Operative bed recurrence of thyroid cancer: utility of a preoperative needle localization technique
Introduction
Over 500,000 people in the United States are estimated to be living with thyroid cancer, with approximately 60,000 new cases reported each year (1). Treatment for well-differentiated thyroid carcinoma entails surgery along with adjuvant iodine-131 therapy, and 10-year overall survival rates are roughly 90% (2). Recurrence rates vary in the literature, with one large series of over 500 patients reporting a recurrence rate of 13% (2). Yet, recurrence rates of up to 35% have been reported. Two-thirds of these recurrent episodes are cervical or mediastinal (3). Surveillance for recurrence can include measuring thyroglobulin levels or imaging including ultrasound (US), computed tomography (CT), magnetic resonance imaging (MRI), or radioiodine scanning (3). For recurrent palpable cervical lymph node involvement, neck dissection is the accepted treatment. However, for nonpalpable recurrent cervical malignancy, management remains controversial.
Recurrent disease is associated with significantly poorer outcomes. Twenty-year disease-specific survival rates in patients without recurrent disease versus multiple sites of recurrence are 100% and 60%, respectively (2). Treatment in these patients can involve reoperation and/or radioactive iodine-131 therapy. However, reoperation is associated with significantly increased morbidity, particularly with a 1.5% chance of recurrent laryngeal nerve palsy and a 2.5% chance of hypoparathyroidism (4). Postoperative scarring can make reoperation difficult, secondary to unclear anatomy, leading to longer operative times and morbidity (5,6). Beyond the complications of recurrent laryngeal nerve injury and hypoparathyroidism, failure to identify and resect the recurrent disease is another risk of reoperative surgery for local recurrence.
With the aforementioned risks in mind, preoperative localization of recurrent disease in a previous operative field may be useful in mitigating morbidity associated with reoperative surgery. Recently, the concept of needle localization typically employed for nonpalpable breast tumors has been extrapolated to head and neck tumors (7-9). In addition, several perioperative techniques to aid in reoperative head and neck surgery have been described in the literature, with varying results (7-14). In this report, we describe our institutional experience utilizing preoperative ultrasound-guided needle localization of recurrent operative bed tumors.
Methods
Patient selection
A retrospective review was conducted on data acquired from institutional review board-approved (Pro20140000469), retrospectively acquired electronic medical records at Robert Wood Johnson University Hospital (New Brunswick, NJ). Patients from 2011–2014 with a diagnosis of thyroid carcinoma at our tertiary care center were identified with the following inclusion criteria: history of thyroidectomy and/or neck dissection, non-palpable disease on surveillance suspicious for recurrence, and ultrasound-guided needle localization before resection of recurrent disease in a previous operative field. Perioperative data and outcomes were analyzed.
Needle localization technique
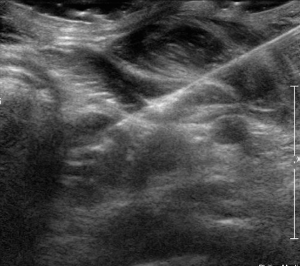
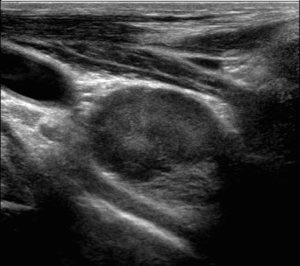
Suspicious nodes or masses were located preoperatively by ultrasound using a standard high-resolution linear transducer 2D probe. Following standard prep and 1% lidocaine for local anesthesia, needle localization was performed under ultrasound guidance with the occasional assistance of CT in the setting of complex anatomy and vessels. The external portion of the needle was removed, and the wire was secured to the skin with gauze. Figures 1,2 depict ultrasound images of this technique.
Follow-up and surveillance
Patients were scheduled to return for postoperative office visits within 1 to 2 weeks after discharge. Follow-up thyroglobulin and anti-thyroglobulin antibody levels were obtained anywhere from approximately three months to 1 year postoperatively. Surveillance imaging with ultrasound was performed yearly at a minimum.
Results
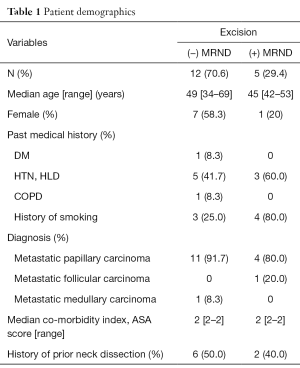
Seventeen patients with a history of total thyroidectomy were identified, of whom five patients underwent concurrent modified radical neck dissection (MRND). Table 1 illustrates the patient demographics separated into the two groups (plus or minus MRND). Fifteen of the patients carried a diagnosis of metastatic papillary carcinoma (88%). All of the patients underwent preoperative fine needle aspiration diagnostic of metastatic carcinoma in 16 of the patients (94%). Median mass size was 1.2 cm (range, 0.3–4.3 cm). The median age was 46 years, and 47% of the cohort was female.
Full table
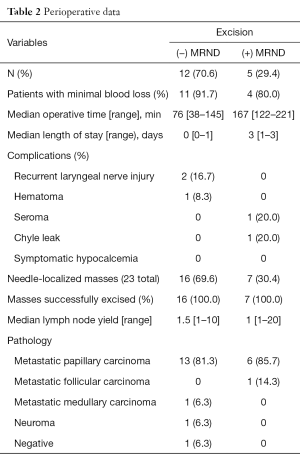
Perioperative data are shown in Table 2. All patients underwent excision of needle-localized operative bed masses, with 5 patients (29%) undergoing an additional MRND. All patients received intraoperative nerve monitoring. Twenty-three needle-localized operative bed masses, 9 (39%) of which were recurrences from previous MRND, were excised successfully. In one patient, the needle was placed through the left internal jugular vein; however, the vein was primarily repaired intraoperatively without major blood loss. Blood loss ranged from minimal (in 88% of patients) to 250 mL. The recurrent laryngeal nerve was involved with tumor in six patients. Two patients experienced new postoperative hoarseness. In these two patients, the tumor was noted intraoperatively to surround the nerve circumferentially and appeared intact after resection, but did not demonstrate activity on nerve monitoring at the end of the operation. Both patients underwent postoperative laryngoscopy demonstrating true vocal cord paralysis and subsequently underwent vocal cord medialization thereafter. Of the two patients, one underwent permanent medialization, while the other underwent temporary medialization. Both patients have experienced improvement in their symptoms, with the latter patient making a full recovery. No patients experienced postoperative hypocalcemia. Other complications included a self-resolving hematoma, a seroma aspirated in the office, and a self-resolving chyle leak. The patients who experienced the latter two complications underwent concurrent MRND at the time of the excision of recurrent thyroid bed disease. All of the preoperatively localized masses were excised successfully.
Full table
Overall, median follow-up was 558 days, with all patients undergoing surveillance ultrasound at least yearly. Papillary and follicular carcinoma patients underwent surveillance thyroglobulin level testing at various times, often beginning within one month postoperatively. All of the patients who underwent MRND in addition to resection of thyroid bed recurrence demonstrated decreasing or undetectable thyroglobulin levels and did not receive postoperative I-131 ablation therapy. Of the patients who underwent excision of recurrent disease without MRND, 2 (16.7%) experienced an increase in postoperative thyroglobulin level and were treated with administration of I-131 ablation therapy. A total of 6 patients (50%) in this group underwent postoperative I-131 ablation therapy. One patient was diagnosed with additional disease in the contralateral supraclavicular node. No local recurrences have been detected in the entire cohort, and 16 of the patients remain with no evidence of disease.
Discussion
Reoperative surgery presents a challenging task in the surgical management thyroid cancer. Morbidity such as recurrent laryngeal nerve injury and hypoparathyroidism in patients undergoing completion thyroidectomy are markedly increased when compared to initial surgery, with permanent complication rates approaching 4% (4). Although patients most commonly undergo reoperative surgery for completion thyroidectomy, an even more challenging endeavor is reoperative surgery for metastatic disease. With metastatic papillary thyroid carcinoma, recurrent disease most commonly occurs in the cervical lymph nodes, but soft tissue recurrences can occur in the thyroid bed/operative field as well (15).
In a series of twenty patients undergoing reoperative central compartment dissection by Kim et al., 20% experienced transient hypocalcemia postoperatively (6). Onkendi et al. reported on a series of 410 patients undergoing a variety of reoperations including removing thyroid bed recurrences and cervical lymphadenectomies, with an overall complication rate of 17% (15). Certainly, morbidity remains high, even in high-volume centers. As a result, the development of novel approaches to aid in operative management of recurrent thyroid cancer can potentially impact morbidity.
Binyousef et al. analyzed outcomes in patients undergoing regional neck dissection for recurrent papillary carcinoma before and after implementing routine preoperative ultrasound neck mapping (10). The authors found that in patients who underwent routine preoperative ultrasound neck mapping, significantly fewer demonstrated signs of recurrent disease postoperatively. Thus, the authors concluded routine preoperative ultrasound to be associated with improved oncological outcomes. McCoy et al. compared outcomes in patients with recurrent papillary carcinoma before and after the implementation of same-day preoperative ultrasound guidance as an adjunct to existing studies in operative planning (11). In 50% of patients in the first group, the surgeon was unable to find the preoperatively imaged lesion, while 100% of patients in the second group underwent successful resection of the lesion, with no change in mean operative time and lower morbidity. Other preoperative adjuncts including methylene blue dye injection and radio-guided surgery have been described, with varying results (12-14).
Recently, the concept of needle localization typically employed for nonpalpable breast tumors has been extrapolated to head and neck tumors. Duprez et al. reported on eight patients with recurrent papillary thyroid cancer in nonpalpable cervical lymph nodes who underwent preoperative hook-needle insertion under ultrasound guidance the morning before surgery (7). There were no complications from the hook-needle insertion, and metastatic lymph nodes were found on final pathology in all eight cases. There have been case reports of successful resection of recurrent cervical and paratracheal disease using ultrasound-guided needle localization in the operating room as well (8,9).
In this study, we report the largest-known cohort of patients who underwent preoperative ultrasound-guided needle localization in patients with non-palpable recurrent disease after surgery for thyroid carcinoma. We demonstrate that preoperative needle localization can be applicable to non-palpable recurrent disease in both the thyroid bed and in the cervical compartments, and that it can be performed potentially safely and effectively. We do recognize the fact that two patients experienced postoperative hoarseness, but this can be attributed to the circumferential involvement of the nerve with tumor. To date, we have not observed any evidence of local recurrence in any of these patients.
We acknowledge several limitations of this study. Its retrospective design introduces selection bias, and precludes the ability to determine a causal relationship with outcomes based on this technique. In addition, our study contains a small cohort of patients, and one surgeon performed all of the operations. As a result, these characteristics limit the generalizability of our findings and our ability to perform meaningful statistical analyses.
In conclusion, preoperative ultrasound-guided needle localization in patients with non-palpable recurrent thyroid carcinoma is a potentially safe and effective technique that can help mitigate the morbidity associated with re-operative thyroid surgery. Future prospective clinical investigations are needed to further investigate this technique as a preoperative aid.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: A retrospective review was conducted on data acquired from institutional review board-approved (Pro20140000469), retrospectively acquired electronic medical records at Robert Wood Johnson University Hospital (New Brunswick, NJ).
References
- Institute NC. SEER Stat Fact Sheets: Thyroid Cancer. Available online: http://seer.cancer.gov/statfacts/html/thyro.html
- Palme CE, Waseem Z, Raza SN, et al. Management and outcome of recurrent well-differentiated thyroid carcinoma. Arch Otolaryngol Head Neck Surg 2004;130:819-24. [Crossref] [PubMed]
- Mazzaferri EL, Kloos RT. Clinical review 128: Current approaches to primary therapy for papillary and follicular thyroid cancer. J Clin Endocrinol Metab 2001;86:1447-63. [Crossref] [PubMed]
- Lefevre JH, Tresallet C, Leenhardt L, et al. Reoperative surgery for thyroid disease. Langenbecks Arch Surg 2007;392:685-91. [Crossref] [PubMed]
- Chao TC, Jeng LB, Lin JD, et al. Reoperative thyroid surgery. World J Surg 1997;21:644-7. [Crossref] [PubMed]
- Kim MK, Mandel SH, Baloch Z, et al. Morbidity following central compartment reoperation for recurrent or persistent thyroid cancer. Arch Otolaryngol Head Neck Surg 2004;130:1214-6. [Crossref] [PubMed]
- Duprez R, Lebas P, Marc OS, et al. Preoperative US-guided hook-needle insertion in recurrent lymph nodes of papillary thyroid cancer: a help for the surgeon. Eur J Radiol 2010;73:40-2. [Crossref] [PubMed]
- Fletcher AM, Preston TW, Kuehn DM, et al. Ultrasound-guided needle localization of recurrent paratracheal thyroid cancer. Ann Otol Rhinol Laryngol 2009;118:475-8. [Crossref] [PubMed]
- Zimmerman P, DaSilva M, Izquierdo R, et al. Intraoperative needle localization during neck reexploration. Am J Surg 2004;188:92-3. [Crossref] [PubMed]
- Binyousef HM, Alzahrani AS, Al-Sobhi SS, et al. Preoperative neck ultrasonographic mapping for persistent/recurrent papillary thyroid cancer. World J Surg 2004;28:1110-4. [Crossref] [PubMed]
- McCoy KL, Yim JH, Tublin ME, et al. Same-day ultrasound guidance in reoperation for locally recurrent papillary thyroid cancer. Surgery 2007;142:965-72. [Crossref] [PubMed]
- Ryan WR, Orloff LA. Intraoperative tumor localization with surgeon-performed ultrasound-guided needle dye injection. Laryngoscope 2011;121:1651-5. [Crossref] [PubMed]
- Travagli JP, Cailleux AF, Ricard M, et al. Combination of radioiodine (131I) and probe-guided surgery for persistent or recurrent thyroid carcinoma. J Clin Endocrinol Metab 1998;83:2675-80. [Crossref] [PubMed]
- Salvatori M, Rufini V, Reale F, et al. Radio-guided surgery for lymph node recurrences of differentiated thyroid cancer. World J Surg 2003;27:770-5. [Crossref] [PubMed]
- Onkendi EO, McKenzie TJ, Richards ML, et al. Reoperative experience with papillary thyroid cancer. World J Surg 2014;38:645-52. [Crossref] [PubMed]