postoperative hypocalcemia and can be discharged safely without supplementation and without the need for laboratory assays in the absence of symptoms.
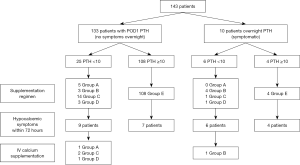
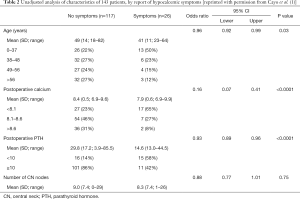
Full table
Taken together, these studies demonstrate that a PTH measurement on the first postoperative day is an effective determinant of postoperative hypocalcemia and the need for supplementation, although limitations of this approach include a potential delay in treatment and subsequent discharge by 1 day.
Immediate postoperative PTH determination
In an effort to reduce costs and target earlier discharge from the hospital without compromising safety, multiple studies have assessed whether a PTH level drawn in the immediate postoperative period can accurately predict hypocalcemia with the goal of earlier discharge in low-risk patients. The Australian Endocrine Surgeons published guidelines for postoperative PTH measurement based on four studies which combined data from 458 patients (20). They recommended that all patients have PTH measured 4 hours postoperatively. The included studies differed in the laboratory assays used for PTH level determination and so the authors defined normal levels based on the standard limits of each assay (>10, >12, >6 pg/mL, or >1.0 pmol/L). Undetectable levels were any value below the lower limit of detection (<3 pg/mL or <0.1 pmol/L), and intermediate levels were any value between the normal and undetectable ranges. The authors made the following recommendations: normal postoperative PTH levels are accurate in predicting normocalcemia and allow for early discharge without supplementation, undetectable PTH levels warrant early initiation of combination therapy with oral calcium and calcitriol and weekly monitoring of laboratory values, and patients with intermediate levels should receive calcium supplementation and should be monitored for hypocalcemia as they may require addition of calcitriol as well. These guidelines are limited by significant variability amongst the four included studies, particularly with respect to calcium supplementation regimens and resultant rates of hypocalcemia.
Several studies have compared the accuracy of PTH levels drawn within the first 6 hours and on POD1. In a prospective cohort study of 100 patients who underwent total thyroidectomy, Sywak et al. (25) evaluated PTH levels at 4 and 23 hours after surgery to determine if the shorter time interval would alter the accuracy with which PTH determination could predict hypocalcemia. Using a threshold of 10 pg/mL, the sensitivity and specificity of the 4- and 23-hour interval (90% vs. 86% and 84% vs. 83%, respectively) were similar. The mean PTH levels were also similar between groups at the 2 time points (22.3 pg/mL at 4 hours and 23.2 pg/mL at 23 hours). The authors advocated for routine supplementation with calcium 1,200 mg twice daily for all patients regardless of postoperative PTH level; in addition, calcitriol 0.5 mcg twice daily was added and hospital stay was extended to 2 days for any patient with PTH <3 pg/mL. For patients with PTH between 3 and 10 pg/mL, calcitriol was added if the serum calcium level dropped below 2.0 mmol/L. This study was limited by a lack of any post-discharge follow up data on rates of hypocalcemia and ongoing need for supplementation. Nonetheless, the authors concluded that postoperative PTH levels drawn at 4 and 23 hours after surgery were equivalent, and the earlier time point could lead to earlier initiation of supplementation and hospital discharge.
Lombardi et al. (24) used serial measurements of calcium and PTH levels to determine the most accurate time point after thyroidectomy. They collected samples preoperatively, at skin closure, 2, 4, 6, 24, and 48 hours after surgery in 53 patients after total or completion thyroidectomy. Hypocalcemia, which they defined as serum calcium <8.0 mg/dL, was noted in 16 (30%) patients, all of whom received supplementation. A standard supplementation schedule of oral calcium 1 g 3 times daily and calcitriol 0.5 mcg twice daily was given to any hypocalcemic patient regardless of symptoms. PTH levels were in the normal range for all normocalcemic patients. The 4- and 6-hour time points were most predictive of developing hypocalcemia (sensitivity 94%, specificity 100%) for a PTH level of <10 pg/mL. The authors concluded that a single PTH measurement at 4 hours was the most useful as it was most accurate in predicting hypocalcemia while allowing early initiation of therapy if needed or early discharge in patients at low risk.
To validate the 4-hour time point, the same authors followed their original study with a large prospective study (10). They evaluated 523 patients after thyroidectomy and found that 199 patients (38%) developed postoperative hypocalcemia (defined as a serum calcium <8.0 mg/dL). They used the same PTH threshold of 10 pg/mL as this provided the highest accuracy for predicting hypocalcemia in their previous study; however, for this cohort, the sensitivity (65%), specificity (90%), and accuracy (80%) were all considerably lower than their previous study. Of the 199 hypocalcemic patients, 70 (35%) had a normal PTH level. For the entire cohort, this coincided with a false-negative rate of 13.4% of patients who were hypocalcemic with a normal PTH level. The authors concluded that this false-negative rate was too high to use a 4-hour PTH level of >10 pg/mL as a justification for early discharge. They therefore recommended serial calcium monitoring and routine supplementation.
A study by Graff et al. (7) similarly investigated the role for an early postoperative 1-hour PTH level in the development of hypocalcemia (defined as <7.5 mg/dL) in 69 patients. With a threshold PTH value of 14 pg/mL, the sensitivity was 100% and specificity was 83% of predicting hypocalcemia in this cohort of patients. The addition of a serum calcium level at 6 hours improved the specificity to 88% without changing the sensitivity. They therefore concluded that patients with an immediate postoperative PTH level >14 pg/mL and serum calcium >8.0 mg/dL at 6 hours were safe for early discharge without supplementation. If these conditions were not met, the authors recommended additional calcium monitoring and inpatient management.
Carr et al. (9) compared the predictive value of PTH levels on POD1 and at 4 hours in 77 patients after total or completion thyroidectomy as a follow up to their previous study (1). PTH levels were drawn at 4 hours and on POD1 and all patients with PTH <10 pg/mL on POD1 were discharged with supplementation (Figure 2A). Of the 77 patients, 21 (27%) received supplementation; 18 had PTH <10 pg/mL at both time points and 9 of them (50%) reported having symptoms. Of the remaining 3 patients, 2 had PTH >10 pg/mL at both time points, but received supplementation because of the development of symptoms in 1 patient and a low serum calcium (7.3 mg/dL) in the other. The remaining patient had an initial PTH <10 pg/mL and POD1 PTH >10 pg/mL, but did develop symptoms. Of note, two patients with a 4-hour PTH 5–10 pg/mL had a POD1 PTH ≥10 pg/mL and were not discharged on supplementation; these patients were asymptomatic and did not require supplementation postoperatively. Comparing the 4-hour data to the POD1 data, the sensitivity (98% vs. 98%), specificity (90% vs. 86%), positive predictive value (96% vs. 95%), and negative predictive value (95% vs. 95%) were similar in their accuracy of predicting hypocalcemia (Table 3). Based on these data, the authors developed a protocol for calcium and calcitriol supplementation after completion/total thyroidectomy; patients with a 4-hour PTH >10 pg/mL do not receive calcium supplementation. Patients with a PTH <10 pg/mL are started on calcium carbonate; calcitriol is added when the PTH is <5 pg/mL and/or at the surgeon’s discretion (Figure 2B).
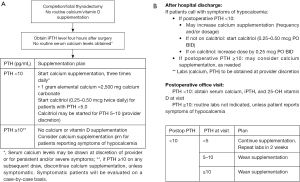
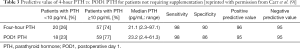
Full table
Intraoperative PTH (IOPTH) measurement
With the increasing use of IOPTH monitoring in parathyroid surgery, several investigators have evaluated its role in predicting postoperative hypocalcemia after thyroid surgery. This was first examined by Richards et al. (26) with a prospective evaluation of 30 patients who had a total thyroidectomy. IOPTH levels were obtained, and postoperative serum calcium levels were drawn at 6 hours and again on POD1. Overall, 10 (33%) patients developed symptomatic hypocalcemia and 5 required IV calcium for tetany; this included 8 patients who had IOPTH levels <10 pg/mL. A threshold of 10 pg/mL had a sensitivity and specificity of 80% and 100%, respectively, for the development of symptomatic hypocalcemia. The authors concluded that IOPTH can be used to accurately predict postoperative hypocalcemia.
Other studies followed validating these results. Lang et al. (13) prospectively evaluated 117 patients that underwent total or completion thyroidectomy. Using a cutoff of 1.0 pmol/L (equal to 9.4 pg/mL), an IOPTH level obtained 5–10 minutes after skin closure was 82% sensitive and 95% specific for developing symptomatic or biochemical hypocalcemia (defined as Ca <1.9 mmol/L or 7.6 mg/dL). Patients with either symptomatic hypocalcemia or serum calcium level <1.9 mmol/L were supplemented with 500–1,500 mg oral calcium daily with the addition of calcitriol 0.25 mcg twice daily for persistent hypocalcemia. With an IOPTH threshold of 9 pg/mL, another prospective study (6) of 100 patients demonstrated similar results. IOPTH levels were drawn 20 minutes after thyroidectomy with the patient still anesthetized. A routine supplementation schedule was given for biochemical hypocalcemia (defined as <8 mg/dL) consisting of oral calcium. With the development of hypocalcemia symptoms, patients received IV calcium gluconate and/or vitamin D in addition to oral calcium. IOPTH was 91% sensitive and 83% specific for predicting symptomatic hypocalcemia. There were only 2 of 64 (3%) patients with PTH >9 pg/mL that became symptomatic and required supplemental calcium. Both were treated successfully as outpatients and neither required addition of calcitriol. Both of these studies concluded that IOPTH was accurate in predicting hypocalcemia with an acceptably low false negative rate, and it can be used to facilitate same-day discharge in patients with IOPTH levels above the threshold. In patients with IOPTH below the threshold level, serial calcium monitoring and an additional inpatient hospital day are warranted for closer monitoring.
Relative decline in perioperative PTH value
While a single PTH measurement postoperatively has been demonstrated to be an accurate predictor of postoperative hypocalcemia at multiple different time points, a PTH decline perioperatively has also been described as a method to predict postoperative hypocalcemia. A systematic review published in 2007 (27) included nine studies with a total of 457 patients that documented the PTH decrease from a level drawn preoperatively to a level drawn at various time points within the first 6 hours after surgery. They evaluated the data in 3 different groups, IOPTH, PTH level drawn at 1–2 hours, and PTH level at 6 hours, and assessed the accuracy with which these different levels could predict hypocalcemia. The definition of hypocalcemia differed for each study and varied from <7.2 to <8.4 mg/dL with or without symptoms. The authors found that the greatest accuracy was determined using the trend to 6 hours, followed by 1–2 hours, and finally intraoperatively with receiver operating characteristic (ROC) area under the curve (AUC) measurements. For this study, the AUCs were 0.97 for the 6-hour group, 0.94 for the 1–2-hour group, and 0.86 for the intraoperative group, with significant differences between the intraoperative group and each of the postoperative groups (P=0.021 for the 1–2-hour group and P=0.002 for the 6-hour group), but not between the two postoperative groups. The authors concluded that all 3 time trends offer an accurate predictor of postoperative hypocalcemia, with the 1–2- and 6-hour determination being the most predictive. Moreover, a greater percent decrease in PTH level correlated with a higher positive predictive value. Based on these data, the authors recommended obtaining preoperative as well as postoperative PTH levels 1–6 hours after thyroidectomy in order to determine a PTH trend, which can be used to guide early calcium supplementation for a larger decline in PTH levels (>90%) and safe early discharge for smaller declines (<50%). The patients that have a decline in the 50–90% range warrant additional calcium monitoring.
A prospective study (3) evaluated the relative decline of PTH 2 hours post-thyroidectomy in a cohort of 75 patients. The relative decline was 72% for hypocalcemic (defined as PTH <8 mg/dL) patients and 44% for patients who remained normocalcemic. A cutoff of 62% decline had the highest accuracy with 100% sensitivity and specificity. The authors recommended oral calcium supplementation for any hypocalcemic patient, or for a patient who is normocalcemic on POD1 with a relative decline in PTH level >62% as this group was at high risk for progression to hypocalcemia. This group did not need additional inpatient monitoring and was safely discharged home on POD1.
Several studies compared the trend in PTH levels to a single intraoperative or postoperative value and demonstrated similar accuracy between the two groups. A study (8) of 90 patients undergoing thyroidectomy with preoperative and IOPTH measurement compared the sensitivity and specificity of a single IOPTH value of 11.3 pg/mL, a PTH decline of 75%, and a combination of the two. Though all tests had high accuracy, the addition of PTH decline to a single IOPTH level did not improve the accuracy of predicting hypocalcemia with a sensitivity of 92% and specificity of 97% for both tests. A similar study of 137 patients (14) used an interval of 4 hours and compared a single 4-hour value to the trend at 4 hours. Both the single value and the trend were highly sensitive and specific for predicting hypocalcemia with a PTH threshold of 19.4 pg/mL and a PTH decline of 69%. The relative decline had a trend toward improved accuracy but this did not reach statistical significance. A prospective study of 160 patients extended the trend until the first postoperative day (11). The accuracy of both the POD1 PTH with threshold 9.6 pg/mL and a negative trend of 69% was similar, and the combination of the two values again did not improve accuracy. Taken together, these studies demonstrate similar accuracy for a single PTH level and a trend of preoperative to postoperative PTH values. This suggests that the additional laboratory values only increase cost and discomfort to the patient without any perceived benefit and may therefore be unnecessary.
Summary
PTH assessment is an accurate predictor of developing postoperative hypocalcemia after total thyroidectomy. Given significant variation in study designs, the optimal timing of this measurement cannot be determined. However, obtaining PTH measurement within 4 hours of surgery is an accurate predictor for the development of hypocalcemia and can guide selective calcium and calcitriol supplementation in high-risk individuals while allowing for the possibility of same-day discharge in low-risk individuals without the need for supplementation or further monitoring in the absence of symptoms.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Cayo AK, Yen TW, Misustin SM, et al. Predicting the need for calcium and calcitriol supplementation after total thyroidectomy: results of a prospective, randomized study. Surgery 2012;152:1059-67. [Crossref] [PubMed]
- McLeod IK, Arciero C, Noordzij JP, et al. The use of rapid parathyroid hormone assay in predicting postoperative hypocalcemia after total or completion thyroidectomy. Thyroid 2006;16:259-65. [Crossref] [PubMed]
- Puzziello A, Gervasi R, Orlando G, et al. Hypocalcaemia after total thyroidectomy: could intact parathyroid hormone be a predictive factor for transient postoperative hypocalcemia? Surgery 2015;157:344-8. [Crossref] [PubMed]
- Selberherr A, Scheuba C, Riss P, et al. Postoperative hypoparathyroidism after thyroidectomy: efficient and cost-effective diagnosis and treatment. Surgery 2015;157:349-53. [Crossref] [PubMed]
- Landry CS, Grubbs EG, Hernandez M, et al. Predictable criteria for selective, rather than routine, calcium supplementation following thyroidectomy. Arch Surg 2012;147:338-44. [Crossref] [PubMed]
- Reddy AC, Chand G, Sabaretnam M, et al. Prospective evaluation of intra-operative quick parathyroid hormone assay as an early predictor of post thyroidectomy hypocalcaemia. Int J Surg 2016;34:103-8. [Crossref] [PubMed]
- Graff AT, Miller FR, Roehm CE, et al. Predicting hypocalcemia after total thyroidectomy: parathyroid hormone level vs. serial calcium levels. Ear Nose Throat J 2010;89:462-5. [PubMed]
- Gupta S, Chaudhary P, Durga CK, et al. Validation of intra-operative parathyroid hormone and its decline as early predictors of hypoparathyroidism after total thyroidectomy: A prospective cohort study. Int J Surg 2015;18:150-3. [Crossref] [PubMed]
- Carr AA, Yen TW, Fareau GG, et al. A single parathyroid hormone level obtained 4 hours after total thyroidectomy predicts the need for postoperative calcium supplementation. J Am Coll Surg 2014;219:757-64. [Crossref] [PubMed]
- Lombardi CP, Raffaelli M, Princi P, et al. Parathyroid hormone levels 4 hours after surgery do not accurately predict post-thyroidectomy hypocalcemia. Surgery 2006;140:1016-23; discussion 1023-5. [Crossref] [PubMed]
- Toniato A, Boschin IM, Piotto A, et al. Thyroidectomy and parathyroid hormone: tracing hypocalcemia-prone patients. Am J Surg 2008;196:285-8. [Crossref] [PubMed]
- Huang SM. Do we overtreat post-thyroidectomy hypocalcemia? World J Surg 2012;36:1503-8. [Crossref] [PubMed]
- Lang BH, Yih PC, Ng KK. A prospective evaluation of quick intraoperative parathyroid hormone assay at the time of skin closure in predicting clinically relevant hypocalcemia after thyroidectomy. World J Surg 2012;36:1300-6. [Crossref] [PubMed]
- Lecerf P, Orry D, Perrodeau E, et al. Parathyroid hormone decline 4 hours after total thyroidectomy accurately predicts hypocalcemia. Surgery 2012;152:863-8. [Crossref] [PubMed]
- Higgins KM, Mandell DL, Govindaraj S, et al. The role of intraoperative rapid parathyroid hormone monitoring for predicting thyroidectomy-related hypocalcemia. Arch Otolaryngol Head Neck Surg 2004;130:63-7. [Crossref] [PubMed]
- Roh JL, Park JY, Park CI. Prevention of postoperative hypocalcemia with routine oral calcium and vitamin D supplements in patients with differentiated papillary thyroid carcinoma undergoing total thyroidectomy plus central neck dissection. Cancer 2009;115:251-8. [Crossref] [PubMed]
- Bellantone R, Lombardi CP, Raffaelli M, et al. Is routine supplementation therapy (calcium and vitamin D) useful after total thyroidectomy? Surgery 2002;132:1109-12; discussion 1112-3. [Crossref] [PubMed]
- Roh JL, Park CI. Routine oral calcium and vitamin D supplements for prevention of hypocalcemia after total thyroidectomy. Am J Surg 2006;192:675-8. [Crossref] [PubMed]
- Wang TS, Cheung K, Roman SA, et al. To supplement or not to supplement: a cost-utility analysis of calcium and vitamin D repletion in patients after thyroidectomy. Ann Surg Oncol 2011;18:1293-9. [Crossref] [PubMed]
- Group AESG. Australian Endocrine Surgeons Guidelines AES06/01. Postoperative parathyroid hormone measurement and early discharge after total thyroidectomy: analysis of Australian data and management recommendations. ANZ J Surg 2007;77:199-202. [Crossref] [PubMed]
- Sabour S, Manders E, Steward DL. The role of rapid PACU parathyroid hormone in reducing post-thyroidectomy hypocalcemia. Otolaryngol Head Neck Surg 2009;141:727-9. [Crossref] [PubMed]
- Alía P, Moreno P, Rigo R, et al. Postresection parathyroid hormone and parathyroid hormone decline accurately predict hypocalcemia after thyroidectomy. Am J Clin Pathol 2007;127:592-7. [Crossref] [PubMed]
- Hermann M, Ott J, Promberger R, et al. Kinetics of serum parathyroid hormone during and after thyroid surgery. Br J Surg 2008;95:1480-7. [Crossref] [PubMed]
- Lombardi CP, Raffaelli M, Princi P, et al. Early prediction of postthyroidectomy hypocalcemia by one single iPTH measurement. Surgery 2004;136:1236-41. [Crossref] [PubMed]
- Sywak MS, Palazzo FF, Yeh M, et al. Parathyroid hormone assay predicts hypocalcaemia after total thyroidectomy. ANZ J Surg 2007;77:667-70. [Crossref] [PubMed]
- Richards ML, Bingener-Casey J, Pierce D, et al. Intraoperative parathyroid hormone assay: an accurate predictor of symptomatic hypocalcemia following thyroidectomy. Arch Surg 2003;138:632-5; discussion 635-6. [Crossref] [PubMed]
- Noordzij JP, Lee SL, Bernet VJ, et al. Early prediction of hypocalcemia after thyroidectomy using parathyroid hormone: an analysis of pooled individual patient data from nine observational studies. J Am Coll Surg 2007;205:748-54. [Crossref] [PubMed]


