Lipofilling in breast cancer surgery
Introduction
Lipofilling is the process of relocating autologous fat to change the shape, volume, consistency and profile of tissues. For over a century, surgeons have used autologous fat to enlarge and reshape the breasts. In 1895, the German surgeon Czerny performed the first breast augmentation by transplanting a lipoma from the lumbar region to a breast defect (1). With the advent of liposuction in the 1980s, large amounts of unwanted fat could be removed from different body areas using small access incisions and a suction cannula. In this setting, fat grafting was re-introduced in the early 80s, pioneered by the American Mel Bircoll, who first described a series of fat transplantation for breast augmentation and reconstruction (2) Bircoll’s contribution to fat grafting was met with a considerable amount of criticism from the plastic surgery leadership, with the American Society of Plastic Surgeons (“ASPS”) releasing a position statement about the procedure in 1987.
In 2007, Coleman published his landmark review of 17 breast augmentation and reconstruction patients who were treated using autologous fat and were followed up with serial photography (3). Some Europeans persisted and continued to push for the technique, though not for cosmetic augmentation. Delay E. et al. in Lyon, France has begun using fat grafting to the breast for reconstruction as early as 2000 and Rigotti G. et al. from Verona, Italy also had a large series that he presented at the European of Aesthetic Plastic Surgeons and Petit JY. et al. and Rietjens M et al. reported several series and clinical outcomes from European Institute of Oncology (EIO), Milan Italy (4-6).
Application of lipofilling in breast cancer surgery
Lipofilling is being indicated for soft tissue defect correction in many sites. It is not only for corrective surgery but also for cosmetic purpose. For breast cancer surgery, we suggest that lipofilling procedure might be advantage in the following situations:
Correction of defects and asymmetry following wide local excision (or breast conservative surgery), with or without radiotherapy.
Improvement of soft tissue coverage following implant based breast reconstruction.
Volume replacement of implants in unsatisfactory oncoplastic breast reconstruction outcomes.
Augmentation of volume and refinement after autologous breast reconstruction.
Whole breast reconstruction with serial fat grafting.
Scar correction.
Oncological safety
Experimental studies have shown that adipocytes can stimulate breast cancer cells. Adipokines are factors that can stimulate breast cancerous cells through endocrine, paracrine and autocrine pathways; theoretically the “tumor stroma interaction” can potentially induce cancer reappearance by fueling dormant tumor cancer cells in the tumor bed (7-9).
At first the oncological safety was considered as a priority problem for lipofilling but many studies now showed the safety of lipofilling as that multicentric study (Milan-Paris-Lyon) performed by Petit et al. There were 513 patients and performed 646 lipofilling procedures. The average interval between oncologic surgical interventions and lipofilling was 39.7 months. Average follow-up after lipofilling was 19.2 months. They observed a low complication rate; the overall oncologic event rate was 5.6 percent (3.6 percent per year). The overall locoregional event rate was 2.4 percent (10).
Petit et al. reported a retrospective matched cohort study on 321 consecutive patients operated for primary breast cancer who subsequently underwent lipofilling for reconstructive purpose. The median follow up of 56 months from primary surgery and 26 months from the lipofilling had shown no significant local recurrence when compared to 642 patients as a control group (11). However, there is a trend of higher risk of local event in subgroup of ductal carcinoma in situ. In 2010, Rietjens et al. reported a series of lipofilling procedures in breast cancer treatment and reconstruction. They followed 158 patients and found that postoperative complication rates are very low (3.6 percent) and that there is little alteration in post lipofilling mammographic finding (5.9%) (6).
Seth et al., made a retrospective comparative study on 886 patients (1,202 breasts) from 1998 to 2008 and revealed no significant differences in demographics, operative characteristics, tumor staging, or radiation therapy exposure between fat-grafting (n=90 breasts) and non-fat grafting (n=1,112 breasts) patients. Ninety-nine fat-grafting procedures were performed an average of 18.3 months after reconstruction, with only one complication (fat necrosis), they concluded that fat grafting did not affect local tumor recurrence or survival when compared with non-fat-grafted breasts (12).
In 2007, the French Society of Plastic Surgery (SOFCPRE) announced that that they would not support the use of lipofilling for treating defects resulting from breast-conserving treatment (BCT) as a result of the lack of evidence on its oncological safety. A phase III multicenter randomized, controlled trial is currently taking place in France with the goal of investigating this issue (13). Also the American society of plastic surgeons had set up a task force in 2009 (ASPS Fat Graft Task Force) to assess the indications, the safety and efficacy of autologous fat grafting on 283 patients it showed the risk of malignancy with lipofilling could not be identified due to lack of standardized techniques and randomized controlled trials (14).
Eventhough lipofilling seems to be a safe procedure in breast cancer patients. Longer follow-up and further experiences from large multicentric oncological series are urgently required to confirm these findings.
Efficacy of lipofilling
Although several teams published results of total breast reconstruction with repeated sessions of lipofilling, for most authors, the technique remains indicated for local improvement of small defects or asymmetry only (15). Many studies had shown efficacy of lipofilling but an important one is in 2009, ASPS Fat Graft Task Force assessed the efficacy on 283 patients, most of them showed satisfactory results, as reported by the patients and/or independent panels of surgeons, Follow-up ranged from 1 month to 10 years. Thirty six complications (12.7%) or unfavorable sequelae were reported: three (1.1%) infections, 14 (4.9%) calcifications, 16 (5.7%) fat necrosis, and three (1.1%) unspecified superficial lumps (14).
Another study by Illouz and Sterodimas, reported a series of 820 consecutive patients who underwent autologous fat transplantation over 25 years. Majority of the study population were breast augmentation but also included patients who underwent lipofilling after breast reconstruction and congenital asymmetry, the authors indicted that the majority of patients were satisfied with the results (15). A questionnaire study on 44 patients who had lipofilling after mastectomy and breast reconstruction or breast conserving surgery for cancer in Karolinska University hospital, Sweden focused on the consistency of the breast, the size of the breast, the shape of the breast, the sensitivity of the skin, its quality, and irregularities of the breast. The result of questionaire revealed an improvement in shape and size after lipofilling and there was only one complication which was liponecrosis (16) While Beck et al. in 2011 published a prospective study on 10 patients who underwent lipofilling after conservative breast surgery, 80% of them failed to achieve a satisfactory outcome from lipofilling and like to have another lipofilling for improvement. Another 20% did not want another augmentation because they were fully satisfied with the primary result (17).
However, the efficacy of the lipofilling procedure depends on the protocol of each institution. Moreover, fat grafting tends to resorb approximately 30% due to various factor, such as timing of grafting, amount of grafting, vascularization of tissue bed, irradiated tissue or even individual objective and subjective evaluation.
Lipofilling in reconstruction of irradiated breast
External radiation is a mandatory part of adjuvant therapy in order to decrease loco regional recurrence in breast cancer local control (18-20). In postmastectomy radiated patients, autologous tissue reconstruction is preferred over implant reconstruction, because the latter is associated with a higher rate of postoperative complications (19). Few studies demonstrated the outcome of lipofilling on irradiated breast (21-23).
Salgarello et al. retrospectively studied 16 patients who underwent lipofilling to the chest wall after irradiation and then followed by prosthesis introduction and found high success rate of prosthetic based procedure with no complication or oncological recurrence (21). Also another series by Sarfati et al. performed lipofilling prior to implant introduction after irradiation in 28 patients. They reported high success rate with only 1 prosthesis exchange due to prosthesis exposure after lipofilling (22). Pannettiere et al. study 60 post mastectomy reconstruction by compared 20 patient who underwent fat grafting before reconstruction with prosthesis and the other 40 patients without fat grafting. They showed that lipofilling is a safe and reliable technique in improving the outcomes of irradiated reconstructed breasts with implants (23).
More studies are needed to assess the efficacy of lipofilling on irradiated breast and the possibility of lipofilling as a sole reconstructive solution for irradiated breast.
Technique of lipofilling
Lipofilling can be performed under general or local anasthesia. Generally, the aim of the technique is to decrease cell damage and to promote survival of the fat tissue and its composition. Lipofilling is not just a simply matter of injecting fat taken from one area of the body to another. Success is heavily dependent on the technique used for harvesting, preparing and grafting of the fat. Furthermore, donor site aesthetics must be considered, not only to minimize morbidity and deformity, but also to improve contour and body profile, thus enhancing the appearance of the areas used.
Identification of the donor site
The various adipose areas of the body were examined to identify the natural fat deposits.
The most common site is the abdominal fat because it is one of the most fat deposit area. Moreover, there is no need to change the patient’s position in the operation room. The second site was the trochanteric region (saddle bags) and the inside of the thighs and knees. The harvesting areas were outlined with a skin marker.
Preparation of the solution
The tumescent solution is prepared to be injected into the donor site before start the procedure. It is so called “Klein’s solution” which contains 1 cc of epinephrine (1:500,000) diluted in 500 cc of 0.001% lactate ringer solution (LRS). The 50 cc of Mepivacaine can be added in the solution if the procedure is planned under local anesthesia. It is injected through a small bore 4 mm blunt cannula that attached to a 60-cc syringe. The estimate volume of solution is 1 cc for each 1 cm3 of target fat harvest volume. The surgeon should wait at least 15 minutes before starting fat harvesting (24), the adrenaline is added to the solution in order to achieve well hemostasis and to decrease postoperative pain (25).
Fat harvesting
Harvesting is a major contributory factor to the success of lipofilling. The most well known technique of fat harvesting is described by Coleman (25). The procedure starts through a small incision made in the abdomen by blade no.11 and gradually apply a blunt tip harvesting cannula (3 mm in diameter and 15 or 23 cm in length). Manually, the syringe is drawn to create to low negative pressure during fat harvesting. The cannula is attached to 10 cc. Luer-Lock syringes. However, various the techniques of fat harvesting with different cannula or liposuction machine system have been reported with different outcome assessments (26-34).
Different cannula size may affect the viability of harvested fat. An experimental study by Ozsoy et al., demonstrated a greater number of viable adipocytes when harvested with a 4-mm-diameter cannula compared with 2- or 3-mm cannulas (26). Erdim et al. also recommended the use of larger cannulas to increase cell viability. Their study showed more viability of fat cells when using 6mm diameter cannula than using 2 or 4 mm in diameter cannula (27).
Different vacuum pressures and some assisted techniques has been used in many clinical series. Rohrich et al. compared traditional liposuction, internal ultrasound-assisted liposuction, external ultrasound assisted liposuction and massage assisted technique liposuction. There was no significant histologic or chemical effect of external ultrasound-assisted liposuction on harvested adipocytes. However, internal ultrasound-assisted aspiration caused a thermal liquefaction of mature adipocytes (28). Shiffman and Mirrafati used various cannulas, needles, suction pressures, external ultrasound, or preoperative massage to determine whether there was an effect on cell viability, and they found that cell damage of greater than 10% occurred when a –700 mmHg vacuum was applied (29). Ferguson et al. demonstrated a significantly higher viable adipocyte count using the syringe aspiration at low vacuum pressure compared with conventional liposuction (30). In contrary, Leong et al. found no differences in cell viability, cell metabolic activity, or adipogenic responses when syringe liposuction and compared to pump-assisted liposuction (31). Pu et al. compared the Coleman technique versus conventional liposuction technique and found significantly higher viable adipocyte level in the Coleman technique (32,33). Crawford et al. examined the hand aspirate at low-force centrifuge versus standard power-assisted liposuction and showed higher cell counts were observed when using the low force centrifuge (34).
Fat processing
There most frequently used methods for fat processing are centrifugation, washing and decantation. The purified fat can be separated from cell debris by centrifugation, as described in the widely used protocol by Coleman (25). After centrifugation, the lipoaspirated specimen can be separated into 4 layers: (I) the oily fraction, leaked out of disrupted adipocytes; (II) the watery fraction consisting of blood, lidocaine and saline, injected before the liposuction; (III) a cell pellet on the bottom; and (IV) the purified fat between the oily and the watery fractions. For washing technique, the fat is washed using normal saline (35) or 5% glucose solution (36) in order to wash out the blood and the oil part and cellular debris from the aspirated fat. The least popular technique is decantation, which use the gravity effect to precipitate the cellular component from the oily and water component. Table 1 shows the different studies on fat processing techniques and the findings.
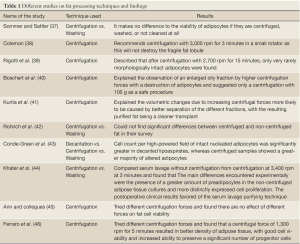
Full Table
Fat transfer (injection)
Our technique at EIO, we using modified Coleman technique (6) by injecting the processed fat via 2 mm diameter cannula attach to 1 mL disposable syringe. The fat was transferred directly to the breast, we try to avoid intraparenchymal injection and avoid creating the bolus injection. The entry site of the cannula can be made by sharp blade or a sharp 17-gauge needle to minimize the scar (4,47,48,).
It is mandatory to overcorrect the defects because 40-60% of the transferred fat is expected to be resorbed. Experimental studies have found that up to 90% of transplanted adipose tissue could be lost (49-51). While clinically reported figures range between 40% and 60% (4,15,52,53), and most of the volume loss occurs within the first 4-6 months following surgery (4,15). Despite, several novel techniques proclaimed that they produced more effective outcomes (14-16). Nonetheless, surgeon should calculate the quantity of fat preparation and injection before the procedure. The limitation of volume inject can be due to the recipient tissues quality such as in irradiation tissue or thick scar. If the target volume cannot be achieved by a single lipofilling procedure, then the patient should be informed for the possibility of another lipofilling (4). In general we employ a 140% overcorrection rule which 140 mL of fat should be injected for a desired final volume of 100 mL (4).
Complication
Complications can be classified into recipient site and donor site complications.
Recipient site complication (4,14,15,54,55)
-
Fat necrosis, oil cyst formation and calcifications can occur due to injection of large volumes into a single area or injecting fat into poorly vascularized areas resulting in failure of “graft take” with palpable mass formation resulting from fat necrosis which may be difficult to distinguish clinically from local recurrence in breast cancer patients and lead to a need for additional imaging and needle biopsy (3-15%). Moreover, post lipofilling calcification can be found in mammogram (0.7-4.9%).
-
Infection (0.6-1.1%).
-
Under-correction or over-correction of deformity.
-
Damage to underlying structures e.g., breast implants, pneumothorax.
-
Intravascular injection with fat embolism.
Donor site complication (4,14,15,54,55)
Complications appear to be minimal and related to liposuction technique. The possible complications include bruising, swelling, hematoma formation, paresthesia or donor site pain, infection, hypertrophic scarring, contour irregularities and damage to underlying structure such as intraperitoneal or intramuscular penetration of the cannula.
Future of lipofilling
One promising future topic is progenitor cell or stem cell selection. Theoretically, adipose tissue-derived mesenchymal stem cells (ASC) and/or ASC-enriched lipofilling have the potential for improving lipofilling result, particularly for its resorption rate, quality of tissue, and oncological safety. Furthermore, the better understanding of cancer stem cells and role of adipocyte and its derivative in cancer stromal interaction and breast tissue engineering should be encourage for future prospective (9,56,57).
Conclusions
Lipofilling in breast cancer surgery can be performed as a day surgery procedure and it has acceptable efficacy in correction of deformities without compromising oncological outcomes. More studies are needed to assess the long term oncological outcome. Application of experimental and fundamental researches on tissue engineering and stem cells can carry more hopes to augment the role of lipofilling in the future.
Acknowledgements
Author Thanks to the colleague in department of the plastic surgery at the European Institute of Oncology.
Disclosure: Authors declare no conflict of commercial interest, no any financial or material support.
References
- Czerny V. Plastischer Ersatz der Brustdruse durch ein Lipom. Zentralbl Chir 1895;27:72.
- Bircoll M, Novack BH. Autologous fat transplantation employing liposuction techniques. Ann Plast Surg 1987;18:327-9.
- Coleman SR, Saboeiro AP. Fat grafting to the breast revisited: safety and efficacy. Plast Reconstr Surg 2007;119:775-85; discussion 786-7.
- Delay E, Garson S, Tousson G, et al. Fat injection to the breast: technique, results, and indications based on 880 procedures over 10 years. Aesthet Surg J 2009;29:360-76.
- Rigotti G, Marchi A, Galiè M, et al. Clinical treatment of radiotherapy tissue damage by lipoaspirate transplant: a healing process mediated by adipose-derived adult stem cells. Plast Reconstr Surg 2007;119:1409-22; discussion 1423-4.
- Rietjens M, De Lorenzi F, Rossetto F, et al. Safety of fat grafting in secondary breast reconstruction after cancer. J Plast Reconstr Aesthet Surg 2011;64:477-83.
- Vona-Davis L, Rose DP. Adipokines as endocrine, paracrine, and autocrine factors in breast cancer risk and progression. Endocr Relat Cancer 2007;14:189-206.
- Hou WK, Xu YX, Yu T, et al. Adipocytokines and breast cancer risk. Chin Med J (Engl) 2007;120:1592-6.
- Lohsiriwat V, Curigliano G, Rietjens M, et al. Autologous fat transplantation in patients with breast cancer: “silencing” or “fueling” cancer recurrence? Breast 2011;20:351-7.
- Petit JY, Lohsiriwat V, Clough KB, et al. The oncologic outcome and immediate surgical complications of lipofilling in breast cancer patients: a multicenter study--Milan-Paris-Lyon experience of 646 lipofilling procedures. Plast Reconstr Surg 2011;128:341-6.
- Petit JY, Botteri E, Lohsiriwat V, et al. Locoregional recurrence risk after lipofilling in breast cancer patients. Ann Oncol 2012;23:582-8.
- Seth AK, Hirsch EM, Kim JY, et al. Long-term outcomes following fat grafting in prosthetic breast reconstruction: a comparative analysis. Plast Reconstr Surg 2012;130:984-90.
- ClinialTrials.gov. Adipose tissue transfer for moderate breast cancer conservative treatment sequel (GRATSEC). NCT01035268.
- Gutowski KA, ASPS Fat Graft Task Force. Current applications and safety of autologous fat grafts: a report of the ASPS fat graft task force. Plast Reconstr Surg 2009;124:272-80.
- Illouz YG, Sterodimas A. Autologous fat transplantation to the breast: a personal technique with 25 years of experience. Aesthetic Plast Surg 2009;33:706-15.
- Schultz I, Lindegren A, Wickman M. Improved shape and consistency after lipofilling of the breast: patients' evaluation of the outcome. J Plast Surg Hand Surg 2012;46:85-90.
- Beck M, Ammar O, Bodin F, et al. Evaluation of breast lipofilling after sequelae of conservative treatment of cancer. Eur J Plast Surg 2012;35:221-8.
- Whelan TJ, Julian J, Wright J, et al. Does locoregional radiation therapy improve survival in breast cancer? A meta-analysis. J Clin Oncol 2000;18:1220-9.
- Clarke M, Collins R, Darby S, et al. Effects of radiotherapy and of differences in the extent of surgery for early breast cancer on local recurrence and 15-year survival: an overview of the randomised trials. Lancet 2005;366:2087-106.
- Benson JR, Jatoi I, Keisch M, et al. Early breast cancer. Lancet 2009;373:1463-79.
- Salgarello M, Visconti G, Barone-Adesi L. Fat grafting and breast reconstruction with implant: another option for irradiated breast cancer patients. Plast Reconstr Surg 2012;129:317-29.
- Sarfati I, Ihrai T, Kaufman G, et al. Adipose-tissue grafting to the post-mastectomy irradiated chest wall: preparing the ground for implant reconstruction. J Plast Reconstr Aesthet Surg 2011;64:1161-6.
- Panettiere P, Marchetti L, Accorsi D. The serial free fat transfer in irradiated prosthetic breast reconstructions. Aesthetic Plast Surg 2009;33:695-700.
- Pitman GH. Liposuction and body contouring. In: Aston SJ, Beasley RW, Thorne CH. eds. Grabb and Smith’s plastic surgery. 5th Edition. Philadelphia: Lippincott-Raven, 1997:673-5.
- Coleman SR. Facial augmentation with structural fat grafting. Clin Plast Surg 2006;33:567-77.
- Ozsoy Z, Kul Z, Bilir A. The role of cannula diameter in improved adipocyte viability: a quantitative analysis. Aesthet Surg J 2006;26:287-9.
- Erdim M, Tezel E, Numanoglu A, et al. The effects of the size of liposuction cannula on adipocyte survival and the optimum temperature for fat graft storage: an experimental study. J Plast Reconstr Aesthet Surg 2009;62:1210-4.
- Rohrich RJ, Morales DE, Krueger JE, et al. Comparative lipoplasty analysis of in vivo-treated adipose tissue. Plast Reconstr Surg 2000;105:2152-8; discussion 2159-60.
- Shiffman MA, Mirrafati S. Fat transfer techniques: the effect of harvest and transfer methods on adipocyte viability and review of the literature. Dermatol Surg 2001;27:819-26.
- Ferguson RE, Cui X, Fink BF, et al. The viability of autologous fat grafts harvested with the LipiVage system: a comparative study. Ann Plast Surg 2008;60:594-7.
- Leong DT, Hutmacher DW, Chew FT, et al. Viability and adipogenic potential of human adipose tissue processed cell population obtained from pump-assisted and syringe-assisted liposuction. J Dermatol Sci 2005;37:169-76.
- Pu LL, Cui X, Fink BF, et al. The viability of fatty tissues within adipose aspirates after conventional liposuction: a comprehensive study. Ann Plast Surg 2005;54:288-92; discussion 292.
- Pu LL, Coleman SR, Cui X, et al. Autologous fat grafts harvested and refined by the Coleman technique: a comparative study. Plast Reconstr Surg 2008;122:932-7.
- Crawford JL, Hubbard BA, Colbert SH, et al. Fine tuning lipoaspirate viability for fat grafting. Plast Reconstr Surg 2010;126:1342-8.
- Asken S. Autologous fat transplantation: micro and macro techniques. Am J Cosmet Surg 1987:4:111-21.
- Fournier PF. Fat grafting: my technique. Dermatol Surg 2000;26:1117-28.
- Sommer B, Sattler G. Current concepts of fat graft survival: histology of aspirated adipose tissue and review of the literature. Dermatol Surg 2000;26:1159-66.
- Coleman SR. Structural fat grafts: the ideal filler? Clin Plast Surg 2001;28:111-9.
- Rigotti G, Marchi A, Galiè M, et al. Clinical treatment of radiotherapy tissue damage by lipoaspirate transplant: a healing process mediated by adipose-derived adult stem cells. Plast Reconstr Surg 2007;119:1409-22; discussion 1423-4.
- Boschert MT, Beckert BW, Puckett CL, et al. Analysis of lipocyte viability after liposuction. Plast Reconstr Surg 2002;109:761-5; discussion 766-7.
- Kurita M, Matsumoto D, Shigeura T, et al. Influences of centrifugation on cells and tissues in liposuction aspirates: optimized centrifugation for lipotransfer and cell isolation. Plast Reconstr Surg 2008;121:1033-41; discussion 1042-3.
- Rohrich RJ, Sorokin ES, Brown SA. In search of improved fat transfer viability: a quantitative analysis of the role of centrifugation and harvest site. Plast Reconstr Surg 2004;113:391-5; discussion 396-7.
- Condé-Green A, de Amorim NF, Pitanguy I. Influence of decantation, washing and centrifugation on adipocyte and mesenchymal stem cell content of aspirated adipose tissue: a comparative study. J Plast Reconstr Aesthet Surg 2010;63:1375-81.
- Khater R, Atanassova P, Anastassov Y, et al. Clinical and experimental study of autologous fat grafting after processing by centrifugation and serum lavage. Aesthetic Plast Surg 2009;33:37-43.
- Pulsfort AK, Wolter TP, Pallua N. The effect of centrifugal forces on viability of adipocytes in centrifuged lipoaspirates. Ann Plast Surg 2011;66:292-5.
- Ferraro GA, De Francesco F, Tirino V, et al. Effects of a new centrifugation method on adipose cell viability for autologous fat grafting. Aesthetic Plast Surg 2011;35:341-8.
- Delay E. Breast deformities. In: Coleman SR, Mazzola RF. eds. Fat Injection: From Filling to Regeneration. Saint Louis: Quality Medical Publishing, 2008.
- Delay E. Fat injections to the breast: the lipomodeling technique. In: Hall-Findlay EJ, Evans GRD, eds. Aesthetic and Reconstructive Surgery of the Breast. NewYork: Saunders Elsevier, 2010: 171-92.
- Smith P, Adams WP Jr, Lipschitz AH, et al. Autologous human fat grafting: effect of harvesting and preparation techniques on adipocyte graft survival. Plast Reconstr Surg 2006;117:1836-44.
- Nguyen A, Pasyk KA, Bouvier TN, et al. Comparative study of survival of autologous adipose tissue taken and transplanted by different techniques. Plast Reconstr Surg 1990;85:378-86; discussion 387-9.
- Kononas TC, Bucky LP, Hurley C, et al. The fate of suctioned and surgically removed fat after reimplantation for soft-tissue augmentation: a volumetric and histologic study in the rabbit. Plast Reconstr Surg 1993;91:763-8.
- Niechajev I, Sevćuk O. Long-term results of fat transplantation: clinical and histologic studies. Plast Reconstr Surg 1994;94:496-506.
- Zocchi ML, Zuliani F. Bicompartmental breast lipostructuring. Aesthetic Plast Surg 2008;32:313-28.
- Coleman SR. Structural fat grafting: more than a permanent filler. Plast Reconstr Surg 2006;118:108S-120S.
- Chan CW, McCulley SJ, Macmillan RD. Autologous fat transfer--a review of the literature with a focus on breast cancer surgery. J Plast Reconstr Aesthet Surg 2008;61:1438-48.
- Trojahn Kølle SF, Oliveri RS, Glovinski PV, et al. Importance of mesenchymal stem cells in autologous fat grafting: a systematic review of existing studies. J Plast Surg Hand Surg 2012;46:59-68.
- Tiryaki T, Findikli N, Tiryaki D. Staged stem cell-enriched tissue (SET) injections for soft tissue augmentation in hostile recipient areas: a preliminary report. Aesthetic Plast Surg 2011;35:965-71.


