Nipple sparing mastectomy and the evolving direct to implant breast reconstruction
Introduction
The Irish writer and philosopher, Bernard Shaw said: “If you have an apple and I have an apple and we exchange these apples then you and I will still each have one apple. But if you have an idea and I have an idea and we exchange these ideas, then each of us will have two ideas.”
Sharing ideas and inspiration is the essence of what promotes the evolution of our specialty. The introduction of the acellular dermal matrix (ADM) hammock in 2001 changed implant based breast reconstruction (1). ADM enabled direct to implant (DTI) breast reconstruction omitting the need for intermediate tissue expansion (1,2). The technique can be used for breast reconstruction following skin sparing as well as nipple sparing mastectomy (NSM) (3). In 2005, Scott Spear concluded that preservation of the nipple provided cosmetic advantages that outweighs any additional oncologic risk and has been a proponent for NSM (4-6). The combined procedure of NSM and DTI breast reconstruction is increasingly performed as it is reputed to provide a reliable result with aesthetically pleasing results and is safeguarded in terms of surgical and oncological risks (7-9). In this paper, we describe the evolving use of the NSM and DTI breast reconstruction according to existing literature and our own experience.
The DTI breast reconstruction
A successful DTI approach relies upon the quality of the mastectomy skin flaps and location of the breast footprint on the chest wall as well as the selection of the correct implant size. The technique is best applied in small to moderate sized breasts without significant ptosis (10). Different incisions have been used to access the glandular tissue with similar results. However, the nipple seems to be at greater risks of necrosis when the trans-areolar or the peri-areolar incisions are used, at least compared to the inframammary fold or radial incisions (8). We prefer to perform NSM through the inframammary fold incision aided by hydrodissection, which provides a good overview for the mastectomy with regards to oncological safety as well as hemostasis and patients seem to prefer it due to its hidden location (10) (Figure 1). The radial incision is a good approach for slightly larger and ptotic breasts in order to preserve the blood supply of the inferior skin and allow for better access to the superolateral part of the breast. The standard dual plane hammock reconstruction requires a release of the pectoralis major insertion inferomedially and suturing the hammock to the edge of the pectoral major muscle and the thoracic wall inferolaterally to create a neo-capsule of muscle/hammock pocket to cover the permanent silicone implant (10). The originally described hammock using human derived ADM has been successfully replicated using a variety of biologic and synthetic materials such as human and pig derived skin, pericardium or peritoneum to the synthetic resorbable Vicryl or TIGR-mesh® or non-resorbable titanium coated polypropylene mesh (11,12). Currently, the choice of material for hammock is based on personal preference and pricing rather than scientific evidence, although the use of ADM is more widely documented especially in long term follow up (13-15).
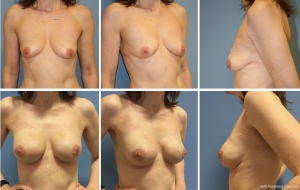
The NSM leaves an intact skin envelope and thereby optimal conditions to achieve the best possible aesthetic result resembling that of a natural breast. The importance of patient selection cannot be overemphasized; the size and shape of the breast, degree of patients’ comorbidity as well as tumor type, size and location are all of paramount importance. It is the obligation of the multidisciplinary team, treating the individual patient, to select possible candidates for NSM and DTI and inform them accordingly of their treatment options. All too often are patients poorly informed of their options and end up having a reconstructive solution favored by their surgeon. This is a habit that must change and the decision should solely be the patients, following an informed consent.
Oncology
Oncological safety is an imperative factor, when NSM and DTI is considered. NSM was shown to be appropriate in carefully selected patients according to a comprehensive review by Mallon et al. and their findings have been further supported by reviews by Endara et al. and Headon et al. (16-18). Headon et al. found an overall pooled locoregional recurrence rate of 2.38%, which is comparable to conventional mastectomy (18). Mallon et al. suggested that patients with early stage invasive breast cancer or ductal carcinoma in situ with a peripherally located tumor less than 5 cm in diameter, located more than 2 cm from the nipple-areola complex (NAC) and not showing HER2 overexpression or exhibiting a positive oestrogen or progesterone receptor status could be considered for NSM with or without adjuvant radiotherapy (17).
Radiation
The literature is questionable whether patients, who have had radiation therapy to the breast or are scheduled for adjuvant postoperative radiation should be considered candidates for NSM and DTI? A recent meta-analysis by Zheng et al. showed a significant association between skin flap necrosis and radiation therapy. However they found no association between nipple areola necrosis and radiation therapy (19). The included studies were heterogenous and the authors mention that their results differ from two large studies by Alperovich et al. and Colwell et al. with divergent results (7,20). Based on our personal experience we do not recommend performing NSM and DTI to patients who have had radiation therapy to the breast. We have also observed poorer results after postoperative radiation therapy compared to non-irradiated and therefore, do not advise NSM and DTI in patients scheduled for adjuvant radiation therapy either.
We have found that reconstructions in radiated patients are most often successful in terms of wound healing in the short run, but the late complications and aesthetic results are worse than breasts that are not irradiated. We have found that radiated patients need several corrections, fat grafting and subsequent implant exchange due to capsular contracture over time. We prefer to perform a delayed breast reconstruction in these cases using some form of autologous tissue, microvascular or pedicled flap transfer. We often use a thoracodorsal artery perforator (TAP) flap for the delayed breast reconstruction. This can be used successfully in one stage to cover the lower pole of an implant fastened with an ADM; TAP, implant and ADM is termed TAPIA and is a delayed form of DTI reconstruction (21-23).
Complications
Complications such as necrosis of the skin and partial to total loss of the NAC, hematoma, seroma, and infection are a risk following NSM and IBR and need to be discussed in advance and dealt with appropriately if they occur. The consequence of such complication can range from conservative outpatient treatment and follow-up to additional surgery and explantation of the implant and a failed reconstruction. The risk of complications is kept to a minimum by correct patient selection and surgeon experience. A meta-analysis by Endara et al. on complication rates in NSM and IBR, skin sparing and nipple sparing with and without hammock technique, showed an overall complication rate of 22% and a nipple necrosis rate of 7%. The incidence of NAC necrosis was slightly higher in one-stage versus two-stage reconstructions, but the overall complication rates were higher in two-stage versus one-stage reconstructions (16).
The most dreaded complication is skin necrosis, which can lead to extrusion and a failed reconstruction. Intraoperative perfusion assessment using indocyanine green color injection and infrared camera technology (SPY) is becoming more common in the United States and seems to enable surgeons to predict a potential postoperative ischemia. One study found that SPY may not be cost-effective in immediate breast reconstruction but the intraoperative assessment seems to contribute to reducing ischemia-related complications (24-26).
It is the current opinion that NSM and DTI is a safe procedure, relatively uncomplicated and results are more aesthetically pleasing than previously seen in alloplastic breast reconstruction, at least in selected patients. As for the patients’ perspective; preservation of the NAC leads to a significantly better body image, sexual well-being and higher satisfaction with the appearance and sensation of the nipple as well as the feeling of being less mutilated compared to patients who had skin-sparing mastectomy (SSM) and a subsequent NAC reconstruction (27-30). There is therefore no doubt that NSM is a great preference from the patient’s point of view and DTI facilitates this process in a single stage.
From a surgeons perspective; NSM and IBR has been found to be a safe method for breast reconstruction, with acceptable risks of complications leaving the patients with very good aesthetic results (13). The best thing about NSM and DTI is the fact that it is a simple and quick procedure and it does not omit any other reconstructive options. It therefore seems to be an optimal first choice when mastectomy is indicated and patient characteristics apply.
Perspectives
A year ago, the late pioneer of plastic surgery Dr. Scott Spear wrote that “advances in breast reconstruction over the past decades are nothing short of amazing”. He followed the birth of the immediate breast reconstruction from nothing in the eighties through a journey of technical advances towards perfection (31). At the end of his journey, the “one and done” NSM and DTI marks merely the end of the beginning of modern breast surgery. The development seems to continue at a rapid pace and it is safe to say that the scope of immediate breast reconstruction, NSM and DTI has increased vastly over the last decade and we will most likely witness further evolution in terms of technical modalities, safety measure and lasting results. At the same time, we also have to be aware of the tendency to publish success stories rather than failures. Increased success leads to higher patient expectations, and disappointments alike. The mastectomy skin flap quality is alpha and omega, when it comes to reconstruction. A sufficient flap is usually the responsibility of a non-plastic surgeon at least until it is insufficient and fails, then it is most often dealt with by a plastic surgeon. The plastic surgeon is not a magician but some of our tricks resemble those of an illusionist. The creation of an illusion of a breast with NSM/DTI is one. Patients need to be aware of this and we must not get ahead of ourselves and remember where we come from knowing that results will be compared in the scrutinizing light of social media. We risk to become victims of our own success as stressed by Scott Spear who stated that we must aim to over deliver and under promise (31).
Long-term follow up after NSM and DTI with the use of a ADM/mesh is warranted. Furthermore, surgeons and researchers should focus more on providing relevant baseline data to ensure the validity of results and also provide clear descriptions of their surgical techniques to facilitate comparability of the results. Moreover, it is not sufficient that we as surgeons evaluate the results. The importance of patient-related outcome measures has become clear and must be applied more avidly in the future (32).
Pre-pectoral placement of the implant with complete ADM coverage is gaining increasing attention in DTI after NSM to overcome animation deformity caused by activation of the pectoralis major muscle (33-35). The literature regarding pre-pectoral implant placement and level of evidence of studies thereof is sparse, however we will most certainly witness a rapid increase in publications of this topic in the near future.
Our overall impression is positive. Improved operative technique for mastectomy and breast reconstruction goes hand in hand with improved cosmetic outcomes and fewer complications. We postulate that the evolution of NSM and DTI breast reconstruction will continue together with the development in biotechnology, implant technology, perhaps with lighter implants in combination with improved bioprosthetics (31)? We have just seen the first report of lighter implants (36). And hybrid approach where implants are used for projection and breast shape together with autologous fat grafting for bolstering of the skin flap with or without a hammock support of the implant are becoming standard of care. It is but natural to apply the tools that are available and the toolbox has grown substantially over the last decade. How we apply these tools must be individualized and remain free from industrial bias or conflict of interest and when we find a tool that works it is our duty to share our findings as we have been fortunate to learn from our peers. One thing is certain, that we need to be realistic in terms of what we can and cannot do and stay honest to ourselves and safeguard our patient’s best interest (31).
This article is written in the honorable memory of Dr. Scott Spear who has inspired us greatly. It has been a pleasant privilege to witness that when we think of something “new”, then Scott Spear had already published it. If he was there before us, it can only mean that we are on the right path and that is always motivating and inspiring.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Salzberg CA. Nonexpansive immediate breast reconstruction using human acellular tissue matrix graft (AlloDerm). Ann Plast Surg 2006;57:1-5. [Crossref] [PubMed]
- Breuing KH, Warren SM. Immediate bilateral breast reconstruction with implants and inferolateral AlloDerm slings. Ann Plast Surg 2005;55:232-9. [Crossref] [PubMed]
- Salzberg CA. Direct-to-implant breast reconstruction. Clin Plast Surg 2012;39:119-26. [Crossref] [PubMed]
- Spear SL, Carter ME, Schwarz K. Prophylactic mastectomy: indications, options, and reconstructive alternatives. Plast Reconstr Surg 2005;115:891-909. [Crossref] [PubMed]
- Spear SL, Hannan CM, Willey SC, et al. Nipple-sparing mastectomy. Plast Reconstr Surg 2009;123:1665-73. [Crossref] [PubMed]
- Spear SL, Willey SC, Feldman ED, et al. Nipple-sparing mastectomy for prophylactic and therapeutic indications. Plast Reconstr Surg 2011;128:1005-14. [Crossref] [PubMed]
- Colwell AS, Tessler O, Lin AM, et al. Breast reconstruction following nipple-sparing mastectomy: predictors of complications, reconstruction outcomes, and 5-year trends. Plast Reconstr Surg 2014;133:496-506. [Crossref] [PubMed]
- Casella D, Calabrese C, Orzalesi L, et al. Current trends and outcomes of breast reconstruction following nipple-sparing mastectomy: results from a national multicentric registry with 1006 cases over a 6-year period. Breast Cancer 2017;24:451-7. [Crossref] [PubMed]
- Nahabedian MY. Implant-based breast reconstruction following conservative mastectomy: one-stage vs. two-stage approach. Gland Surg 2016;5:47-54. [PubMed]
- Gunnarsson GL, Børsen-Koch M, Wamberg P, et al. How to perform a NAC sparing mastectomy using an ADM and an implant. Gland Surg 2014;3:252-7. [PubMed]
- Logan Ellis H, Asaolu O, Nebo V, et al. Biological and synthetic mesh use in breast reconstructive surgery: a literature review. World J Surg Oncol 2016;14:121. [Crossref] [PubMed]
- Dieterich M, Paepke S, Zwiefel K, et al. Implant-based breast reconstruction using a titanium-coated polypropylene mesh (TiLOOP Bra): a multicenter study of 231 cases. Plast Reconstr Surg 2013;132:8e-19e. [Crossref] [PubMed]
- Salzberg CA, Ashikari AY, Berry C, et al. Acellular Dermal Matrix-Assisted Direct-to-Implant Breast Reconstruction and Capsular Contracture: A 13-Year Experience. Plast Reconstr Surg 2016;138:329-37. [Crossref] [PubMed]
- Seth AK, Hirsch EM, Fine NA, et al. Utility of acellular dermis-assisted breast reconstruction in the setting of radiation: a comparative analysis. Plast Reconstr Surg 2012;130:750-8. [Crossref] [PubMed]
- Spear SL, Parikh PM, Reisin E, et al. Acellular dermis-assisted breast reconstruction. Aesthetic Plast Surg 2008;32:418-25. [Crossref] [PubMed]
- Endara M, Chen D, Verma K, et al. Breast reconstruction following nipple-sparing mastectomy: a systematic review of the literature with pooled analysis. Plast Reconstr Surg 2013;132:1043-54. [Crossref] [PubMed]
- Mallon P, Feron JG, Couturaud B, et al. The role of nipple-sparing mastectomy in breast cancer: a comprehensive review of the literature. Plast Reconstr Surg 2013;131:969-84. [Crossref] [PubMed]
- Headon HL, Kasem A, Mokbel K. The Oncological Safety of Nipple-Sparing Mastectomy: A Systematic Review of the Literature with a Pooled Analysis of 12,358 Procedures. Arch Plast Surg 2016;43:328-38. [Crossref] [PubMed]
- Zheng Y, Zhong M, Ni C, et al. Radiotherapy and nipple-areolar complex necrosis after nipple-sparing mastectomy: a systematic review and meta-analysis. Radiol Med 2017;122:171-8. [Crossref] [PubMed]
- Alperovich M, Tanna N, Samra F, et al. Nipple-sparing mastectomy in patients with a history of reduction mammaplasty or mastopexy: how safe is it? Plast Reconstr Surg 2013;131:962-7. [Crossref] [PubMed]
- Børsen-Koch M, Gunnarsson GL, Udesen A, et al. Direct delayed breast reconstruction with TAP flap, implant and acellular dermal matrix (TAPIA). J Plast Reconstr Aesthet Surg 2015;68:815-21. [Crossref] [PubMed]
- Heidemann LN, Gunnarsson GL, Bille C, et al. Reconstructive breast surgery using implant and mesh. Ugeskr Laeger 2017;179. [PubMed]
- Gunnarsson GL, Børsen-Koch M, Nielsen HT, et al. Bilateral Breast Reconstruction with Extended Thoracodorsal Artery Perforator Propeller Flaps and Implants. Plast Reconstr Surg Glob Open 2015;3:e435. [Crossref] [PubMed]
- Kanuri A, Liu AS, Guo L. Whom should we SPY? A cost analysis of laser-assisted indocyanine green angiography in prevention of mastectomy skin flap necrosis during prosthesis-based breast reconstruction. Plast Reconstr Surg 2014;133:448e-54e. [Crossref] [PubMed]
- Sood M, Glat P. Potential of the SPY intraoperative perfusion assessment system to reduce ischemic complications in immediate postmastectomy breast reconstruction. Ann Surg Innov Res 2013;7:9. [Crossref] [PubMed]
- Newman MI, Jack MC, Samson MC. SPY-Q analysis toolkit values potentially predict mastectomy flap necrosis. Ann Plast Surg 2013;70:595-8. [Crossref] [PubMed]
- El Hage Chehade H, Headon H, Wazir U, et al. Nipple-sparing mastectomy using a hemi-periareolar incision with or without minimal medial-lateral extensions; clinical outcome and patient satisfaction: A single centre prospective observational study. Am J Surg 2017;213:1116-24. [Crossref] [PubMed]
- Metcalfe KA, Cil TD, Semple JL, et al. Long-Term Psychosocial Functioning in Women with Bilateral Prophylactic Mastectomy: Does Preservation of the Nipple-Areolar Complex Make a Difference? Ann Surg Oncol 2015;22:3324-30. [Crossref] [PubMed]
- Didier F, Radice D, Gandini S, et al. Does nipple preservation in mastectomy improve satisfaction with cosmetic results, psychological adjustment, body image and sexuality? Breast Cancer Res Treat 2009;118:623-33. [Crossref] [PubMed]
- Wei CH, Scott AM, Price AN, et al. Psychosocial and Sexual Well-Being Following Nipple-Sparing Mastectomy and Reconstruction. Breast J 2016;22:10-7. [Crossref] [PubMed]
- Spear SL. Underpromise. Plast Reconstr Surg 2016;137:1961-2. [Crossref] [PubMed]
- Pusic AL, Chen CM, Cano S, et al. Measuring quality of life in cosmetic and reconstructive breast surgery: a systematic review of patient-reported outcomes instruments. Plast Reconstr Surg 2007;120:823-37; discussion 838-9. [Crossref] [PubMed]
- Downs RK, Hedges K. An Alternative Technique for Immediate Direct-to-Implant Breast Reconstruction-A Case Series. Plast Reconstr Surg Glob Open 2016;4:e821. [Crossref] [PubMed]
- Reitsamer R, Peintinger F. Prepectoral implant placement and complete coverage with porcine acellular dermal matrix: a new technique for direct-to-implant breast reconstruction after nipple-sparing mastectomy. J Plast Reconstr Aesthet Surg 2015;68:162-7. [Crossref] [PubMed]
- Kobraei EM, Cauley R, Gadd M, et al. Avoiding Breast Animation Deformity with Pectoralis-Sparing Subcutaneous Direct-to-Implant Breast Reconstruction. Plast Reconstr Surg Glob Open 2016;4:e708. [Crossref] [PubMed]
- Govrin-Yehudain J, Dvir H, Preise D, et al. Lightweight breast implants: a novel solution for breast augmentation and reconstruction mammaplasty. Aesthet Surg J 2015;35:965-71. [Crossref] [PubMed]


