Are there disparities in the presentation, treatment and outcomes of patients diagnosed with medullary thyroid cancer?—An analysis of 634 patients from the California Cancer Registry
Introduction
Racial and healthcare-related disparities are well described in the cancer literature with underserved minorities frequently presenting with increased stage of disease (1,2) and having a disproportionately increased mortality for a broad range of cancer types. Harari and colleagues have published that Black patients and those with a low socioeconomic status (SES) have worse outcomes for differentiated thyroid cancers than their White and higher SES counterparts (3). Similarly, Keegan et al. recently published that adolescents and young adult patients residing in low SES neighborhoods and nonmetropolitan areas had worse thyroid cancer-specific survival among adolescent and young adult men (4). Although the underlying cause of these racial and SES disparities are unknown, the etiology is thought to include differences in exposures and access to medical care (5).
Medullary thyroid cancer (MTC) is an uncommon malignancy accounting for 1–2% of thyroid cancers in the United States (6). The management of MTC is challenging because MTC has a high rate of lymph node metastasis even with early stage tumors (7,8) and its neuroendocrine origin does not allow for the use of radioactive iodine. Small lymph nodes in the central neck are difficult to detect on both preoperative imaging and intraoperatively (9) and therefore both the American Thyroid Association and the National Comprehensive Cancer Network (NCCN) recommend that almost all patients with MTC undergo a total thyroidectomy with central lymph node dissection (CLND) (10).
Racial, socioeconomic and gender disparities in the presentation, management and outcomes for MTC have not been adequately investigated. The California Cancer Registry (CCR) is a statewide population-based cancer surveillance system that captures all cancer diagnosis and management in one of the most diverse regions within the United States and is well suited to study rare cancers such as MTC. We hypothesized that minorities and those with a low SES present with more advanced disease, have a less adequate surgical resection and have worse outcomes for MTC than white patients or higher SES patients.
Methods
This was a retrospective, cross-sectional analysis of patients diagnosed with MTC in California. The CCR is the single largest population-based cancer registry in the US and contains demographic, diagnostic, treatment and outcome information extracted from medical records for every reportable cancer diagnosed among residents of the state since 1988. By law (Health and Safety Code, Section 103885), all new cancer cases diagnosed in California residents since January of 1988 have been reported to the CCR, with strict guidelines to maintain patient confidentiality. To ensure current follow up for vital status and cause of death, the CCR database is linked annually to death certificates, hospital discharge data, Medicare files, the Department of Motor Vehicles and other administrative databases. Linkage to the National Death Index ensures capture of deaths occurring outside California. The CCR is part of both the Centers for Disease Control National Program of Cancer Registries and the National Cancer Institute Surveillance Epidemiology and End Results (SEER) program and meets or exceeds the standards of both groups for data quality and completeness.
We identified patients diagnosed with MTC (M8510) between 1988 and 2011 through the CCR. We collected data on patient demographics (age, gender, race, SES), tumor characteristics (summary stage at diagnosis and tumor size), and first course treatment (whether or not patients received thyroidectomy and lymph nodes dissection). Patients who had five or more nodes removed were classified as having CLND. Patients were followed through December 31, 2011, and only patients for whom MTC was the first or only cancer diagnosis were included. Patients diagnosed at autopsy were excluded from analysis.
Race/ethnicity in the CCR is based on information collected from medical records supplemented with linkage to algorithms to better identify Hispanics and Asian/Pacific Islanders. We categorized race/ethnicity as non-Hispanic white, non-Hispanic black, Hispanic, and non-Hispanic Asian/Pacific Islander (API). Neighborhood SES was based on U.S. Census characteristics combined into the summary Yost index (11), and was categorized into quintiles. Stage at diagnosis was defined based on the SEER summary stage, and it was categorized as localized, regional, and remote/metastatic disease.
Patient demographic and tumor characteristics, and first course of treatment were summarized using descriptive statistics. ANOVA was used to determine if the distributions of tumor size were significantly different by gender, race, or SES. Ordinal multivariable logistic regression was used for predictors of the tumor stage. Generalized linear regression model with Poisson distribution was used for potential predictors of number of lymph nodes examined, and binomial distribution was used for predictors of receipt of surgical treatment. The overall survival function was estimated using Kaplan-Meier method within gender, race, and SES. Log-rank tests were conducted to examine whether the unadjusted differences in survival patterns between the groups were statistically significant. Multivariable Cox proportional hazards regression analysis was performed for overall and cause-specific survival analysis with gender, race, and SES as the main predictors of interest, adjusting for patient’s age, tumor stage, and receipt of radiation and surgical therapy. We considered P values less than 0.05 as statistically significant. All statistical analyses were conducted using SAS for Windows, version 9.3 (SAS Institute Cary, NC, USA). Ethics approval was not obtained as this study used existing data collections containing only non-identifiable data about human beings.
Results
We identified 634 patients with MTC diagnosed between 1988 and 2011. Forty-one percent (51%) were under age 50 years at diagnosis and 58% were women. The majority of patients were non-Hispanic white (60%); but 25% were Hispanic, 8% API and 6% black. Patients were distributed evenly across quintiles of SES. Almost everyone (85%) underwent thyroidectomy with 50% having a CLND (Table 1).

Full table
Characteristics of patients by tumor size, stage at diagnosis and surgical treatment are shown in Table 2. There were no statistically significant differences by age, race or SES in mean tumor size or the proportion of patients diagnosed with localized disease, but men were diagnosed with larger tumors than women and women were much more likely to be diagnosed at a localized stage. Younger patients and women were more likely to be treated with a thyroidectomy. There were no statistically significant differences in surgical treatment by race or SES. Patients aged 65 and older were less likely than younger patients to receive a CLND.
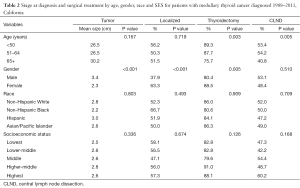
Full table
Predictors of overall and cause specific survival are shown in Table 3. There were no statistically significant differences in survival by gender or race. Patients over age 50 had poorer survival than younger patients. Patients in the highest SES category had a better overall survival, but not disease specific survival, than those in the lowest SES (HR =0.3, CI =0.1–0.7). Patients treated with thyroidectomy had better overall and cause specific survival, but the effect of CLND was not statistically significant after adjustment for other factors.

Full table
Discussion
Many population groups across the United States suffer disproportionately from a variety of cancers. Since the Institute of Medicine 2002 report entitled Unequal Treatment: Confronting Racial and Ethnic Disparities in Health Care (12) major organizations such as the American Medical Association and the National Institute of Health have launched initiatives to address disparities in health care; however, most of the previous research on health care disparities has focused on common cancers such as breast, lung, colon and prostate. The scope of the problem in uncommon cancers, such as MTC, has not been adequately studied probably because of the inherent difficulties in accumulating enough data on rare cancers. Our study, using a population-based cancer surveillance system from a diverse population, suggests that although patients with MTC in the highest SES had a better overall survival than patients in the lowest SES, the stage at presentation and treatment did not differ between different racial/ethnic groups or across SES classifications.
One concern in health care disparities research has been the access to quality care by persons from minority groups and low SES. It is theorized that lack of access to quality care may lead to delayed diagnosis (2) and delayed treatment of life threatening conditions. Although we hypothesized this may be true in MTC, our study suggests otherwise. Patients across all race groups and SES categories presented with similar size tumors and the same incidence of regionally metastatic and distant metastatic disease. This differs from the findings of Harari et al. who previously reported that racial minorities presented with more advanced differentiated thyroid cancer than their white counterparts. The disparities in that study were independent of SES, age, gender or insurance status (3). The reason we did not observe this association is likely multifactorial. The average tumor size in our study was fairly large at 2.6 cm. In our experience, when a tumor reaches this size it is often visible and palpable. The finding of a neck mass of this size immediately raises concern in the patient and practitioner for a cancer, which facilitates an evaluation using fine-needle aspiration. This is different from the more common differentiated cancers described by Harari et al., which are frequently found incidentally on imaging studies done for another reason. Therefore, persons who have access to high quality care are probably more likely to have incidental differentiated cancers discovered and evaluated while they are small.
The role of a CLND in patients with MTC has been debated in the past three decades. Because of the early spread to MTC to the regional lymph nodes and the lack of effective adjuvant treatments for MTC, most experts agree that a CLND is warranted at the time of thyroidectomy (9). We hypothesized that racial minorities and patients in a low SES group would be less likely to have a CLND because of lack of access to a high-volume thyroid surgeon. We did not find this to be true. However we did find that only 47% of patients with MTC had a CLND. This is concerning that perhaps a significant proportion of the population is getting less than the recommended treatment of MTC regardless of race or SES. A recent study showed a difference between surgeon volume and outcomes among 106,314 patients of different race and ethnicities undergoing thyroid or parathyroid surgery. Higher surgeon volume was associated with improved patient outcomes, however, African Americans had significantly less access to intermediate- (10–99 cases) and high- (100 cases) volume surgeons compared with Caucasians—45 versus 49%, and 16 versus 19%, respectively (P<0.001) (13). This access to care can be related to geographic distance, insurance coverage, and availability.
We found that patients in the highest SES categories had a better overall, but not cause-specific, survival when compared to those in the lowest SES. One explanation for this is that our patients in the lowest SES groups may have suffered from a disproportionately higher number of comorbidities. Paeratakul et al. described SES as an important predictor of chronic medical conditions such as diabetes, hypertension, and obesity. These conditions likely contribute to a decreased long-term survival (14). In recent studies, lowest income patients with hepatocellular carcinoma were less likely to receive curative treatment (15), non-Hispanic blacks were more likely than non-Hispanic whites to die of ovarian cancer (16), and melanoma patients with low SES suffered increase in mortality rate relative to patients with high SES (17).
We did not see a statistically significant difference in disease specific survival across SES categories or race. We hypothesize that this may be because of the low number of MTC specific deaths. MTC is a slow growing cancer and previous authors have described long-term survival even with residual and recurrent disease (18).
We found that men are more likely to present with larger tumors and are more likely to have regional and distant metastatic disease than women. Our results are similar to those found by Roman et al. who reported that men with MTC had a worse survival (19). The reason for this disparity is unclear, but may be because men are less likely to seek medical care than woman (20). Unexpectedly, despite presenting with more advanced disease, men had a similar probability of getting a CLND and had similar cancer specific and overall survival. These findings are consistent with Kandil et al. who found that lymph node disease confers a poor prognosis in MTC, but the extent of lymph node dissection and the overall number of lymph nodes removed did not improve outcomes (21). This discrepancy has been found to be true in other cancers including rectal cancers and melanoma and may be attributable to patient characteristics, tumor location, and surgical and pathologic technique.
The number of patients and quality of data afforded by the CCR is a major strength in this study. The CCR is California’s statewide population-based cancer surveillance system and has a nearly 100% capture of patients in California with a reportable cancer. With rare cancers such as MTC, large databases such CCR provide valuable information that cannot be obtained from single institution studies. Unfortunately, population database studies do have well known limitations. For example, we do not have any information as whether the MTC listed in the CCR are sporadic or from germ-line mutations. This is potentially limiting as patients with known genetic mutations may have undergone thyroidectomy earlier and therefore influenced their tumor size, lymph node status and survival. Furthermore, we know that patients with RET proto-oncogene mutations can suffer from other comorbidities such as pheochromocytomas which may influence survival. Additionally, we do not have information on other co-morbid conditions prior to the diagnosis of MTC that are not related to the tumor, but may affect overall survival. We also have little information on the type of lymph node resection that was done such and whether lymph nodes were resected prophylactically or because they appeared to harbor metastatic disease. This is a problem inherent in most database studies. As a surrogate for having a CLND we used any patient who had at least five lymph nodes removed. We chose this number, because in our experience it is rare that a patient has five lymph nodes removed incidentally with a thyroidectomy and it is also uncommon that any patient who had a CLND would have less than five lymph nodes removed. Finally we have no information on patients’ preoperative and postoperative calcitonin and CEA levels which could have been used to assess the adequacy of the surgical resection and the recurrence rates in our groups.
In conclusion, we did not find that race, gender or SES influenced the presentation, treatment or outcomes of patients with MTC. Men with MTC present with larger tumors and are less likely to have localized disease. Half of the MTC patients in California do not undergo a CLND at the time of thyroidectomy, which may suggest a lack appropriate care across a range of healthcare systems. Ongoing education of providers should highlight the importance of a CLND in the care of patients with MTC.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: Ethics approval was not obtained as this study used existing data collections containing only non-identifiable data about human beings.
References
- Altekruse SF, Kosary CL, Krapcho M, et al, editors. Surveillance, Epidemiology, and End Results Cancer Statistics Review, 1975-2007. Bethesda, MD: National Cancer Institute, 2010.
- Singh GK, Miller BA, Hankey BF, et al, editors. Area Socioeconomic Variations in U.S.Cancer Incidence, Mortality, Stage, Treatment, and Survival, 1975-1999. NCI Cancer Surveillance Monograph Series, No. 4. Bethesda, MD: National Cancer Institute, 2003.
- Harari A, Li N, Yeh MW. Racial and socioeconomic disparities in presentation and outcomes of well-differentiated thyroid cancer. J Clin Endocrinol Metab 2014;99:133-41. [Crossref] [PubMed]
- Keegan TH, Grogan RH, Parsons HM, et al. Sociodemographic disparities in differentiated thyroid cancer survival among adolescents and young adults in California. Thyroid 2015;25:635-48. [Crossref] [PubMed]
- Ghafoor A, Jemal A, Ward E, et al. Trends in breast cancer by race and ethnicity. CA Cancer J Clin 2003;53:342-55. [Crossref] [PubMed]
- Cancer of the Thyroid Invasive: Trends in SEER Incidence and U.S. Mortality Using the Joinpoint Regression Program, 1975-2011 (SEER). Available online: www.seer.cancer.gov
- Machens A, Hinze R, Thomusch O, et al. Pattern of nodal metastasis for primary and reoperative thyroid cancer. World J Surg 2002;26:22-8. [Crossref] [PubMed]
- Moley JF. Medullary thyroid carcinoma: management of lymph node metastases. J Natl Compr Canc Netw 2010;8:549-56. [PubMed]
- Chan AC, Lang BH, Wong KP. The pros and cons of routine central compartment neck dissection for clinically nodal negative (cN0) papillary thyroid cancer. Gland Surg 2013;2:186-95. [PubMed]
- Wells SA Jr, Asa SL, Dralle H, et al. Revised American Thyroid Association guidelines for the management of medullary thyroid carcinoma. Thyroid 2015;25:567-610. [Crossref] [PubMed]
- Yost K, Perkins C, Cohen R, et al. Socioeconomic status and breast cancer incidence in California for different race/ethnic groups. Cancer Causes Control 2001;12:703-11. [Crossref] [PubMed]
- Nelson A. Unequal treatment: confronting racial and ethnic disparities in health care. J Natl Med Assoc 2002;94:666-8. [PubMed]
- Noureldine SI, Abbas A, Tufano RP, et al. The impact of surgical volume on racial disparity in thyroid and parathyroid surgery. Ann Surg Oncol 2014;21:2733-9. [Crossref] [PubMed]
- Paeratakul S, Lovejoy JC, Ryan DH, et al. The relation of gender, race and socioeconomic status to obesity and obesity comorbidities in a sample of US adults. Int J Obes Relat Metab Disord 2002;26:1205-10. [Crossref] [PubMed]
- Jembere N, Campitelli MA, Sherman M, et al. Influence of socioeconomic status on survival of hepatocellular carcinoma in the Ontario population; a population-based study, 1990-2009. PLoS One 2012;7:e40917. [Crossref] [PubMed]
- Brewer KC, Peterson CE, Davis FG, et al. The influence of neighborhood socioeconomic status and race on survival from ovarian cancer: a population-based analysis of Cook County, Illinois. Ann Epidemiol 2015;25:556-63. [Crossref] [PubMed]
- Fiala MA, Finney JD, Liu J, et al. Socioeconomic status is independently associated with overall survival in patients with multiple myeloma. Leuk Lymphoma 2015;56:2643-9. [Crossref] [PubMed]
- Kebebew E, Ituarte PH, Siperstein AE, et al. Medullary thyroid carcinoma: clinical characteristics, treatment, prognostic factors, and a comparison of staging systems. Cancer 2000;88:1139-48. [Crossref] [PubMed]
- Roman S, Lin R, Sosa JA. Prognosis of medullary thyroid carcinoma: demographic, clinical, and pathologic predictors of survival in 1252 cases. Cancer 2006;107:2134-42. [Crossref] [PubMed]
- Utilization of Ambulatory Medical Care by Women: United States, 1997-98. Centers for Disease Control and Prevention (CDCP). National Center for Health Statistics: Vital and Health Statistics, Series 13, #149: July 2001.
- Kandil E, Gilson MM, Alabbas HH, et al. Survival implications of cervical lymphadenectomy in patients with medullary thyroid cancer. Ann Surg Oncol 2011;18:1028-34. [Crossref] [PubMed]


