Bupivacaine application reduces post thyroidectomy pain: Cerrahpasa experience
Introduction
Thyroid surgery evolved rapidly with the help of the advanced equipment and increased experience of the surgeons. Even though the size of skin incision and operating time has been minimized currently, postoperative neck pain persists, especially in the early post-operative period (1). Treatment of post-operative pain is essential and the primary target is to improve the post-operative comfort and to decrease hospital stay after surgery (2). However, no standard protocol has been described for the management of post-operative pain after thyroidectomy.
Local anesthetics have been used in pain management after various types of operations with different doses via different routes (3). Despite conflicting outcomes of these studies, administration of local anesthetics into the surgical wound has demonstrated promising results in neck surgery (3). A long-acting local anesthetic, bupivacaine, is easily used by infiltrating intra-incisional and decreases the post-operative pain effectively (3,4). Also, administration of local anesthetics or opioid through chest-tube or drainage placed between pleural sheets is used for the management of pain in chest-tube placed patients and patients to whom thoracotomy was performed (5,6). With administration of the drug through drainage, the amount of used drug is reduced in addition to avoidance of systemic effects. Additionally, it shortens hospital stay and the postoperative period till mobilization (5,6). In our study, we aimed to evaluate the impact of bupivacaine administered into the surgical field after total thyroidectomy on post-operative pain and analgesic requirement with a prospective randomized study.
Methods
The study was performed between 1 January 2010 and 1 January 2011 at Istanbul University, Cerrahpasa Medical Faculty, Department of General Surgery, Division of Endocrine Surgery. The study was approved by institutional ethics board of Istanbul University, Cerrahpasa Medical Faculty (No. 19178) and written informed consent was obtained from all patients. Patients with a history of previous thyroid disease, neck operations, hypersensitivity to the drugs that were used in the study protocol, spinal bone disease, depression, kidney, heart, liver, and pulmonary failure, patients with American Society of Anesthesiologists (ASA) score III or higher, sedative, steroid, and chronic analgesic drug users, alcoholics, patients with expected airway difficulty according to Mallampati scores, patients with mental retardation, and who could not ascertain pain scoring with the visual analog score (VAS) were excluded from the study (7).
VAS evaluation scale [from 0 (painless) to 10 (worst possible)] was introduced to the patients before surgery and the patients who were suitable and compatible with the formation of the study were included.
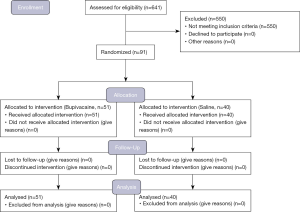
Six hundred forty-one consecutive patients underwent thyroidectomy in the study period. Hundred and thirty-five of patients were re-operation for thyroid diseases, 312 were ASA score III or higher, 103 had Mallampati 3 or 4. Those patients were excluded from the study and remaining 91 patients were included in the study. CONSORT diagram of the patients is given in Figure 1.
Patients were randomly divided into two groups before the surgery with random number generator to receive either bupivacaine or 0.9% NaCl solution in the same volume. All patients were anesthetized and operated with the same anesthesia and surgical team. All patients underwent sutureless total thyroidectomy as described in the literature (8).
Procedure of general anesthesia
After the patient was taken into the operation room, a 20 G cannula was inserted into a superficial vein on the dorsum of the left hand and an infusion of “Isolyte-S” (sodium 140 mEq/L; potassium 5 mEq/L, magnesium 3 mEq/L; chloride 98 mEq/L; acetate 27 mEq/L, gluconate 23 mEq/L. Eczacibasi Baxter, Istanbul, Turkey) at a rate of 5 mL/kg/h was started and 0.02 mg/kg midazolam for premedication was given intravenously.
Monitoring consisted of continuous measurements of the electrocardiogram, heart rate, non-invasive arterial blood pressure (systolic, diastolic, mean), peripheral arterial saturation by pulse oximetry, end-tidal pCO2 (ETCO2), axillary temperature and inhaled anesthetic gas concentration (Datex S5 monitor, USA). Anesthesia was induced with intravenous (IV) 2 mg/kg propofol and 1 µg/kg fentanyl, and orotracheal intubation was facilitated with 0.6 mg/kg rocuronium. Female and male patients were intubated with 7.5 ID and 8.0 ID tubes respectively. Patients were ventilated with 40% oxygen in the air at a frequency of 12 breaths/min and tidal volume was adjusted to maintain ETCO2 at 4.6–5.3 kPa (tidal volume =8 mL/kg, positive end-expiratory pressure =5 cmH2O, peak airway pressure =15–20 cmH2O). Anesthesia was maintained with sevoflurane at an inspiratory concentration of 2–3% (1–1.5 MAC). A second bolus dose, 50 µg of fentanyl, was given immediately before skin incision. Additional boluses of 50 µg fentanyl were given if the heart rate increased by more than 30%. No anti-edema therapy was given during the operation. After completion of surgical procedure, following 1 mg atropine, 2 mg neostigmine was given IV when spontaneous breathing returned, and the patients were extubated when respiration was adequate.
Before extubation, a registered nurse who knew the distribution of the randomization, prepared 10 mL of bupivacaine 0.5% [(5 mg/mL), Marcaine, Astra-Zeneca, Istanbul, Turkey] or 0.9% NaCl (Eczacibasi Baxter, Istanbul, Turkey) solution in order to be administered through the minivac drain after completion of total thyroidectomy procedure and delivered to the surgical team without exposing the used drug. The control group who did not receive bupivacaine received 0.9% NaCl. The minivac drain was occluded for 15 minutes after injecting the prepared solution. Patients, the surgeon who performed the operations and the anesthetists who intubated, monitored patients during surgery and recorded VAS score were also blind to which patient received bupivacaine or 0.9% NaCl.
Prior to the end of surgery, 15–30 minutes before the estimated extubation time, 4 mg ondansetron was administered to all patients in both groups intravenously to prevent postoperative nausea.
After the extubation, all patients were sent to postoperative care unit for routine care. Postoperative pain was evaluated at post-operative minute one (at operation room), five, thirty and hour one in postoperative care unit. The patients whose scores were 9 or higher according to modified Aldrete’s criteria (9) were sent to their rooms. Post-operative pain was further scored at post-operative hour 8 and 24. No analgesia had been performed to the patients unless they requested. Paracetamol IV 1 gram was given as a rescue analgesic.
Postoperative nausea was assessed by a nausea scale between 0–3 (0: no postoperative nausea, 1: mild nausea with no need for treatment, 2: moderate-severe nausea that is responsive to treatment, 3: severe nausea that is resistant to treatment with vomiting). If patients expressed scores 2 or above, an additional 4 mg ondansetron was administered intravenously. The assessment was done by the same postoperative care unit nurse at the 30th minute, 1st, 6th and 12th hours.
The investigators received the data when the study was finished.
Statistics
Power analysis was done before the study to determine the minimum patient number. The sample size, n=36, in each group was required based on 50% reduction in pain score with an α error of 0.05 and a β of 80%. Comparisons of the bupivacaine and control groups were performed using chi-square or Fisher’s exact tests with respect to categorical data and using the Wilcoxon’s rank sum test with respect to quantitative data. Parametric data was reported as mean ± standard deviation and nonparametric data as numbers. Statistical significance was established at P<0.05 for all results.
This study is recorded in Clinical Trials Protocol Registration System (NCT02981095).
Results
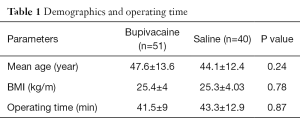
Ninety-one patients (71 females, 20 males) were included in the study. Fifty-one patients received bupivacaine, and 40 patients received 0.9% NaCl solution. No patient dropped out of the study during the procedures. Preoperative diagnoses were multinodular goiter (n=57), malignancy (n=21), thyrotoxicosis (n=8), and substernal goiter (n=5). Solely total thyroidectomy was performed in all patients and no lymph node dissection was performed. No mortality was encountered during the study and in the short-term postoperative period. Mean age, body mass index, and operating time of the patients were comparable between both groups (Table 1).
Full table
No significant difference was observed between the two groups in terms of the fentanyl doses used intraoperatively.
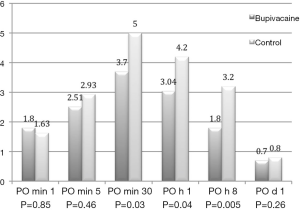
While the VAS scores of the two groups were similar at first (1.8±3.02 vs. 1.63±2.41; P=0.85) and fifth minute (2.51±3 vs. 2.93±2.97; P=0.46) after surgery, they were significantly lower in the bupivacaine administered group at post-operative 30 minutes (3.7±3.2 vs. 5±2.9; P=0.03), hour one (3.04±2.4 vs. 4.2±2.8; P=0.04), and hour eight (1.8±2.04 vs. 3.2±2.1; P=0.005) (Figure 2). The VAS scores were at similar levels on the following day of surgery (0.7±1.5 vs. 0.8±1.2; P=0.26) in both bupivacaine, and 0.9% NaCl received patients. Thirteen patients required rescue analgesia with IV paracetamol during their postoperative hospital stay in the bupivacaine group while this number was twenty-two in the control group (P=0.005). Each patient in both groups needed no more than one dose rescue analgesic in the postoperative period.
One patient in the bupivacaine and two patients in the control group vomited after surgery (P=0.58). Nausea scores at 30th minute were 0.5±0.7 and 0.5±0.8 respectively in bupivacaine and control groups (P=0.83). Nineteen patients in bupivacaine group and 14 patients in the control group experienced nausea (37% and 35% respectively). Of these patients with nausea, four patients in bupivacaine group and three patients in control group needed an additional dose of ondansetron in the postoperative care unit (P=0.67). No patient in both groups needed additional ondansetron dose after they were released from the postoperative care unit or had nausea score above one. Transient hypocalcemia developed in one patient in each group (P=1). Transient hoarseness was observed in one patient in the control group (P=0.44) and post-operative bleeding developed in one patient in the bupivacaine group (P=1).
Discussion
Most of the studies concerning this issue had similar outcomes, but none of them evaluated the application of bupivacaine directly into the surgical field. Additionally, the necessity of analgesia after total thyroidectomy had never been questioned in settings of bupivacaine administration via the drain. Preventing any major nerves from the paralytic effect of the bupivacaine could be one of the reasons why surgeons do not prefer to apply bupivacaine directly into the surgical field after total thyroidectomy. However, as it is shown in our study bupivacaine has no paralytic effects on laryngeal nerves even if it is infused to the surgical field through drain because bupivacaine sustains analgesia without motor blockage in low concentrations, has higher potency and well tissue penetration with an effecting time of 5–16 hours (10,11). This could be an advantage for the direct application of bupivacaine when compared to the subcuticular injection since it presents easy application, and there are no aesthetic concerns that could occur due to diminished suture line formation after subcuticular or trans-incision injection (3,4). In our study, at first and fifth minutes, VAS scores of the patients between bupivacaine and control group are similar while it is significantly lower in bupivacaine administered group at postoperative minute 30, hour 1 and hour eight. This significance of late pain relief may be due to the delayed effect of bupivacaine due to its pharmacokinetics.
Wound incision is one of the origins of post-operative pain. Hyperextension of the neck, retraction of the wound edges during surgery and dissection itself are the other causes of post-operative pain. Infusion of bupivacaine into the surgical field could also be effective against post-operative pain related to the factors mentioned above. In our post-operative pain assessment with the VAS, bupivacaine administration through the drain into the surgical field reduced VAS scores during the patients completely awoke after surgery on postoperative day one. In our study, thirteen patients required analgesia during their postoperative hospital stay in the bupivacaine group while this number was twenty-two in the control group. When the patients were retrospectively investigated, these cases have low tolerances to pain also before the surgery.
The second important finding of our study was the reduced postoperative analgesia requirement of the patients who underwent total thyroidectomy after bupivacaine treatment during their post-operative hospital stay. Surgeons and anesthesiologists have spent great effort in reducing post-operative pain and its management to increase the comfort of the patients after surgery. Minimally invasive techniques and patient controlled anesthesia are the major ones of these approaches. Since total thyroidectomy is a short-stay procedure, reduction of postoperative pain becomes a major issue that needs to be relieved during the post-operative hospital stay. Various types of non-steroid anti-inflammatory drugs were assessed for this issue; ketorolac is one of them (12). In Western literature, there are some intra-operative modifications that also tried to reduce post-operative pain after thyroid surgery: It has been reported that remifentanil was associated with a significant increase in immediate postoperative pain and the length of stay in the post-operative intensive care unit (4). Opioid based analgesia was one of their parameters. We never use opioid-based analgesia after uncomplicated total thyroidectomy procedures.
Bilateral superficial cervical plexus block, greater occipital nerve block, and patient controlled analgesia are the other techniques that were evaluated after thyroid operations about their benefit for post-operative pain control (12,13). The superiority of sublingual buprenorphine to wound infiltration of bupivacaine before skin closure was proven with a prospective study (4), but the action mechanism and application routes of both drugs are completely different, and only wound infiltration of bupivacaine could be insufficient to reduce the pain arising from neck and dissection area. All the additional pain management techniques showed 24-hour post-operative effectivity (14). We observed the same results in our study, but most of the patients are discharged from hospital on postoperative day one after thyroid surgery. This is the real issue limiting the further pain assessment for investigative purposes. However, it is probably not necessary since patients’ pain complaints are significantly low at the day following thyroid surgery. Additionally, the bilateral superficial block is insufficient for pain relief by itself (15). Post-operative analgesia was not required in 74.5% of the patients treated with bupivacaine in our study. Another advantage of bupivacaine could be the cost advantage compared to other techniques or drugs such as patient controlled analgesia or opioid use after thyroidectomy for pain management. However, this hypothesis needs to be verified by further studies. Our study was also comparable with the others in terms of occurrence of transient hypocalcaemia, hoarseness, and post-operative bleeding (16).
The rate of postoperative nausea and vomiting (PONV) was 37% and 35% in our groups. There are studies stating that PONV rate after thyroidectomy can exceed 80%. However, no prophylactic antiemetic agents were used in those studies (17). In our study, we have used prophylactic antiemetics, and the PONV rate is similar to the studies where prophylaxis was used (18).
Limitation of this study is not being designed in a comparative manner with another analgesic. In addition, patient number could be higher but as a tertiary referral center, most of our patients were recurrent thyroid cancer or goiter patients that we should exclude from the study.
Conclusions
We observed that bupivacaine administration via drain into the surgical field after total thyroidectomy reduces pain and analgesic requirement.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The study was approved by institutional ethics board of Istanbul University, Cerrahpasa Medical Faculty (No. 19178) and written informed consent was obtained from all patients.
References
- Youssef T, Mahdy T, Farid M, et al. Thyroid surgery: use of the LigaSure Vessel Sealing System versus conventional knot tying. Int J Surg 2008;6:323-7. [Crossref] [PubMed]
- Ruggieri M, Straniero A, Pacini FM, et al. Video-assisted surgery of the thyroid diseases. Eur Rev Med Pharmacol Sci 2003;7:91-6. [PubMed]
- Bagul A, Taha R, Metcalfe MS, et al. Pre-incision infiltration of local anesthetic reduces postoperative pain with no effects on bruising and wound cosmesis after thyroid surgery. Thyroid 2005;15:1245-8. [Crossref] [PubMed]
- Lacoste L, Thomas D, Kraimps JL, et al. Postthyroidectomy analgesia: morphine, buprenorphine, or bupivacaine? J Clin Anesth 1997;9:189-93. [Crossref] [PubMed]
- Demmy TL, Nwogu C, Solan P, et al. Chest tube-delivered bupivacaine improves pain and decreases opioid use after thoracoscopy. Ann Thorac Surg 2009;87:1040-6; discussion 1046-7. [Crossref] [PubMed]
- Schneider RF, Villamena PC, Harvey J, et al. Lack of efficacy of intrapleural bupivacaine for postoperative analgesia following thoracotomy. Chest 1993;103:414-6. [Crossref] [PubMed]
- Mallampati SR, Gatt SP, Gugino LD, et al. A clinical sign to predict difficult tracheal intubation: a prospective study. Can Anaesth Soc J 1985;32:429-34. [Crossref] [PubMed]
- Teksoz S, Arikan AE, Erbabacan SE, et al. Effect of total thyroidectomy on mortality and morbidity in geriatric patients: Cerrahpasa experience. Turk Geriatri Dergisi 2014;16:365-71.
- Aldrete JA. The post-anesthesia recovery score revisited. J Clin Anesth 1995;7:89-91. [Crossref] [PubMed]
- Kuan YM, Smith S, Miles C, et al. Effectiveness of intra-operative wound infiltration with long-acting local anaesthetic. ANZ J Surg 2002;72:18-20. [Crossref] [PubMed]
- Kayhan Z. editor. Clinical Anesthesia. Ankara: Logos Yayıncılık, 2004.
- Kim SY, Kim EM, Nam KH, et al. Postoperative intravenous patient-controlled analgesia in thyroid surgery: comparison of fentanyl and ondansetron regimens with and without the nonsteriodal anti-inflammatory drug ketorolac. Thyroid 2008;18:1285-90. [Crossref] [PubMed]
- Eti Z, Irmak P, Gulluoglu BM, et al. Does bilateral superficial cervical plexus block decrease analgesic requirement after thyroid surgery? Anesth Analg 2006;102:1174-6. [Crossref] [PubMed]
- Shih ML, Duh QY, Hsieh CB, et al. Bilateral superficial cervical plexus block combined with general anesthesia administered in thyroid operations. World J Surg 2010;34:2338-43. [Crossref] [PubMed]
- Dieudonne N, Gomola A, Bonnichon P, et al. Prevention of postoperative pain after thyroid surgery: a double-blind randomized study of bilateral superficial cervical plexus blocks. Anesth Analg 2001;92:1538-42. [Crossref] [PubMed]
- Lee J, Na KY, Kim RM, et al. Postoperative functional voice changes after conventional open or robotic thyroidectomy: a prospective trial. Ann Surg Oncol 2012;19:2963-70. [Crossref] [PubMed]
- Sonner JM, Hynson JM, Clark O, et al. Nausea and vomiting following thyroid and parathyroid surgery. J Clin Anesth 1997;9:398-402. [Crossref] [PubMed]
- Shilpa SN, Shailaja S, Hilda SS. Comparison of Efficacy of Clonidine versus Ondansetron for Prevention of Nausea and Vomiting Post Thyroidectomy: A Double Blind Randomized Controlled Trial. J Clin Diagn Res 2015;9:UC01-3. [PubMed]